Masterclass in Distal Upper Extremity Skin Grafting & Substitutes: An Intraoperative Guide
Key Takeaway
Welcome, fellows, to an immersive masterclass on skin grafting and substitutes for distal upper extremity wounds. We'll meticulously cover patient positioning, comprehensive anatomy, advanced wound bed preparation, precise graft harvesting, and application techniques. Learn critical pearls and pitfalls for both split-thickness and full-thickness grafts, ensuring optimal functional and aesthetic outcomes in complex cases.
Introduction and Terminology
The reconstruction of soft tissue defects in the distal upper extremity requires a rigorous understanding of microvascular anatomy, wound healing physiology, and the biomechanical demands of the hand and wrist. Wounds in this anatomic region that are candidates for skin grafting adhere to the universal principles of free tissue transfer; however, the unique functional requirements of the hand—specifically the need for durable, sensate, and pliable coverage over highly mobile joints—dictate a highly selective approach to graft and donor site selection.
Core Terminology
A precise understanding of graft classification is fundamental to surgical decision-making:
* Autograft: Tissue harvested from and applied to the same individual. This remains the gold standard for permanent wound coverage.
* Isograft: Tissue transferred between genetically identical individuals (monozygotic twins). Immunologically, it behaves identically to an autograft.
* Allograft: Tissue harvested from a genetically non-identical individual of the same species (cadaveric skin). Due to major histocompatibility complex (MHC) mismatch, allografts are subject to immunologic rejection and eventual separation, serving primarily as a temporary biologic dressing unless the host is severely immunosuppressed.
* Xenograft: Tissue harvested from a different species (e.g., porcine skin). Like allografts, these provide only temporary coverage and are associated with a higher rate of wound bed infection and inflammatory reaction.
* Split-Thickness Skin Graft (STSG): A graft containing the entire epidermis and a variable portion of the underlying dermis.
* Full-Thickness Skin Graft (FTSG): A graft incorporating the entire epidermis and the complete dermal layer, excluding the subcutaneous fat.
* Skin Substitutes: Semisynthetic or purely synthetic bilaminar constructs designed to replace lost skin structures, often providing a scaffold for cellular ingrowth. The criteria for an ideal skin substitute were classically defined in 1984, emphasizing qualities such as lack of antigenicity, structural stability, and capacity for fibrovascular ingrowth (Pruitt BA Jr, Levine NS. Characteristics and uses of biologic dressings and skin substitutes. Arch Surg 1984;119:312-322.).
Wound Bed Preparation and Pathophysiology of Graft Survival
The success of any free skin graft is entirely dependent on the vascularity and preparation of the recipient bed. A skin graft initially lacks an intrinsic blood supply and must rely on the diffusion of nutrients and subsequent neovascularization from the host bed.
Microanatomy and Vascular Substrate
A graft bed must consist of healthy, well-vascularized tissue. Acceptable substrates include healthy subcutaneous fat, viable muscle, paratenon, and periosteum. The graft will uniformly fail if placed over denuded bone (lacking periosteum), denuded tendon (lacking paratenon), denuded cartilage (lacking perichondrium), or heavily irradiated tissue. These avascular surfaces cannot support the robust capillary ingrowth required for graft inosculation.
Bacterial Colonization and Debridement
Radical debridement of all nonviable, necrotic, or fibrotic tissue is mandatory. Furthermore, the bacterial bioburden of the wound must be strictly controlled. Quantitative tissue cultures demonstrating greater than $10^5$ colony-forming units (CFUs) per gram of tissue serve as a reliable negative predictor of graft survival. Enhanced graft survival achieved by aggressive reduction of bacterial counts has been well documented (Perry AW, Sutkin HS, Gottlieb LJ, et al. Skin graft survival—the bacterial answer. Ann Plast Surg 1989;22:479-483.). Pulse lavage systems and meticulous surgical excision are primary modalities for achieving this.
Negative Pressure Wound Therapy
The application of a vacuum-assisted closure (VAC) device provides continuous microdebridement, manages exudate, reduces interstitial edema, and promotes the formation of a robust granulation tissue bed. Furthermore, the VAC device is an exceptional adjunct when applied directly over a newly placed skin graft, acting as an optimal bolster that eliminates shear forces and prevents subgraft fluid collection (hematoma/seroma).
Properties and Biomechanics of Skin Grafts
The Mechanism of Graft "Take"
The incorporation of a skin graft into the host bed occurs in three distinct, sequential physiologic phases:
1. Plasmatic Imbibition (Days 0-2): The graft survives via passive diffusion of oxygen and nutrients from the wound bed exudate. The graft becomes edematous and increases in weight. A friable fibrin network anchors the graft to the bed.
2. Inosculation (Days 3-4): Capillary buds from the recipient bed align with and penetrate the preexisting vascular channels within the graft dermis.
3. Capillary Ingrowth / Neovascularization (Days 4+): True blood flow is reestablished. The graft transitions from a pale/white appearance to a healthy pink hue. Fibrovascular ingrowth establishes definitive secondary adherence.
Contraction Mechanics
Graft contraction is a critical biomechanical consideration, particularly in the hand where unchecked contraction leads to severe joint contracture and loss of function.
* Primary Contraction: Occurs immediately upon harvest due to the recoil of intrinsic dermal elastin fibers. Because FTSGs contain the entire dermal layer, they exhibit significant primary contraction compared to STSGs.
* Secondary Contraction: Occurs as the graft heals in the wound bed, mediated by myofibroblasts. STSGs undergo substantially more secondary contraction than FTSGs. Consequently, FTSGs are strongly preferred over or adjacent to joints to prevent functional contractures. FTSGs also possess the unique capacity to grow proportionally with a pediatric patient, whereas STSGs remain static or shrink.
Reinnervation and Dyspigmentation
Sensory recovery in skin grafts proceeds via both peripheral ingrowth from the wound margins and deep neural ingrowth from the bed. The speed and quality of reinnervation are directly proportional to the accessibility of empty neural sheaths within the graft. Because FTSGs contain a higher density of intact neural sheaths, they exhibit more rapid and complete sensory recovery compared to STSGs. Protective sensation typically begins to return at 4 to 6 weeks, with maximal recovery taking 12 to 24 months.
Dyspigmentation occurs due to the transient ischemia post-harvest, leading to a temporary reduction in melanoblast populations. While melanocytes eventually recover, STSGs are notoriously prone to permanent dyschromia, whereas FTSGs tend to maintain a color match closer to their native donor site.
Patient Evaluation and Preoperative Planning
History and Physical Examination
A comprehensive evaluation must identify systemic factors that impair microvascular perfusion and wound healing, such as poorly controlled diabetes mellitus, peripheral vascular disease, malnutrition, and particularly tobacco use. The wound bed must be meticulously inspected to confirm the presence of a vascularized substrate (paratenon, periosteum, muscle) and the absence of gross purulence.
Diagnostic Studies
In cases of chronic or heavily contaminated wounds, a punch biopsy of the viable wound bed should be obtained for quantitative culture. The biopsy must target viable tissue, avoiding superficial slough or eschar. As previously noted, a bioburden exceeding $10^5$ CFUs/gram necessitates further debridement and targeted antimicrobial therapy prior to grafting.
Differential Diagnosis and Defect Classification
The reconstructive ladder dictates that the surgeon accurately classify the defect:
* Superficial/partial-thickness skin loss (amenable to secondary intention or biologic dressings).
* Full-thickness skin loss over broad flat surfaces (amenable to STSG).
* Full-thickness skin loss over joints or volar glabrous surfaces (requires FTSG).
* Composite defects with exposed avascular structures (requires local/regional flaps, free tissue transfer, or dermal regeneration templates).
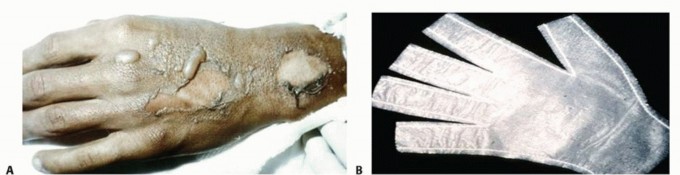
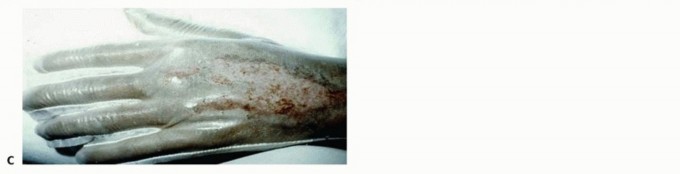
Superficial partial-thickness injuries, such as the dorsal hand burn shown above, can often be managed with biosynthetic dressings like the Biobrane glove, which provides a protective scaffold and limits pain while native reepithelialization occurs. Deeper, full-thickness injuries mandate surgical debridement and grafting.
Donor Site Selection and Surgical Anatomy
Split-Thickness Skin Graft Donor Sites
- Anterior Thigh: The traditional workhorse donor site due to its large surface area, ease of access in the supine position, and relatively straightforward postoperative care.
- Scalp: An exceptional, albeit less commonly utilized, donor site. The scalp's robust vascularity and high density of epidermal appendages allow for extremely rapid reepithelialization (often within 5-7 days), minimizing donor site morbidity. Harvesting requires subgaleal injection of tumescent fluid (e.g., Klein solution) to provide turgor and hemostasis. If harvested superficially within the hair-bearing region, alopecia is avoided, and the donor scar is perfectly concealed.
Full-Thickness Skin Graft Donor Sites
- Lower Abdomen / Groin: Provides a large surface area of redundant tissue that allows for primary closure. The incision can be hidden within the bikini line. The surgeon must account for the hair-bearing nature of the central groin versus the relatively glabrous lateral extensions.
- Medial Upper Arm: The junction of the medial biceps and triceps yields thin, pliable, and typically hairless skin that is an excellent color match for the upper extremity.
- Glabrous Skin: For defects on the palmar surface of the hand, replacing "like with like" is ideal to withstand shear forces. Glabrous FTSG can be harvested from the hypothenar eminence (ulnar border of the hand) or the non-weight-bearing medial arch (instep) of the plantar foot.
Surgical Technique: Split-Thickness Skin Grafts
Preoperative Templating
Accurate templating minimizes donor site morbidity. A sterile glove wrapper is pressed into the wound bed; the absorbed moisture outlines the defect. This template is excised and transferred to the donor site, where it is outlined with a surgical marker, typically adding a slight margin to accommodate the geometry of the dermatome.
Harvesting the Graft
The donor site is infiltrated with an epinephrine-containing local anesthetic to provide hydrodissection and hemostasis. A pneumatic or electric dermatome is set to the desired thickness (typically 0.012 to 0.015 inches for a medium STSG). Mineral oil is applied to the skin and the dermatome to reduce friction. The dermatome is advanced with steady, downward pressure at a consistent 45-degree angle.

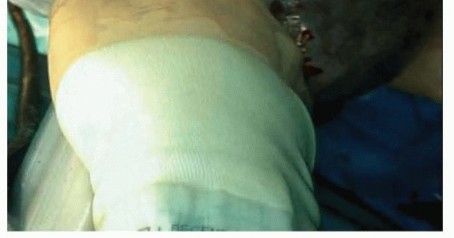
Technique of split-thickness skin graft harvest utilizing a powered dermatome.
Meshed Versus Unmeshed (Sheet) Grafts
The harvested STSG can be applied as a contiguous sheet or passed through a mesher.
* Sheet Grafts: Provide superior cosmetic outcomes and are preferred for the dorsal hand and digits. However, they lack avenues for fluid egress, making them highly susceptible to failure secondary to subgraft hematoma or seroma.
* Meshed Grafts: Passing the graft through a mesher (e.g., 1:1.5 ratio) creates fenestrations. Even if not expanded, these fenestrations allow for the drainage of blood and exudate, significantly increasing the likelihood of graft adherence in contaminated or highly exudative beds.


Appearance of a meshed split-thickness graft, oriented dermis side up prior to application.
Graft Application and Fixation
The graft is placed over the wound bed with the dermis facing down. It is secured peripherally using staples or fine absorbable sutures (e.g., 4-0 or 5-0 chromic gut). To eliminate dead space in irregular wound beds, quilting sutures should be placed centrally.
A nonadherent interface (e.g., Xeroform or petrolatum-impregnated gauze) is applied. Fixation is paramount; shear forces will disrupt the fragile fibrin bonds and vascular ingrowth. A tie-over bolster using mineral oil-soaked cotton can be utilized, or alternatively, a VAC device set to -75 to -125 mmHg continuous pressure provides unparalleled graft immobilization and fluid evacuation. The extremity is then placed in a rigid sugar-tong or volar splint to prevent wrist and digital motion.
Surgical Technique: Full-Thickness Skin Grafts
Harvesting and Defatting
Following templating, the donor site is infiltrated with local anesthetic containing epinephrine. The graft is sharply incised with a No. 15 blade and elevated. It is critical to stay in the superficial subcutaneous plane to avoid deep neurovascular structures, yet ensure the entire dermis is captured.
Defatting is the most critical step in FTSG preparation. Any residual subcutaneous fat acts as an avascular barrier that prevents inosculation. The graft is stretched over the surgeon's non-dominant index finger, and curved tenotomy or dissecting scissors are used to meticulously excise all yellow adipose tissue until the stark white, glistening undersurface of the dermis is uniformly exposed.
Application and Pie-Crusting
Because FTSGs undergo significant primary contraction, they must be stretched back to their original dimensions and sutured into the defect under physiologic tension. To prevent subgraft fluid accumulation (as FTSGs cannot be meshed without destroying their structural integrity), the surgeon must perform "pie-crusting"—making multiple small, random stab incisions through the graft with a No. 11 blade.
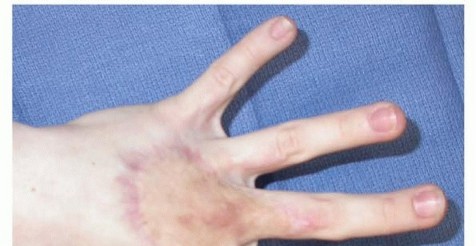
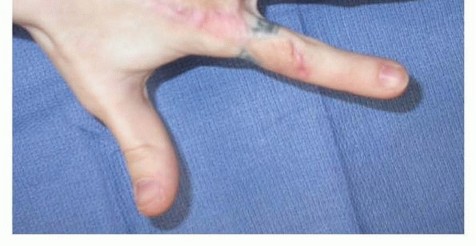
Appearance of a mature, well-incorporated full-thickness skin graft applied over the dorsal aspect of the hand and metacarpophalangeal joints, demonstrating excellent contour and lack of secondary contracture.
Skin Substitutes in the Distal Upper Extremity
When a defect is too large for an FTSG, but an STSG is contraindicated due to the need for durability, tendon gliding, or joint mobility, dermal regeneration templates and skin substitutes are indicated.
AlloDerm
AlloDerm (LifeCell Corp.) is an acellular human cadaveric dermal matrix. It provides a structurally intact basement membrane complex and dermal collagen scaffold. It is sutured into the defect and monitored. Over 2 to 3 weeks, host fibrovascular tissue populates the matrix. Once robust granulation is evident through the matrix, a thin STSG is applied in a secondary procedure.
Integra Dermal Regeneration Template
Integra (Integra LifeSciences) is a bilaminar construct comprising a porous matrix of cross-linked bovine tendon collagen and glycosaminoglycan (chondroitin-6-sulfate) covered by a temporary silicone epidermal substitute.
Upon application, it acts as a scaffold for endothelial cells and fibroblasts. Over 14 to 21 days, the matrix vascularizes, transitioning from white to a peach/rosy hue. Once fully vascularized, the silicone layer is peeled away, and an ultra-thin STSG (0.004 - 0.006 inches) is applied to the neo-dermis. This technique provides a highly pliable, durable reconstruction that mimics an FTSG but covers a massive surface area.
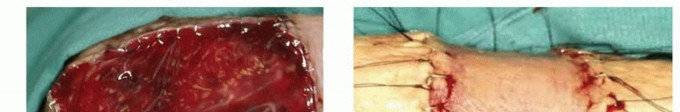
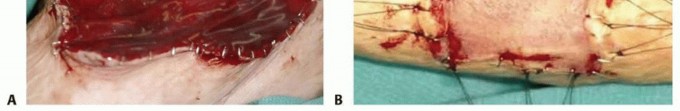
*(A) Appearance of mature, vascularized Integra applied over an open forearm wound, indicated by the rosy
You Might Also Like