
Comprehensive Introduction and Patho-Epidemiology
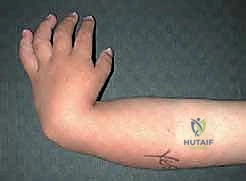
Radial dysplasia, historically referred to as radial clubhand, represents a profound and complex spectrum of longitudinal failure of formation affecting the preaxial border of the upper extremity. This condition is not merely a localized skeletal anomaly; it is a profound multi-tissue deficiency that encompasses osteological, myological, neurological, and vascular hypoplasia or aplasia. The incidence of radial dysplasia is estimated to be between 1 in 30,000 to 1 in 100,000 live births, presenting bilaterally in approximately half of all documented cases. As orthopedic surgeons, our approach to this deformity must be rooted in a deep understanding of its embryological origins, as the insult occurs between the fourth and eighth weeks of gestation, directly impacting the apical ectodermal ridge (AER) and the zone of polarizing activity (ZPA).
The molecular basis of radial dysplasia is increasingly understood through the lens of developmental genetics. Disruptions in the Sonic hedgehog (Shh) signaling pathway, which is crucial for radio-ulnar patterning, and mutations in the Hox gene clusters (specifically Hox 9-13) are heavily implicated in the failure of radial longitudinal growth. The natural history of the untreated severe radial clubhand is one of progressive deformity. Without the supportive strut of the radius, the carpus and hand slide radially and volarly, pivoting around the distal ulnar articular surface. This leads to profound ulnar bowing, a shortened forearm segment, and a severe mechanical disadvantage for the extrinsic flexors and extensors of the hand, severely compromising grip strength and overall upper extremity kinematics.
Surgical intervention is fundamentally aimed at realigning the hand with the forearm axis to optimize the biomechanical advantage of the extrinsic musculature, while simultaneously maximizing functional reach and preserving the growth potential of the distal ulnar physis. The paradigm of treatment has shifted significantly over the decades. While historical approaches favored rigid centralization or radialization—often at the cost of wrist mobility and ulnar physeal arrest—contemporary masterclasses in radial dysplasia reconstruction increasingly emphasize radical soft tissue release, meticulous rebalancing, and sophisticated local tissue transfer, such as the bilobed flap, to accommodate the corrected posture without placing undue compressive forces on the carpus.
The Bayne and Klug Classification: Our Radiographic Compass
To standardize our surgical approach and prognostic discussions, we rely heavily on the Bayne and Klug classification system, which categorizes the anatomical deficiency based on radiographic evaluation. Type I dysplasia is characterized by a deficient distal radial epiphysis; the radius is present but demonstrably shorter than the ulna, leading to mild radial deviation. In these cases, the proximal radial epiphysis is typically normal, and the overall functional deficit is minimal, often precluding the need for major surgical reconstruction.
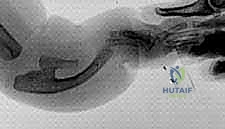
Type II dysplasia involves defective growth in both the proximal and distal epiphyses. The radius is essentially present in miniature. While the radio-ulnar articulation may be somewhat preserved, the length discrepancy becomes more pronounced as the child grows, leading to moderate radial deviation and potential functional impairment. Type III represents a partial absence of the radius, most commonly the distal two-thirds. The wrist is entirely unsupported, leading to significant radial deviation and volar subluxation of the carpus. The proximal radial remnant is often fused to the ulna, creating a complex synostosis that further complicates forearm rotation.
Type IV is the total absence of the radius and is, by far, the most common and severe clinical presentation. The hand is completely unsupported on the radial side, resulting in a perpendicular relationship between the hand and the forearm. The distal ulna is frequently bowed, thickened, and serves as the sole longitudinal skeletal support for the forearm. It is primarily in Type III and Type IV patients that the sophisticated soft tissue releases and bilobed flap reconstructions discussed in this chapter are most rigorously applied.
The Systemic Context: Beyond the Forearm
It is an absolute clinical imperative to recognize that radial dysplasia is rarely an isolated orthopedic event; it is frequently the cardinal musculoskeletal manifestation of a broader, potentially life-threatening systemic syndrome. Before any surgical intervention is contemplated, a comprehensive systemic workup is strictly non-negotiable. Holt-Oram syndrome, an autosomal dominant condition linked to TBX5 gene mutations, presents with radial ray anomalies alongside congenital heart defects, most notably atrial or ventricular septal defects. A preoperative echocardiogram and cardiology clearance are mandatory to prevent catastrophic intraoperative cardiac events.
Thrombocytopenia-absent radius (TAR) syndrome presents a unique clinical and surgical challenge. These patients exhibit bilateral absence of the radius but characteristically preserve their thumbs—a key diagnostic differentiator from other syndromes. The profound thrombocytopenia, which is most severe in the first year of life, poses a massive hemorrhagic risk during any surgical procedure. Hematologic optimization, often requiring platelet transfusions and close coordination with a pediatric hematologist, is a prerequisite for surgical clearance.
Fanconi anemia represents the most insidious associated condition. This rare, autosomal recessive disorder of DNA repair (chromosomal breakage) leads to progressive bone marrow failure, pancytopenia, and a significantly elevated risk of distinct malignancies, including acute myeloid leukemia and squamous cell carcinomas. Diagnosis is confirmed via chromosomal breakage analysis (diepoxybutane test). Furthermore, the VACTERL association (Vertebral, Anal, Cardiac, Tracheoesophageal, Renal, Limb anomalies) necessitates a sweeping evaluation, including renal ultrasounds and spinal radiographs, as undiagnosed renal agenesis or severe tethered cord can drastically alter the patient's surgical candidacy and life expectancy.
Detailed Surgical Anatomy and Biomechanics
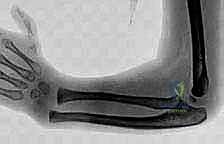
Navigating the forearm in a patient with severe radial dysplasia requires a complete mental recalibration of standard upper extremity anatomy. The standard landmarks and neurovascular planes are profoundly distorted by the failure of preaxial formation and the subsequent mechanical adaptations. Understanding this pathoanatomy is the cornerstone of a safe and effective surgical reconstruction. The skeletal foundation is defined by the absence of the radius and the compensatory hypertrophy and bowing of the ulna. The distal ulnar epiphysis is the singular growth center for the forearm; it is incredibly vulnerable and must be protected at all costs during dissection, as any iatrogenic injury will result in catastrophic cessation of forearm longitudinal growth.
The carpus in a Type IV dysplasia does not articulate with a radius; instead, it frequently articulates directly with the radial aspect of the distal ulna or is entirely displaced, resting against the radial shaft of the ulna. This lack of a stable fulcrum leads to severe biomechanical collapse. The extrinsic flexors and extensors, lacking a stable skeletal axis to pull against, act as bowstrings, further exacerbating the radial deviation and volar flexion of the wrist. The mechanical advantage of these muscles is severely compromised, resulting in profound weakness in grip and pinch strength, even when the digits themselves are fully formed.
Neurovascular Structures: The Unseen Dangers
The neurovascular anatomy in radial dysplasia is notoriously anomalous and presents the highest risk for intraoperative iatrogenic injury. The superficial sensory branch of the radial nerve and the radial artery are almost universally absent or severely hypoplastic. Consequently, the median nerve assumes a critical, expanded role. It frequently bifurcates proximally and sends anomalous branches to supply the sensory territory typically innervated by the radial nerve.
Crucially, the median nerve is displaced from its standard central location. Due to the severe radial deviation and the pull of contracted soft tissues, the median nerve often lies superficially and extremely radially, sometimes positioned directly beneath the subcutaneous tissue in the exact location where a standard radial incision might be placed. It is often tethered by thick fascial bands and can be compressed. Identifying, neurolysing, and protecting the median nerve is the absolute first priority upon opening the deep fascia.
The ulnar nerve and ulnar artery are typically present and functionally intact, but they are subject to significant traction due to the profound ulnar bowing of the forearm. During the surgical correction of the radial deviation, the ulnar neurovascular bundle can become redundant and kinked, or conversely, if the ulna is osteotomized and straightened, these structures can be acutely stretched. Continuous intraoperative assessment of distal perfusion and nerve tension is required throughout the reconstructive process.
Muscular Intervals and Soft Tissue Tethering
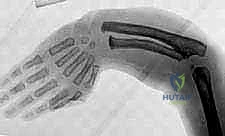
The primary deforming forces in radial dysplasia are the contracted, fibrotic soft tissues on the radial aspect of the forearm and wrist. The most significant of these is the radial anlage, a dense, unyielding fibrocartilaginous band that represents the embryological remnant of the absent radius. This anlage originates proximally near the lateral epicondyle or proximal ulna and inserts distally into the radial carpus. It acts as a massive tether, preventing any passive correction of the deformity and must be completely excised to achieve a neutral wrist position.
In addition to the anlage, the muscular anatomy is highly variable. The brachioradialis, extensor carpi radialis longus (ECRL), and extensor carpi radialis brevis (ECRB) may be absent, fused, or insert anomalously into the carpus or the anlage itself. When present, these radial-sided muscles are severely contracted and contribute to the deforming force. The flexor carpi radialis (FCR) is also frequently contracted.
Conversely, the skin and subcutaneous tissues on the radial side are severely deficient, creating a physical barrier to realignment even after deep structures are released. The skin on the ulnar side, however, is redundant and forms a prominent bursa or skin fold over the prominent distal ulna. This disparity in soft tissue envelope is the precise anatomical rationale for the bilobed flap reconstruction, allowing us to recruit redundant ulnar skin to fill the gaping radial defect created following radical soft tissue release.
Exhaustive Indications and Contraindications
The decision to proceed with surgical reconstruction in radial dysplasia is highly nuanced and must be individualized to the patient's specific anatomical deficit, systemic health, and functional adaptations. The overarching goal of surgery is to improve the biomechanics of the hand, enhance cosmetic appearance, and facilitate better activities of daily living, without compromising the patient's existing compensatory mechanisms.
The ideal candidate is a child between 6 and 18 months of age with a Type III or Type IV radial dysplasia who has undergone extensive preoperative soft tissue stretching. Early intervention takes advantage of the inherent plasticity of the pediatric soft tissues and allows the child to incorporate the corrected limb position into their developing motor cortex patterns. However, surgery is not universally indicated, and a rigorous assessment of contraindications is paramount to avoid converting a functional, albeit deformed, limb into a stiff, non-functional one.
A critical functional consideration is the status of the elbow. Many patients with severe radial dysplasia have limited elbow flexion. In these cases, the severe radial deviation of the wrist is actually a necessary compensatory mechanism that allows the patient to bring their hand to their mouth for feeding and hygiene. Correcting the wrist to a neutral position in a patient with a stiff, extended elbow will disastrously eliminate their ability to reach their face, rendering the surgery an absolute functional failure.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Absolute Indications | Severe Type III or IV Deformity | Profound lack of skeletal support requires realignment for extrinsic muscle function. |
| Progressive Volar/Radial Subluxation | Prevents irreversible carpal deformity and skin breakdown over the prominent distal ulna. | |
| Good Elbow Flexion (>90 degrees) | Ensures the patient can reach their mouth and face after the wrist is centralized/realigned. | |
| Relative Indications | Type II Deformity with Functional Deficit | Addressed if progressive bowing or significant weakness impairs daily activities. |
| Older Child (Untreated) | Surgery is possible but carries a higher risk of stiffness and requires more extensive soft tissue releases or external fixation. | |
| Absolute Contraindications | Stiff Elbow in Extension | The radial deviation is a mandatory compensatory mechanism for hand-to-mouth function. |
| Unstable/Untreated Medical Comorbidities | Severe cardiac defects (Holt-Oram) or profound thrombocytopenia (TAR) must be managed prior to elective orthopedic surgery. | |
| Mild Type I Deformity | Functional impairment is minimal; surgery risks iatrogenic injury with little functional gain. | |
| Relative Contraindications | Older Adults with Adapted Function | The brain has fully adapted to the deformity; surgery may decrease overall function despite improving radiographic alignment. |
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning for radial dysplasia reconstruction is a meticulous process that begins months before the actual surgical date. Once the systemic workup is complete and the patient is medically cleared, the focus shifts to optimizing the local soft tissue environment. Nonoperative management is the mandatory first phase of treatment. Beginning shortly after birth, the patient undergoes serial manipulation and casting to stretch the contracted radial structures.
The casting technique requires careful attention; the fulcrum of the stretch must be applied across the carpus and radial soft tissues, taking extreme care to avoid placing sheer stress across the delicate distal ulnar physis. After 3 to 6 months of age, casting is transitioned to custom-fabricated static progressive splinting, worn primarily at night, supplemented by daily active stretching exercises performed by the parents. In cases of severe, rigid deformity where conservative stretching fails to yield adequate soft tissue compliance, preoperative application of a multi-planar external fixator (such as a Taylor Spatial Frame or a specialized Ilizarov apparatus) may be indicated to achieve gradual, controlled distraction histogenesis of the radial soft tissues prior to definitive open reconstruction.
Advanced imaging is critical for surgical templating. While standard orthogonal radiographs of the forearm and hand are standard, we increasingly rely on high-resolution ultrasonography or MRI in younger infants to delineate the unossified cartilaginous anlage, the exact position of the displaced median nerve, and the specific muscular anomalies present. Templating involves calculating the degree of ulnar bowing to determine if a concurrent ulnar closing-wedge osteotomy will be required to achieve a straight forearm axis.
Patient Positioning and Hemostasis Management
In the operating theater, the patient is placed in the standard supine position. The affected upper extremity is abducted onto a radiolucent hand table to allow for unencumbered access to the entire forearm and wrist, and to facilitate intraoperative fluoroscopy. Meticulous padding of all bony prominences is required. The anesthesiologist must be highly experienced in pediatric cases, particularly given the potential for associated airway or cardiac anomalies inherent to the syndromic nature of this condition.
Tourniquet management in infants requires a specialized approach. Standard pneumatic tourniquets are often too wide for the short pediatric forearm, encroaching on the surgical field, and can cause unpredictable pressure distribution on a conical limb. Instead, we utilize a sterile Esmarch bandage. After standard skin preparation and draping, the limb is elevated and exsanguinated with the Esmarch. The bandage is then wrapped tightly three to four times around the proximal humerus to serve as a reliable, low-profile tourniquet.
This technique provides an exquisitely bloodless field, which is absolutely vital for the microscopic dissection required to identify and protect the anomalous median nerve and delicate vascular structures. The time of Esmarch application is strictly recorded, and we adhere to a rigid limit of 90 to 120 minutes of ischemia time before requiring a reperfusion interval.
Step-by-Step Surgical Approach and Fixation Technique
The operative execution of a soft tissue release and bilobed flap reconstruction is a masterclass in spatial geometry, delicate tissue handling, and comprehensive anatomical release. The procedure begins with the precise geometric design of the bilobed flap, a critical step that dictates the success of the entire soft tissue reconstruction.
Step 1: Pre-Incision Markings and Bilobed Flap Design
The bilobed flap is a double-transposition flap designed to move redundant tissue from the ulnar aspect of the wrist to the deficient radial aspect, while allowing the donor site to be closed primarily without tension. Using a sterile surgical marker, the apex of the deformity—the point of maximal radial skin contracture—is identified. This point will serve as the recipient site for the primary lobe.
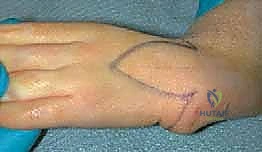
The primary lobe is designed over the redundant ulnar bursa. Its width must equal the anticipated width of the radial defect once the wrist is fully corrected to neutral. The length of the primary lobe is designed to reach the radial apex. The secondary lobe is then designed adjacent to the primary lobe, extending further ulnarly and proximally. The secondary lobe is typically half the width of the primary lobe, and its geometric axis is designed at a 90-degree angle to the primary lobe.
The pivot point is established at the base of the flap, typically volar to the distal ulna. When executed correctly, the primary lobe will rotate 90 degrees to fill the radial defect, and the secondary lobe will rotate 90 degrees to fill the defect left by the primary lobe. The remaining defect from the secondary lobe is closed primarily by undermining the supple ulnar skin.
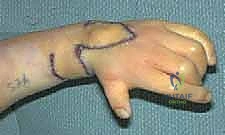
Step 2: Flap Elevation and Median Nerve Identification
Incisions are made sharply through the dermis along the templated lines. The flaps are elevated at the level of the deep investing fascia to preserve the subdermal vascular plexus, ensuring robust perfusion. As the primary and secondary lobes are elevated and reflected proximally and volarly, the underlying distorted anatomy is exposed.
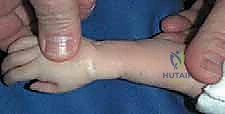
The immediate next step is the identification and protection of the median nerve. Dissection proceeds cautiously on the radial-volar aspect of the incision. The median nerve is frequently found immediately deep to the subcutaneous fat, displaced far radially from its normal anatomical position. It is often encased in thickened fascial bands. Using tenotomy scissors and loupe magnification, a meticulous neurolysis is performed, freeing the nerve from the proximal forearm down to the carpal tunnel. A vessel loop is placed around the nerve for continuous identification and protection throughout the remainder of the case.
Step 3: Radical Soft Tissue Release and Anlage Excision
With the nerve secured, attention turns to the deforming radial structures. The radial anlage is identified as a dense, white, fibrocartilaginous band extending from the proximal forearm to the radial carpus. This structure must be completely excised. We resect the anlage aggressively, taking care not to injure the adjacent protected median nerve or the underlying joint capsules.
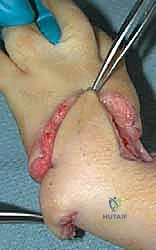
Following anlage excision, the anomalous and contracted radial musculature is addressed. The brachioradialis, ECRL, and ECRB (if present) are identified. To achieve correction, these tendons often require Z-lengthening or fractional lengthening at their musculotendinous junctions. The flexor carpi radialis (FCR) is similarly lengthened. A comprehensive capsulotomy of the radial aspect of the radio-carpal (or ulnocarpal) joint is performed to allow the carpus to be mobilized out of its volarly and radially subluxated position.
Step 4: Carpal Realignment, Fixation, and Flap Inset
Once the soft tissue release is complete, the hand should passively correct to a neutral position over the distal ulna without significant force. If severe ulnar bowing prevents alignment, a concurrent closing-wedge osteotomy of the ulnar diaphysis is performed and stabilized with a dynamic compression plate or intramedullary wire.
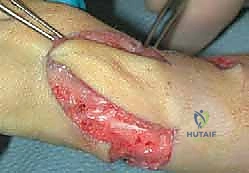
To maintain the corrected alignment during the initial healing phase, a smooth, stout Kirschner wire (typically 0.062 inch) is driven retrogradely through the third metacarpal, across the carpus, and down the medullary canal of the ulna. Extreme care is taken to place this wire centrally through the distal ulnar epiphysis to minimize the risk of physeal arrest. The wire is left protruding distally and bent to facilitate later removal.
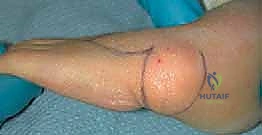
Finally, the bilobed flap is rotated into position. The primary lobe is inset into the massive defect created on the radial side of the wrist by the soft tissue release. The secondary lobe is rotated into the primary donor site. The flaps are secured using interrupted 5-0 or 6-0 absorbable sutures (such as chromic gut or fast-absorbing plain gut) to avoid the need for suture removal in a pediatric patient. The tourniquet is released, and flap perfusion is meticulously confirmed via capillary refill and dermal edge bleeding before the final dressings are applied.

Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, reconstruction of radial dysplasia carries a significant risk of complications, primarily due to the inherent biological drive of the deformity to recur and the fragility of the anomalous tissues. The most common and frustrating complication is the recurrence of radial deviation. This occurs due to the differential growth between the normal ulnar side and the deficient radial side, combined with the relentless pull of the radial musculature. Recurrence rates vary widely in the literature but can approach 30% to 50% over the long term, particularly during the adolescent growth spurt.
Another catastrophic complication is premature physeal arrest of the distal ulna. The distal ulnar physis is responsible for the vast majority of forearm longitudinal growth. Injury to this physis—either from excessive intraoperative pressure during centralization, multiple passes of the intramedullary K-wire, or ischemic injury—will result in a severely shortened forearm. If physeal arrest occurs, limb lengthening procedures utilizing external fixation (distraction histogenesis) may be required in older childhood or adolescence to restore functional length.
Wound complications and flap necrosis are also significant risks. The bilobed flap relies on a random pattern blood supply. If the flap is designed with excessive tension, or if the base is made too narrow, distal tip necrosis of the primary lobe can occur. This is usually managed conservatively with local wound care, allowing healing by secondary intention, though severe necrosis may require subsequent skin grafting, which can re-introduce radial-sided contracture.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Recurrence of Deformity | 30% - 50% | Inadequate initial release, differential growth, intact radial anlage. | Revision soft tissue release, ulnar osteotomy, or radialization procedure. |
| Distal Ulnar Physeal Arrest | 10% - 20% | Iatrogenic injury from K-wire, excessive compression during centralization. | Observation if mild; Ilizarov distraction osteogenesis for severe length discrepancy. |
| Median Nerve Injury | < 5% | Failure to identify the anomalously positioned nerve during initial dissection. | Primary microscopic repair or nerve grafting; often results in permanent sensory/motor deficits. |
| Flap Tip Necrosis | 5% - 15% | Excessive tension on inset, inadequate flap base width, vascular compromise. | Local wound care, healing by secondary intention; full-thickness skin graft if severe. |
| Pin Site Infection | 10% - 15% | Superficial bacterial colonization of the protruding K-wire. | Oral antibiotics, more frequent dressing changes; early pin removal if deep infection suspected. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following radial dysplasia reconstruction is as critical as the surgical procedure itself. The protocol is designed to protect the delicate soft tissue reconstruction, maintain the skeletal alignment achieved by the K-wire, and gradually reintroduce functional movement while preventing the recurrence of contractures. This requires a dedicated hand therapist and highly compliant parents.
Phase 1 encompasses the initial immobilization period, typically lasting from postoperative day 1
Detailed Chapters & Topics
Dive deeper into specialized chapters regarding radial-dysplasia-reconstruction
