Radial Shortening Osteotomy for Kienböck Disease: An Intraoperative Masterclass
Key Takeaway
This masterclass guides fellows through radial shortening osteotomy for Kienböck disease. We cover intricate lunate vascularity, ulnar variance assessment, and meticulous preoperative planning. The core focus is a real-time, step-by-step intraoperative execution, detailing incision, dissection, precise osteotomy, and hardware fixation. We emphasize critical anatomical landmarks, potential pitfalls, and advanced rehabilitation protocols to optimize patient outcomes.
Introduction and Epidemiology
Kienböck disease is an insidious and progressive disorder characterized by avascular necrosis (AVN) of the carpal lunate. The precise etiology remains undetermined, though it is widely considered to be multifactorial, involving a combination of anatomical, mechanical, and vascular predispositions. The condition predominantly affects young, active adults, typically presenting between the second and fourth decades of life (20 to 40 years of age).
Epidemiologically, there is a pronounced male-to-female predominance, with reported ratios ranging from 3:1 to 7:1. Bilateral involvement is exceedingly rare. Notably, more than 95% of patients diagnosed with Kienböck disease are engaged in heavy manual labor, suggesting that repetitive microtrauma and repetitive compressive loading across the radiocarpal joint play a significant role in its pathogenesis. While Kienböck disease has been sporadically reported in association with systemic conditions such as sickle cell disease, gout, cerebral palsy, septic emboli, carpal coalition, and chronic corticosteroid use, there is no definitively established correlation with any systemic or neuromuscular process that mandates routine systemic screening during diagnostic workup.
Surgical Anatomy and Biomechanics
Lunate Vascularity
The lunate relies on a complex and highly variable extraosseous and intraosseous vascular network. The extraosseous blood supply is robust: the dorsal lunate plexus is formed by branches of the radial and anterior interosseous arteries, while the volar plexus is supplied by branches of the radial, ulnar, and anterior interosseous arteries, supplemented by the recurrent deep palmar arch.
Despite this rich extraosseous network, the intraosseous vascularity presents significant vulnerabilities. Because the lunate is extensively covered by articular cartilage proximally (radiocarpal joint) and distally (midcarpal joint), nutrient vessels can only penetrate the bone at its non-articular dorsal and volar poles.
Anatomical studies have identified specific intraosseous vascular patterns that place certain "lunates at risk" for ischemic events. In 7% to 20% of the population, large regions of the lunate are entirely dependent on a single intraosseous vessel. Furthermore, up to 31% of lunates exhibit a Y-shaped or I-shaped internal vascular pattern with absolutely no internal arterial branching or collateral anastomoses. These precarious internal vascular arrangements render the lunate highly susceptible to AVN; a traumatic or microtraumatic injury to a single nutrient vessel cannot be compensated for by collateral flow, precipitating focal or global ischemia.
Ulnar Variance and Radiocarpal Mechanics
Ulnar variance is a critical biomechanical parameter in the evaluation of Kienböck disease. It is assessed via a standardized posteroanterior (PA) wrist radiograph, obtained with the patient's shoulder abducted to 90 degrees, the elbow flexed to 90 degrees, and the forearm in neutral rotation. Ulnar variance is defined as the relative length of the distal articular surface of the ulna compared to the distal articular surface of the radius.
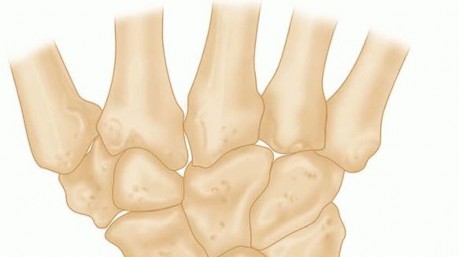
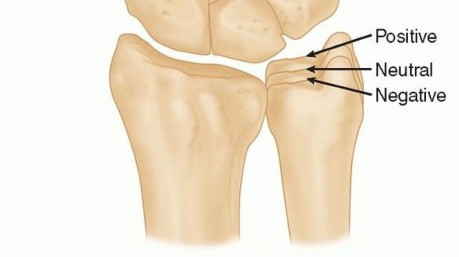
Neutral ulnar variance exists when the articular surfaces are collinear. Negative ulnar variance occurs when the ulna is shorter than the radius, and positive ulnar variance occurs when the ulna is longer.
Biomechanically, negative ulnar variance is theorized to increase the shear and compressive forces transmitted directly to the lunate. In a negative variance wrist, the triangular fibrocartilage complex (TFCC) is anatomically thicker. The discrepancy in compliance between the thick, compliant TFCC and the rigid ulnar edge of the distal radius accentuates shear forces on the lunate during carpal motion. Consequently, axial loads across the radiocarpal joint are borne disproportionately by the radiolunate articulation rather than being shared optimally with the ulnocarpal articulation.
In North American demographics, Kienböck disease demonstrates a strong statistical association with negative ulnar variance. However, this correlation is notably absent in the Japanese literature, and there is currently no definitive evidence establishing a direct causal relationship between negative ulnar variance and the onset of Kienböck disease. Some morphometric studies also indicate a tendency toward volumetrically smaller lunates in affected patients, which may further concentrate focal stresses.
Pathogenesis and Natural History
The pathogenesis of Kienböck disease must be differentiated from simple transient ischemia of the lunate. Transient ischemia lacks the progressive radiographic collapse that is the hallmark of true Kienböck disease.
Contemporary theories suggest that the initial vascular insult may stem from venous outflow obstruction resulting in intraosseous vascular congestion, rather than pure arterial insufficiency. Elevated intraosseous pressures have been documented in lunates affected by Kienböck disease (analogous to the pathophysiology of AVN in the femoral head). This hemodynamic profile aligns more closely with venous stasis. Alternatively, elevated intraosseous pressure may be a secondary phenomenon resulting from structural bony collapse.
Once the lunate becomes avascular, repetitive loading leads to subchondral stress fractures. These fractures predictably initiate in the proximal lunate adjacent to the radial articular surface—the zone with the most tenuous blood supply. Consequently, the proximal lunate undergoes flattening and fragmentation earlier than the distal pole. Furthermore, the radial aspect of the lunate (articulating with the rigid radius) is typically more severely involved than the ulnar aspect (overlying the compliant TFCC), a discrepancy that is magnified in wrists with negative ulnar variance.
As lunate structural integrity fails, coronal plane fractures frequently develop. The capitate acts as a wedge, driving compressive forces that displace these fracture fragments volarly and dorsally. This leads to a predictable cascade: lunate collapse, loss of carpal height, scaphoid rotary subluxation (flexion), proximal migration of the capitate, and ultimately, advanced radiocarpal and midcarpal degenerative joint disease (perilunate arthritis).
Despite this progressive radiographic deterioration, the natural history does not universally correlate with catastrophic clinical decline. Long-term follow-up studies comparing immobilized wrists to non-treated wrists (mean follow-up of 20.5 years) demonstrate that a majority of patients experience a gradual decrease in subjective symptoms over time. In one cohort, 83% of immobilized and 77% of untreated wrists were pain-free or experienced pain only during heavy manual labor, despite 67% demonstrating radiographic radiocarpal arthritis. This indicates that Kienböck disease may exhibit a naturally benign clinical course in a subset of patients, and that radiographic appearance correlates poorly with long-term residual symptoms.
Patient History and Physical Examination
Patients typically present with an insidious onset of dorsal central wrist pain, progressive stiffness, and a profound reduction in grip strength—often diminished to 50% of the contralateral, unaffected extremity. The duration of symptoms prior to formal presentation can be protracted, ranging from months to years. Pain intensity is highly variable, ranging from mild, activity-related aching to severe, constant, debilitating pain. Symptoms generally improve with rest and splint immobilization. A distinct history of an acute traumatic event is variable and frequently absent.
On physical examination, the wrist often exhibits mild dorsal swelling indicative of reactive radiocarpal synovitis. Point tenderness is localized directly over the dorsal aspect of the lunate. Range of motion is predictably restricted. Wrist flexion is typically more limited than extension; this occurs because the volar pole of the collapsing lunate frequently extrudes volarly, mechanically impinging against the volar rim of the distal radius during flexion. Forearm pronation and supination are generally preserved and unaffected.
Imaging and Diagnostic Studies
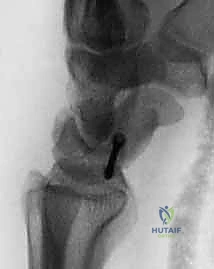
Radiographic Classification (Lichtman Staging)
Standard plain radiography remains the cornerstone for staging Kienböck disease, utilizing the modified Lichtman classification system to guide therapeutic decision-making.
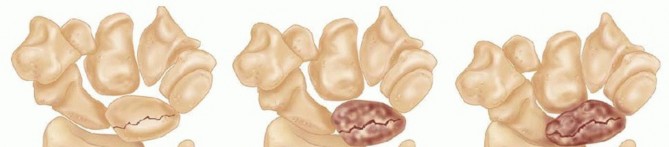
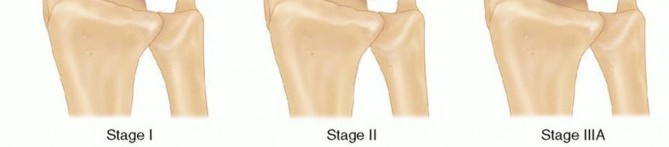
- Stage I: Normal radiographic appearance (diagnosis relies on MRI).
- Stage II: Sclerosis of the lunate without structural collapse.
- Stage IIIA: Lunate collapse and fragmentation, but normal carpal alignment and carpal height.
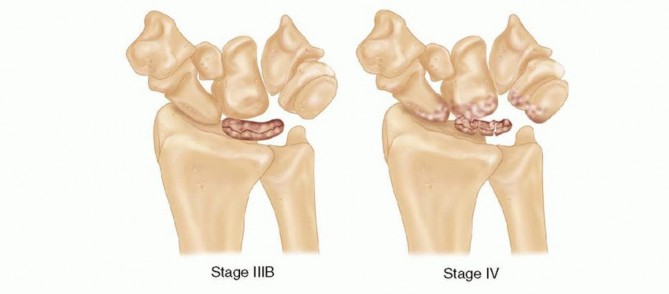
- Stage IIIB: Lunate collapse with fixed carpal instability (scaphoid flexion, proximal capitate migration, loss of carpal height).
- Stage IV: Advanced lunate collapse accompanied by secondary degenerative osteoarthritis of the radiocarpal and/or midcarpal joints (joint space narrowing, osteophyte formation, subchondral sclerosis, and cystic changes).
Advanced Imaging: MRI and CT
Magnetic Resonance Imaging (MRI) is the gold standard for early diagnosis, particularly in Stage I disease before trabecular destruction is radiographically apparent.
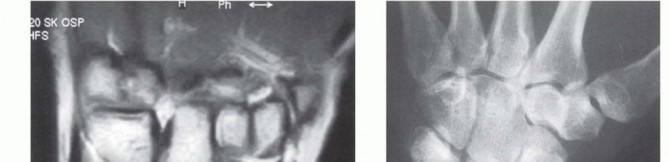
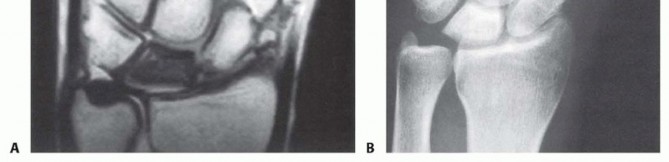
T1-weighted images demonstrate diffuse loss of normal marrow fat signal. T2-weighted images typically reveal low signal intensity, representing the replacement of normal fatty marrow with fibrotic tissue. However, if T2 images demonstrate hyperintense (increased) signal, this indicates active intramedullary edema or ongoing revascularization, suggesting an earlier stage of disease and a potentially more favorable prognosis. Gadolinium-enhanced MRI provides superior sensitivity for evaluating residual lunate perfusion. It must be noted that large enchondromas, interosseous ganglia, or other marrow-replacing lesions can mimic these MRI findings; thus, there is no single pathognomonic MRI sign.
Computed Tomography (CT) is highly valuable once structural changes commence. CT provides superior resolution of trabecular architecture and fracture lines. Studies indicate that CT imaging upstages the disease (compared to plain radiographs) in 89% of apparent Stage I cases, 71% of Stage II cases, and 9% of Stage III cases.
Differential Diagnosis
The clinical and radiographic presentation of Kienböck disease must be differentiated from:
* Ulnocarpal impaction syndrome
* Rheumatoid arthritis
* Radial-sided triangular fibrocartilage complex (TFCC) tears
* Posttraumatic radiocarpal arthritis
* Acute carpal fractures (especially scaphoid or lunate)
* Carpal instability patterns (DISI/VISI)
* Benign bone tumors (Enchondroma, Osteoid osteoma, Bone island)
* Occult or intraosseous ganglion cysts
* Transient lunate ischemia or "Bone bruise"
* Metabolic/Systemic conditions (Paget disease, Gaucher disease)
Management Strategies
Nonoperative Management
A conservative trial of immobilization (casting or rigid splinting) for 2 weeks to 3 months may be considered exclusively for early Stage I disease, particularly in young patients demonstrating hyperintense (edematous) lunates on T2 MRI. The biomechanical rationale is that reducing active forces across the carpus may facilitate spontaneous revascularization.
However, clinical data heavily discourages prolonged nonoperative management. Immobilization fails to neutralize the passive compressive forces exerted by the capitate onto the lunate. Consequently, the capitate continues to drive fracture fragments apart, leading to predictable structural collapse. Furthermore, prolonged immobilization induces significant radiocarpal stiffness.
Literature demonstrates poor outcomes with conservative care. In a series of 22 patients managed with immobilization, 17 exhibited progressive lunate collapse, and 5 showed no improvement. Another series reported unsatisfactory results in 19 of 22 patients. For patients with Stage II or advanced disease, conservative management is contraindicated; one study showed 76% of such patients eventually required total wrist arthrodesis or suffered daily debilitating pain at an 8-year follow-up. Early surgical intervention is therefore strongly advocated to unload the lunate before irreversible collapse occurs.
Surgical Indications and Options
Surgical intervention aims to alter carpal biomechanics, decompress the lunate, or salvage the joint in end-stage disease.
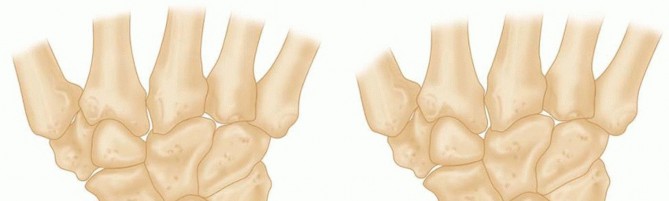
- Joint Leveling Procedures (Radius Shortening / Radial Closing Wedge Osteotomy): The primary treatment for Lichtman Stages I, II, and IIIA. In patients with negative ulnar variance, a radius shortening osteotomy is the gold standard. In patients with neutral or positive ulnar variance, a lateral radial closing wedge osteotomy or capitate shortening (with or without capitohamate fusion) is utilized. Interestingly, radius shortening has shown excellent clinical results even in ulnar neutral or positive wrists.
- Revascularization: Vascularized bone grafting (e.g., 4+5 ECA pedicle) may be utilized for Stages I to IIIA, often in conjunction with a joint leveling procedure.
- Salvage Procedures (Stage IIIB and IV): Proximal row carpectomy (PRC), scaphotrapeziotrapezoid (STT) fusion, or scaphocapitate (SC) fusion. For Stage IV disease, PRC or total wrist arthrodesis is indicated.
- Adjunct Procedures: Arthroscopic debridement has shown temporary palliative benefit in advanced stages. Metaphyseal core decompression of the distal radius (targeting venous hypertension) and wrist denervation (AIN/PIN neurectomy) can be utilized as adjuncts at any stage.
Preoperative Planning and Patient Positioning
High-quality, standardized PA radiographs are mandatory to accurately calculate ulnar variance.
Biomechanical Planning for Osteotomy: While historical algorithms recommended shortening the radius to achieve neutral or +1 mm positive variance, modern biomechanical studies prove that 90% of the strain reduction on the lunate occurs within the first 2 mm of shortening. Consequently, a conservative shortening of exactly 2 to 3 mm is highly recommended, regardless of the initial magnitude of negative variance.
Attempting to shorten the radius by more than 4 mm is strictly contraindicated. Excessive shortening drastically alters the kinematics of the distal radioulnar joint (DRUJ), leading to severe DRUJ incongruity, restricted forearm rotation, and iatrogenic ulnocarpal impaction syndrome. Patients over 30 years of age subjected to >4 mm of shortening predictably yield poor clinical outcomes. In cases with a highly oblique sigmoid notch, shortening should be strictly limited to a maximum of 2 mm to prevent catastrophic DRUJ compression.
Positioning: The patient is positioned supine with the operative extremity extended onto a radiolucent hand table. A well-padded proximal tourniquet is applied.
Detailed Surgical Approach and Technique
Volar Approach to the Distal Radius
A standard volar (Henry) approach to the distal radius is utilized.



The incision is made over the flexor carpi radialis (FCR) tendon. The FCR sheath is incised, and the tendon is retracted ulnarward to protect the median nerve. The floor of the FCR sheath is incised to expose the pronator quadratus. The distal border and radial insertion of the pronator quadratus, along with the radial insertion of the flexor pollicis longus, are sharply elevated. The radial artery is carefully protected and retracted radially; formal skeletonization of the artery is unnecessary. The volar surface of the radius is exposed subperiosteally from radial to ulnar. Strict care must be taken to avoid circumferential periosteal stripping, which would devascularize the osteotomy site and increase the risk of nonunion.
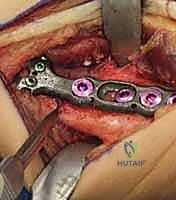
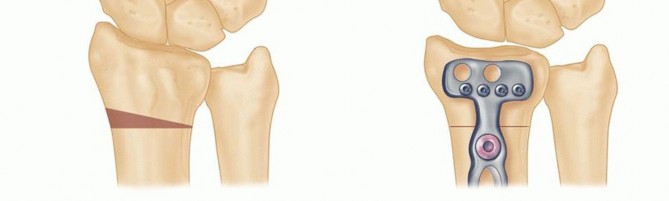
Radius Shortening Osteotomy
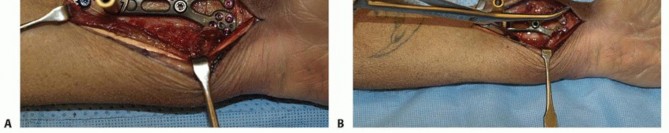
Initial Plate Application:
A volar locking plate (often a standard distal radius fracture plate) is provisionally applied.

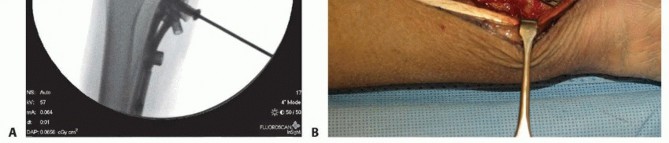
The plate is positioned such that the distal locking screws (or provisional K-wires) are placed just proximal to the radiocarpal subchondral surface. The distal locking screws are fully inserted to secure the distal fragment.
Creation of the Osteotomy:
The planned osteotomy site is marked on the volar cortex, ensuring it is positioned strictly proximal to the distal radioulnar joint (DRUJ) and between the distal and proximal plate holes.
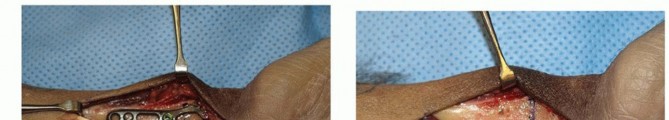
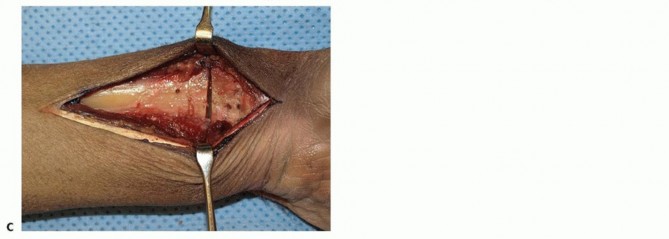
The plate is temporarily removed or rotated out of the way. A transverse osteotomy is highly preferred over an oblique osteotomy. While an oblique cut theoretically increases surface area and allows for an interfragmentary lag screw, it is technically demanding, and lag screw purchase is often poor.
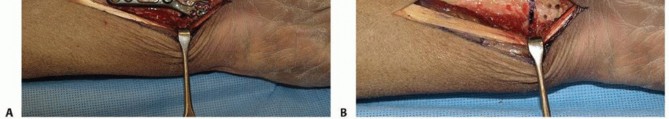
Using an oscillating saw under continuous cool saline irrigation (to prevent thermal osteonecrosis), parallel transverse cuts are made to remove exactly 2 to 3 mm of bone. A periosteal elevator is placed dorsally to protect the extensor tendons. The cuts are made from volar to dorsal, deliberately leaving the dorsal cortex intact until the very end to maintain stability during the cutting process. Once the measured segment is mobilized, the dorsal cortex is completed. The flat transverse cut allows for easy intraoperative assessment of rotational alignment.
Final Plate Application and Compression:
The plate is reapplied to the distal fragment.
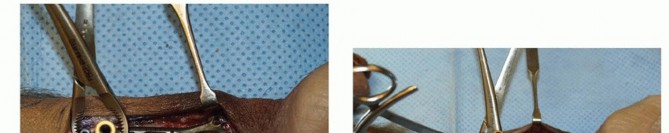

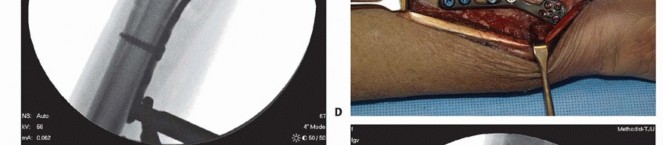

To achieve robust compression across the osteotomy, a bicortical screw (deliberately chosen to be a few millimeters longer than the bone width) is placed
You Might Also Like