Masterclass: Split Posterior Tibial Tendon Transfer for Equinovarus Deformity
Key Takeaway
This masterclass provides an immersive, step-by-step guide to Split Posterior Tibial Tendon Transfer for equinovarus deformity. We cover essential preoperative planning, precise patient positioning, and granular intraoperative execution, including specific techniques for tendon splitting, routing, and fixation. Fellows will gain insights into critical surgical anatomy, neurovascular considerations, pearls, pitfalls, and comprehensive postoperative rehabilitation protocols to ensure optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The Split Posterior Tibial Tendon Transfer (SPTTT) remains a foundational surgical intervention in the armamentarium of the orthopedic surgeon managing complex pediatric and adult foot deformities. This operation is a critical cornerstone in the correction of dynamic equinovarus foot deformities, most notably within the cerebral palsy (CP) population, but equally applicable to patients with traumatic brain injuries, cerebrovascular accidents, and specific peripheral neuropathies. The overarching goal of this procedure is not merely anatomical realignment, but the restoration of dynamic biomechanical balance, the optimization of the gait cycle, and the enhancement of the patient’s overall functional independence and quality of life.
To master this procedure, one must first deeply understand the underlying patho-epidemiology. The equinovarus deformity is a complex, multiplanar distortion characterized by a synergistic combination of hindfoot equinus (plantarflexion at the tibiotalar joint) and hindfoot varus (inversion at the subtalar joint). In the context of upper motor neuron lesions, this is rarely a static, isolated problem; rather, it is a dynamic biomechanical imbalance. It primarily stems from an overactive, spastic, or out-of-phase tibialis posterior muscle, frequently coupled with profound weakness or delayed activation in its antagonist groups, specifically the peroneus brevis and the tibialis anterior.
Epidemiologically, the spastic equinovarus foot is the most common foot deformity encountered in patients with spastic hemiplegic cerebral palsy, though it presents with significant frequency in diplegic and quadriplegic variants as well. The natural history of this deformity is one of predictable deterioration if left unmanaged. It initiates as a purely dynamic imbalance, visible only during specific phases of the gait cycle—typically manifesting as excessive inversion and plantarflexion during the swing phase, leading to inadequate foot clearance and toe drag. During the stance phase, the initial contact occurs abnormally over the lateral border of the forefoot, forcing the hindfoot into a rigid varus position, severely compromising the stability of the support base and increasing the energy expenditure of ambulation.
Over time, the chronic, unrelenting dynamic tethering forces exerted on the growing pediatric skeleton inevitably lead to myostatic contractures. The muscle-tendon units physically shorten, losing their intrinsic extensibility, which transitions the deformity from a flexible, dynamic state to a rigid, fixed structural pathology. If permitted to progress to this stage, the constant abnormal loading alters the morphological development of the tarsal bones, resulting in fixed bony adaptations such as a structural cavovarus foot. Therefore, timely intervention with a procedure like the SPTTT is paramount to intercept this pathophysiological cascade, redirecting deforming muscular forces to act as corrective, balancing vectors before irreversible bony changes mandate complex, joint-sacrificing arthrodesis.
Detailed Surgical Anatomy and Biomechanics
A profound, three-dimensional understanding of the surgical anatomy of the leg and foot is non-negotiable for the safe and efficacious execution of the SPTTT. The tibialis posterior muscle, the primary pathogenic driver of the varus deformity, originates broadly from the posterior aspect of the interosseous membrane, the posterolateral surface of the tibia, and the medial surface of the fibula. Situated deep within the posterior compartment of the leg, it is a pennate muscle designed for significant force generation. Its tendon courses distally, passing through a distinct fibro-osseous tunnel immediately posterior to the medial malleolus, securely contained within its own synovial sheath beneath the flexor retinaculum.
The insertional anatomy of the tibialis posterior is remarkably complex and biomechanically significant. While its primary and most robust insertion is onto the tuberosity of the navicular bone, it possesses broad, diffuse plantar extensions. These tendinous slips fan out to insert onto the medial, intermediate, and lateral cuneiforms, the cuboid, the sustentaculum tali of the calcaneus, and the bases of the second, third, and fourth metatarsals. This extensive plantar footprint explains the muscle's profound influence on the entire midfoot and hindfoot architecture. Biomechanically, it acts as the primary invertor of the subtalar joint and a secondary plantarflexor of the ankle joint. By splitting the tendon and transferring its lateral half, we aim to selectively eliminate its pathological inversion moment while preserving its stabilizing plantarflexion contribution to the medial longitudinal arch.
Equally critical to understand is the gastrocsoleus complex, which drives the equinus component of the deformity. The gastrocnemius, a biarticular muscle originating from the posterior femoral condyles, and the soleus, a uniarticular muscle originating from the proximal tibia and fibula, converge to form the Achilles tendon. Differentiating the contribution of each muscle to the equinus contracture via the Silfverskiöld test is a fundamental prerequisite for surgical planning. When the knee is extended, both muscles are under tension; when the knee is flexed, the gastrocnemius is relaxed, isolating the soleus. This anatomical distinction dictates whether a gastrocnemius recession or a comprehensive Achilles tendon lengthening is required concomitantly with the tendon transfer.
Navigating the neurovascular anatomy is the most perilous aspect of this operation. The posterior tibial neurovascular bundle—comprising the posterior tibial artery, posterior tibial nerve, and paired venae comitantes—is the critical structure at risk during the posterior routing of the tendon. This bundle courses immediately lateral and slightly posterior to the tibialis posterior tendon, deep to the deep transverse fascia of the leg. Iatrogenic injury to this bundle during blind tunneling can precipitate catastrophic complications, including ischemic necrosis of the plantar foot, profound paresthesias, and paralysis of the intrinsic pedal musculature. Furthermore, the surgeon must remain hyper-vigilant of the saphenous nerve and vein medially, and the sural nerve laterally, as these superficial structures are highly vulnerable during the initial surgical exposures.
Exhaustive Indications and Contraindications
The decision to proceed with a Split Posterior Tibial Tendon Transfer requires meticulous patient selection. The primary indication is a dynamic, flexible equinovarus foot deformity that significantly impairs ambulation, brace tolerance, or overall functional mobility. The deformity must be demonstrably driven by an overactive or spastic tibialis posterior muscle. A critical prerequisite is that the varus deformity must be passively correctable to at least a neutral hindfoot alignment. If the hindfoot cannot be passively brought out of varus, soft tissue transfer alone will inevitably fail, and concomitant bony procedures (such as a calcaneal osteotomy) are strictly mandated.
Furthermore, the patient should ideally demonstrate some degree of selective motor control. While pure isolation is rare in spastic cerebral palsy, the presence of voluntary, albeit synergistic, activation of the muscle groups suggests a higher likelihood of successful postoperative neuromuscular re-education. Gait analysis demonstrating continuous swing and stance phase firing of the tibialis posterior, coupled with lateral forefoot initial contact and a varus thrust during stance, provides objective, quantifiable data supporting the intervention. The procedure is generally delayed until the child is at least 6 to 8 years of age; performing the transfer in younger, less neurologically mature children carries an unacceptably high rate of recurrence or overcorrection as their gait pattern continues to evolve.
Contraindications must be rigorously respected to avoid disastrous outcomes. A rigid, fixed bony deformity that cannot be corrected with adjunctive osteotomies is an absolute contraindication for a standalone soft tissue transfer; such cases require salvage procedures like a triple arthrodesis. Severe, unremitting athetosis or dystonia represents a relative to absolute contraindication, as unpredictable, fluctuating muscle tone will rapidly overpower the transfer, leading to failure or unpredictable secondary deformities. Additionally, complete absence of tibialis posterior activity on dynamic electromyography (EMG) contraindicates the procedure, as transferring a non-functioning muscle will yield no dynamic balancing benefit.
The decision between a split transfer (SPTTT) and a whole tendon transfer is also critical. Transferring the entire tibialis posterior tendon is generally contraindicated in ambulatory patients with cerebral palsy due to the exceptionally high risk of catastrophic overcorrection into a severe, progressive planovalgus deformity. The split transfer elegantly circumvents this by leaving the medial half of the tendon attached to the navicular, thereby maintaining crucial support for the medial longitudinal arch while simultaneously providing an eversion force via the transferred lateral half.
| Category | Split Posterior Tibial Tendon Transfer (SPTTT) Parameters |
|---|---|
| Primary Indications | Dynamic, flexible equinovarus deformity; Spastic hemiplegia/diplegia with swing-phase inversion; Intolerance to Ankle-Foot Orthosis (AFO) due to lateral pressure. |
| Prerequisites | Passively correctable hindfoot varus (to neutral or valgus); Patient age > 6-8 years; Demonstrable tibialis posterior activity on EMG/clinical exam. |
| Absolute Contraindications | Rigid, fixed bony deformity (uncorrectable by osteotomy); Severe athetosis or dystonia; Flaccid paralysis of the tibialis posterior. |
| Relative Contraindications | Poor selective motor control; Severe midfoot breakdown; Prior extensive posterior compartment trauma/scarring. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the bedrock upon which surgical success is built. The clinical assessment must go far beyond a simple range of motion check. We meticulously employ the modified Ashworth scale to quantify spasticity in the gastrocsoleus and tibialis posterior. Observational gait analysis is mandatory, but whenever feasible, three-dimensional instrumented gait analysis should be utilized. This provides invaluable kinematic and kinetic data, alongside dynamic EMG, definitively confirming whether the tibialis posterior is firing continuously or out of phase. Pedobarography is also highly useful, often revealing a pathological concentration of pressure under the lateral midfoot and fifth metatarsal base, confirming the varus weight-bearing axis.
Radiographic evaluation is essential, even in seemingly flexible deformities, to rule out subtle structural adaptations. Weight-bearing anteroposterior (AP) and lateral radiographs of the foot and ankle are standard. On the AP view, we assess the talocalcaneal angle (Kite's angle), which is typically narrowed in varus deformities. The lateral view allows assessment of Meary's angle to evaluate the longitudinal arch and rule out a subtle cavus component. A Harris axial heel view is absolutely critical to accurately quantify the degree of coronal plane hindfoot varus and to assess the morphology of the subtalar joint. If a bony procedure is anticipated, preoperative templating for calcaneal osteotomy wedges or displacement distances is performed on these radiographs.
The Examination Under Anesthesia (EUA) is the final, definitive phase of preoperative planning, performed immediately prior to incision. By completely eliminating spasticity, the EUA allows the surgeon to discern the true extent of myostatic contracture. The Silfverskiöld test is repeated to finalize the decision between gastrocnemius recession and Achilles lengthening. The hindfoot is aggressively manipulated to confirm that it can be passively overcorrected into valgus. If residual varus persists despite maximal force under anesthesia, the surgical plan must be immediately adapted to include a lateral closing wedge or sliding calcaneal osteotomy.
Patient positioning must facilitate circumferential access to the lower extremity. The patient is positioned strictly supine on the operating table. A bump may be placed under the ipsilateral hip to correct for external rotation and present the medial aspect of the leg more favorably, though the leg must remain freely mobile. All bony prominences are meticulously padded. A well-padded pneumatic thigh tourniquet is applied to ensure a pristine, bloodless surgical field, which is absolutely vital for the safe identification and protection of the posterior tibial neurovascular bundle. The entire lower extremity is prepped and draped free from the toes to the proximal thigh, allowing the surgeon to dynamically assess tension, alignment, and range of motion throughout the procedure.
Step-by-Step Surgical Approach and Fixation Technique
The operative execution of the SPTTT demands precision, respect for soft tissues, and an intimate knowledge of the posterior compartment anatomy. We will describe the posterior routing technique (Kaufer technique), which is our preferred method for balancing inversion-eversion forces.
Medial Exposure and Tendon Splitting
The procedure begins with a 4 to 5 cm longitudinal incision centered directly over the prominent tuberosity of the navicular. Careful subcutaneous dissection protects the terminal branches of the saphenous nerve. The deep fascia is incised, and the robust sheath of the tibialis posterior tendon is opened longitudinally, revealing the tendon at its primary insertion site.
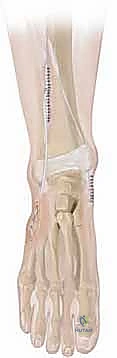
Using a #15 blade, the tendon is meticulously dissected from its broad plantar insertions. We aim to release the plantar/lateral half of the tendon from the navicular and cuneiforms, preserving the dorsal/medial half to maintain the medial longitudinal arch.
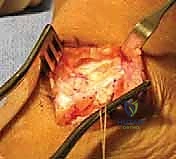
Once the lateral half is freed distally, it is immediately secured with a running, locking whipstitch using a heavy, non-absorbable suture (e.g., #0 or #1 Ethibond). This suture tag provides absolute control over the tendon slip during the subsequent splitting process.

Maintaining tension on the tagged lateral half, a #15 blade is used to carefully split the tibialis posterior tendon longitudinally, following its natural fiber orientation. This split is carried proximally within the tendon sheath, extending behind the medial malleolus and up into the distal leg.
Proximal Medial Dissection and Tendon Retrieval
A second longitudinal incision, approximately 5 cm in length, is made on the posteromedial aspect of the distal third of the leg, positioned 1 to 2 cm posterior to the palpable medial border of the tibia.
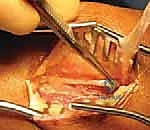
Sharp dissection is carried down through the subcutaneous tissue and the deep transverse fascia. The muscle belly and proximal musculotendinous junction of the tibialis posterior are identified deep in the compartment. The longitudinal split previously initiated distally is identified and meticulously extended proximally into the musculotendinous junction.
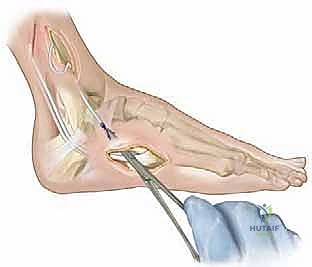
Using steady, gentle traction on the distal suture tag, the freed lateral half of the tendon is smoothly pulled proximally, delivering it out of the proximal medial wound. It is imperative to ensure the split extends high enough into the muscle belly to allow for a direct, unobstructed line of pull during the subsequent transfer, preventing any tethering or kinking of the muscle fibers.
Lateral Exposure and Posterior Routing
Attention is then directed to the lateral aspect of the foot. A 4 cm longitudinal incision is made over the lateral aspect of the distal fibula, extending distally toward the base of the fifth metatarsal, following the course of the peroneal tendons.
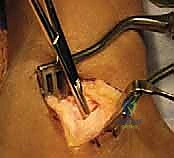
The sural nerve must be identified and protected during this exposure. The peroneal tendon sheath is incised, and the peroneus brevis tendon is clearly identified and isolated.
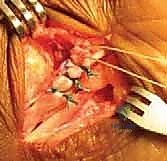
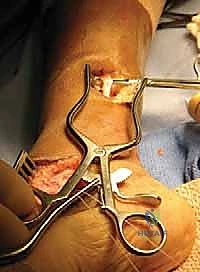
The most critical step of the procedure—the posterior routing—now commences. A large, blunt tendon passer (or a curved Kelly clamp) is introduced through the lateral incision, passing deep to the peroneal tendons, and directed carefully posterior to the fibula and tibia.
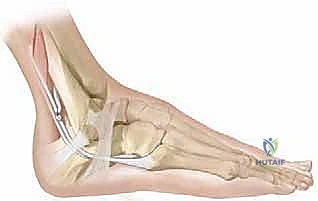
The passer must glide anterior to the soleus muscle belly but, crucially, anterior to the posterior tibial neurovascular bundle. The tip of the passer is delivered into the proximal medial wound. The suture tag of the split tibialis posterior is grasped by the passer.
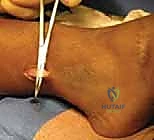
With extreme care, the tendon is gently pulled through the newly created posterior tunnel and delivered into the lateral wound. The surgeon must verify that the tendon glides smoothly without catching, ensuring it is not wrapped around the neurovascular bundle or tethered by fascial bands.
Tendon Weave, Tensioning, and Fixation
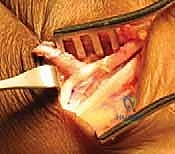
With the transferred tendon slip now residing laterally, the recipient peroneus brevis tendon is prepared.
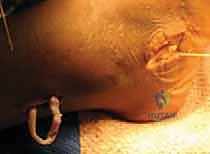
A longitudinal slit is created in the substance of the peroneus brevis tendon to facilitate a robust Pulvertaft weave. Alternatively, a side-to-side anastomosis can be utilized if tendon length is a limiting factor, though the weave provides superior biomechanical pull-out strength.
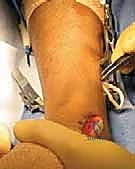
Setting the correct tension is the most challenging and subjective aspect of the operation. The ankle is held in neutral dorsiflexion (0 degrees), and the hindfoot is positioned in approximately 5 to 10 degrees of eversion.
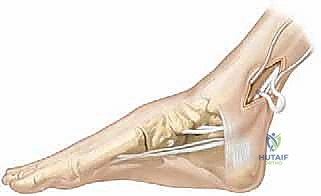
While an assistant rigidly maintains this precise position, the transferred tibialis posterior tendon is pulled distally under maximal physiological tension to remove all crimp, woven through the peroneus brevis, and sutured securely upon itself and the recipient tendon using multiple interrupted 2-0 non-absorbable braided sutures.
The final construct is visually inspected. The surgeon should be able to passively invert the foot, but upon release, the tension of the transfer should immediately spring the foot back to the neutral or slightly everted resting posture.
Once hemostasis is achieved following tourniquet deflation, the wounds are copiously irrigated and closed in layers. Subcutaneous tissue is approximated with 3-0 absorbable sutures, and the skin is closed with a running subcuticular stitch or interrupted nylons.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, the SPTTT carries a distinct complication profile that the orthopedic surgeon must be prepared to manage. The most frequent and concerning complication is overcorrection, leading to an iatrogenic planovalgus deformity. This occurs far more frequently when the entire tendon is transferred, which is why the split technique is heavily favored. Overcorrection can also result from over-lengthening of the Achilles tendon, leading to a calcaneovalgus posture and a devastating crouch gait pattern. If mild, overcorrection can be managed with orthotics (UCBL or AFO). However, severe, progressive valgus requires surgical salvage, often necessitating medial column plication, lateral column lengthening (Evans osteotomy), or subtalar arthrodesis.
Undercorrection or recurrence of the equinovarus deformity is another significant risk, particularly if the procedure is performed on children under the age of 8, or if an unrecognized fixed bony deformity was present preoperatively. Recurrence may also stem from inadequate tensioning of the transfer or failure to adequately lengthen the gastrocsoleus complex. Salvage for recurrent varus typically involves a return to the operating room for a lateral closing wedge calcaneal osteotomy, revision of the Achilles lengthening, and potentially a secondary soft tissue transfer if the initial transfer has ruptured or stretched out.
Neurovascular injury is the most catastrophic intraoperative complication. Blind passage of the tendon passer posterior to the tibia can easily ensnare or lacerate the posterior tibial artery or nerve. This manifests postoperatively as profound ischemia of the plantar foot, loss of sensation, or intrinsic muscle paralysis. Avoidance is paramount: the passer must stay directly on the posterior periosteum of the tibia and fibula. If vascular compromise is suspected postoperatively, immediate vascular surgery consultation and possible exploration are mandatory. Tendon pull-out or rupture at the anastomosis site is rare if a proper Pulvertaft weave is utilized and the patient is strictly immobilized, but it requires prompt surgical revision if it occurs.
| Complication | Estimated Incidence | Avoidance Strategy | Salvage Management |
|---|---|---|---|
| Overcorrection (Planovalgus) | 5% - 15% | Utilize split transfer; Avoid over-lengthening Achilles; Tension in neutral, not excessive eversion. | Orthotics; Medial column plication; Lateral column lengthening; Subtalar arthrodesis. |
| Undercorrection / Recurrence | 10% - 20% | Delay surgery until age > 8; Ensure rigid bony varus is treated with osteotomy; Adequate tensioning. | Calcaneal osteotomy (Dwyer); Revision Achilles lengthening; Triple arthrodesis (in adults). |
| Neurovascular Injury | < 1% | Direct visualization during proximal dissection; Keep tendon passer flush against posterior bone. | Immediate surgical exploration; Vascular repair; Nerve grafting/repair. |
| Tendon Pull-out / Rupture | 1% - 3% | Robust Pulvertaft weave; Non-absorbable sutures; Strict 6-week cast immobilization. | Surgical re-exploration and revision of the tendon anastomosis. |
Phased Post-Operative Rehabilitation Protocols
The immediate postoperative phase prioritizes the absolute protection of the tendon transfer and any concomitant osteotomies. In the operating room, before the patient awakens, a well-padded, rigid short leg cast is applied with the ankle locked in neutral dorsiflexion and the hindfoot in neutral to 5 degrees of eversion. This cast remains in place for a minimum of 6 weeks. During this initial phase, the patient is strictly non-weight-bearing on the operative extremity to prevent any tension across the healing tendon anastomosis. Deep vein thrombosis (DVT) prophylaxis is instituted based on patient age and risk factors, and adequate analgesia/antispasmodic medications (e.g., Diazepam) are provided to prevent violent muscle spasms from disrupting the repair.
At the 6-week mark, the cast is removed, and clinical healing is assessed. The patient is then transitioned into a rigid, custom-molded Ankle-Foot Orthosis (AFO) or a controlled ankle motion (CAM) boot. The rehabilitation protocol shifts to the transition phase, where weight-bearing is gradually advanced as tolerated over the next 2 to 4 weeks. Physical therapy is initiated immediately upon cast removal. The initial focus is on gentle, passive, and active-assisted range of motion exercises for the ankle and subtalar joints to prevent debilitating stiffness, taking care not to aggressively stretch the transfer into inversion.
The active rehabilitation phase, commencing around 8 to 10 weeks postoperatively, focuses heavily on neuromuscular re-education. The patient must essentially "re-learn" how to fire the transferred lateral half of the tibialis posterior in its new biomechanical role as an evertor. Biofeedback and targeted strengthening exercises for the peroneal musculature and the anterior compartment are critical. Gait training is paramount to integrate the newly balanced foot into a functional, reciprocal gait pattern. The patient will typically require the AFO for ambulation for at least 6 to 12 months postoperatively to
