Masterclass: Open Dorsal Approach Scaphoid Fracture Fixation with Corticocancellous Bone Grafting
Key Takeaway
This masterclass details dorsal scaphoid fracture fixation with bone grafting. Fellows will learn preoperative planning, precise anatomical dissection, K-wire guidance, and meticulous hardware placement. We cover neurovascular risks, pearls for successful union, and comprehensive postoperative care, ensuring optimal patient outcomes for challenging scaphoid nonunions or acute fractures.
Comprehensive Introduction and Patho-Epidemiology
The management of scaphoid nonunions represents one of the most formidable challenges in hand and upper extremity surgery, demanding an exacting synthesis of biomechanical understanding, anatomical fluency, and technical precision. When we encounter a Stage IIIB nonunion of the scaphoid waist, characterized by significant bone loss, sclerosis, and a classic "humpback" kyphotic deformity, the surgical stakes are exceptionally high. This pathology typically arises in young, active patients—often following a high-energy fall on an outstretched hand (FOOSH) that was either misdiagnosed initially or failed conservative management. The definitive intervention necessitates an open reduction, internal fixation (ORIF), and the meticulous harvest and interposition of a corticocancellous bone graft to restore both biology and architecture.
Epidemiologically, the scaphoid is the most frequently fractured carpal bone, accounting for approximately 60% to 70% of all carpal fractures. The peak incidence occurs in males between the ages of 15 and 30, a demographic characterized by high participation in contact sports and vocational activities prone to high-velocity trauma. Because the initial radiographic presentation of a scaphoid fracture can be notoriously occult, up to 10% to 15% of these injuries progress to nonunion. The progression to a Stage IIIB nonunion specifically implies not just a failure of osteogenesis, but a structural collapse of the scaphoid architecture, leading to a profound disruption of the entire carpal kinematic chain.
The pathophysiology of scaphoid nonunion is intrinsically linked to its precarious vascular anatomy. The scaphoid relies on a retrograde blood supply, with the primary arterial inflow entering via the dorsal ridge and supplying the proximal pole in a distal-to-proximal direction. Consequently, fractures at the waist or proximal pole critically interrupt this vascular tether, rendering the proximal fragment highly susceptible to avascular necrosis (AVN). As the nonunion persists, cyclical loading across the wrist joint exacerbates the mechanical instability. The distal scaphoid fragment tends to flex volarly under the pull of the radioscaphocapitate ligament and the trapezium, while the proximal fragment extends with the lunate, driven by the intact scapholunate interosseous ligament.
This divergent rotation results in the pathognomonic "humpback" deformity of the scaphoid and a secondary Dorsal Intercalated Segment Instability (DISI) pattern of the carpus. If left untreated, this altered biomechanical environment inevitably leads to Scaphoid Nonunion Advanced Collapse (SNAC), a predictable cascade of degenerative radiocarpal and midcarpal arthritis. Therefore, the utilization of a dorsal approach combined with structural corticocancellous bone grafting is not merely a method of achieving union; it is a vital reconstructive procedure designed to restore the volar carpal length, correct the DISI deformity, and preserve the longevity of the radiocarpal articulation.
Detailed Surgical Anatomy and Biomechanics
Osteology and Carpal Kinematics
The scaphoid functions as the critical mechanical linkage between the proximal and distal carpal rows, essentially serving as the tie rod of the wrist. Its complex, twisted-peanut geometry is almost entirely covered by articular cartilage, leaving only limited non-articular regions at the dorsal ridge and the volar tubercle for ligamentous attachments and vascular ingress. The scaphoid articulates proximally with the elliptical fossa of the distal radius, medially with the lunate and capitate, and distally with the trapezium and trapezoid. This multi-faceted articulation dictates that any alteration in scaphoid morphology—such as the foreshortening seen in a humpback nonunion—will profoundly derange the contact mechanics of the entire wrist joint.
Biomechanically, the scaphoid is subjected to immense shear and bending forces during normal wrist motion and load-bearing. During wrist extension and ulnar deviation, the scaphoid assumes an extended posture, whereas during wrist flexion and radial deviation, it flexes volarly. This dynamic motion is essential for the "dart thrower's motion," the primary functional axis of the human wrist. When a waist fracture occurs, the structural integrity of this linkage is lost. The distal fragment flexes, and the proximal fragment extends, creating a localized kyphosis.
Correcting this kyphosis is the paramount mechanical goal of the procedure. The insertion of a volar wedge or a dorsal structural corticocancellous graft acts as a mechanical strut, forcibly restoring the scaphoid to its anatomical length and alignment. This restoration of the radial column length subsequently realigns the lunate out of its extended DISI posture, thereby normalizing the load transmission across the radiocarpal and midcarpal joints. Failure to adequately restore this geometry, even if osseous union is achieved, will result in persistent kinematic dysfunction and accelerated degenerative changes.
Vascular Anatomy and Perfusion
A masterful comprehension of the scaphoid's vascularity is non-negotiable for the orthopedic surgeon, as it dictates both the pathophysiology of the nonunion and the surgical approach. The extraosseous blood supply is predominantly derived from the radial artery. The most critical vessel is the dorsal carpal branch of the radial artery, which gives rise to multiple small nutrient vessels that penetrate the scaphoid at the dorsal ridge, located just distal to the articular surface for the radius. These dorsal vessels supply approximately 70% to 80% of the scaphoid, including the entire proximal pole and the majority of the waist, via a retrograde intraosseous network.
The volar blood supply is less dominant, originating from the superficial palmar arch and the volar carpal branches of the radial artery. These vessels enter the scaphoid at the distal tubercle and supply only the distal 20% to 30% of the bone. This unique anatomical arrangement explains why proximal pole fractures have an exceptionally high rate of avascular necrosis and nonunion. The retrograde flow is easily disrupted by the fracture line, leaving the proximal fragment completely isolated from its vascular source.
During a dorsal surgical approach, the surgeon must be acutely aware of this vascular anatomy. While the approach provides unparalleled access to the proximal pole and the dorsal ridge, aggressive stripping of the dorsal capsule and periosteum can further compromise the fragile remaining blood supply to the scaphoid. The dissection must be meticulously precise, preserving the capsular attachments where possible and avoiding indiscriminate electrocautery near the dorsal ridge. The rationale for using a corticocancellous graft in this avascular environment is twofold: the cancellous portion provides vital osteoinductive and osteoconductive elements, while the cortical strut provides the necessary mechanical stability to protect the revascularization process.
Dorsal Extensor Compartments and Neurovascular Structures
Navigating the dorsal wrist requires a precise mental map of the six extensor compartments and their associated neurovascular structures. The dorsal approach relies on exploiting the anatomical intervals between these compartments. Compartment 1 (abductor pollicis longus and extensor pollicis brevis) and Compartment 2 (extensor carpi radialis longus and brevis) form the radial border of our primary working zone. Compartment 4 (extensor digitorum communis and extensor indicis proprius) forms the ulnar border. The critical landmark is Lister's tubercle, a bony prominence on the dorsal distal radius that acts as a pulley for the contents of Compartment 3.
Compartment 3 houses the extensor pollicis longus (EPL) tendon. The EPL takes a sharp 45-degree turn around Lister's tubercle to insert on the distal phalanx of the thumb. In the dorsal approach to the scaphoid, opening Compartment 3 and transposing the EPL radially is the key maneuver. This transposition creates a safe, avascular window directly over the dorsal wrist capsule and the underlying scaphoid, without necessitating the retraction of the bulky muscle bellies of the EDC.
Superficial to the extensor retinaculum, the surgeon must identify and protect the superficial branch of the radial nerve (SBRN). The SBRN emerges from beneath the brachioradialis in the distal third of the forearm and arborizes over the radial dorsum of the wrist, providing critical sensation to the dorsal thumb and radial digits. Iatrogenic injury to the SBRN, either through direct transection or aggressive traction, can result in debilitating neuromas and complex regional pain syndrome (CRPS). Deep to the extensor compartments, resting directly on the dorsal wrist capsule, lies the posterior interosseous nerve (PIN). The PIN is a terminal sensory branch of the radial nerve that innervates the dorsal capsule. Routine identification and prophylactic neurectomy of the PIN during capsulotomy are advocated by many surgeons to denervate the joint and alleviate chronic wrist pain associated with the nonunion.
Exhaustive Indications and Contraindications
The decision to proceed with an open dorsal approach, structural corticocancellous bone grafting, and internal fixation for a scaphoid nonunion is highly nuanced. It requires a comprehensive evaluation of the fracture morphology, the chronicity of the nonunion, the viability of the proximal pole, and the patient's physiological and functional status. This approach is not a panacea; it is a highly specific, technically demanding procedure reserved for distinct pathological patterns where percutaneous or purely cancellous grafting techniques would inevitably fail.
The primary indication for this procedure is a Stage IIIB scaphoid nonunion, characterized by a significant structural defect, a foreshortened scaphoid waist, and a humpback deformity. In these cases, the dorsal approach allows for excellent visualization of the proximal pole and the nonunion interface. The structural corticocancellous graft is mandatory here; cancellous bone alone lacks the mechanical rigidity required to correct the kyphosis and maintain the restored radial column length against the compressive forces of the carpus. Furthermore, proximal pole nonunions, even without significant deformity, are often best approached dorsally due to the anatomical proximity of the proximal pole to the dorsal rim of the radius, allowing for direct, perpendicular screw trajectory.
Conversely, absolute contraindications must be strictly respected to avoid catastrophic outcomes. The most critical contraindication is the presence of advanced Scaphoid Nonunion Advanced Collapse (SNAC) arthritis. If irreversible degenerative changes have occurred in the radioscaphoid or midcarpal joints (SNAC Stage II or III), attempting to reconstruct the scaphoid is futile and will likely exacerbate the patient's pain. In such scenarios, salvage procedures like a proximal row carpectomy (PRC) or a four-corner fusion are indicated. Active local or systemic infection is an absolute contraindication to any internal fixation or bone grafting procedure.
Relative contraindications revolve around patient-specific factors that severely compromise osteogenesis. Active tobacco smoking is perhaps the most significant modifiable risk factor; nicotine induces potent vasoconstriction and cellular hypoxia, drastically increasing the risk of graft failure and persistent nonunion. Many surgeons require documented smoking cessation prior to undertaking complex scaphoid reconstruction. Additionally, severe medical comorbidities, uncontrolled diabetes mellitus, and a demonstrated inability to comply with strict postoperative immobilization protocols are strong relative contraindications that must be carefully weighed against the potential benefits of the surgery.
| Category | Specific Criteria | Clinical Rationale and Impact |
|---|---|---|
| Primary Indications | Stage IIIB Scaphoid Waist Nonunion | Requires structural graft to bridge the defect and restore carpal length. |
| Humpback Deformity with DISI | Corticocancellous strut is necessary to mechanically correct the kyphosis. | |
| Proximal Pole Nonunions | Dorsal approach provides optimal visualization and direct screw trajectory. | |
| Failed Previous Fixation | Requires robust structural revision and rigid headless compression. | |
| Absolute Contraindications | SNAC Stage II or III Arthritis | Scaphoid reconstruction will not reverse established radiocarpal degeneration. |
| Active Local or Systemic Infection | High risk of osteomyelitis and catastrophic hardware failure. | |
| Avascular Necrosis of Proximal Pole (Severe) | May necessitate vascularized bone grafting rather than non-vascularized. | |
| Relative Contraindications | Active Tobacco Smoking | Profoundly impairs microvascular ingrowth and osteogenesis. |
| Patient Non-Compliance | Inability to adhere to strict non-weight-bearing and cast immobilization. | |
| Severe Osteopenia/Osteoporosis | Compromises hardware purchase and increases risk of screw pull-out. |
Pre-Operative Planning, Templating, and Patient Positioning
Advanced Imaging and Templating
The foundation of a successful scaphoid reconstruction is laid long before the patient enters the operating theater, beginning with an exhaustive radiographic analysis. Standard plain radiographs—including a posteroanterior (PA), true lateral, pronated oblique, and a specific scaphoid view (PA with the wrist in ulnar deviation and extension)—are mandatory. These views allow for the initial assessment of the nonunion site, the degree of sclerosis, the presence of cystic changes, and the overall carpal alignment. The true lateral view is particularly critical for evaluating the radiolunate angle and identifying the presence of a Dorsal Intercalated Segment Instability (DISI) deformity, which dictates the need for structural deformity correction.
However, plain radiography is frequently insufficient for complex nonunions. A high-resolution computed tomography (CT) scan is the gold standard for preoperative planning. The CT must be reformatted along the true longitudinal and sagittal axes of the scaphoid, rather than the anatomical planes of the wrist. This scaphoid-specific reformatting provides a precise volumetric assessment of the bone defect, the exact angle of the humpback deformity, and the presence of any bridging trabeculae. Furthermore, Magnetic Resonance Imaging (MRI), particularly with gadolinium contrast, is highly recommended to evaluate the vascular status of the proximal pole. A completely avascular proximal pole may alter the surgical plan, pushing the surgeon toward a vascularized bone graft rather than the non-vascularized corticocancellous graft discussed herein.
Digital templating is the final step in the imaging review. Using the sagittal CT reformats, the surgeon must calculate the exact dimensions of the corticocancellous graft required to restore the scaphoid to its pre-injury length and morphology. The graft is typically templated as a trapezoidal wedge. Additionally, the trajectory and length of the headless compression screw must be templated. The ideal trajectory runs down the central axis of the scaphoid, maximizing thread purchase in both the proximal and distal fragments. Templating prevents intraoperative guesswork, minimizes the risk of articular penetration, and ensures that the appropriate hardware inventory is available in the suite.
Patient Positioning and Operating Room Setup
Optimal patient positioning and operating room ergonomics are critical for minimizing surgical fatigue and ensuring precise execution of this technically demanding procedure. The patient is placed in the supine position on a standard operating table. A radiolucent hand table extension is securely attached to the operative side. It is imperative that the hand table is completely radiolucent and free of any metallic underpinnings that could obstruct fluoroscopic visualization during critical maneuvering. A well-padded high-thigh tourniquet is applied to the proximal arm to provide a bloodless surgical field, which is absolutely essential for identifying fine neurovascular structures and assessing the quality of the debrided bone.
The patient's arm is abducted to approximately 90 degrees, and the elbow is flexed to 90 degrees. The forearm is placed in full pronation. This pronated posture naturally presents the dorsal aspect of the wrist to the surgeon, providing a direct, unobstructed line of sight to Lister's tubercle and the dorsal carpus. The surgeon typically sits in the axilla, facing cephalad, while the surgical assistant sits opposite, facing the surgeon. This arrangement allows both operators comfortable access to the dorsal wrist and facilitates the seamless passing of delicate instruments.
The setup of the fluoroscopy unit (C-arm) requires meticulous coordination with the radiology technician prior to draping. The C-arm should be positioned parallel to the longitudinal axis of the patient, entering from the distal end of the hand table. This configuration allows the C-arm to roll in and out of the field effortlessly without disrupting the surgical team. The surgeon must confirm that true AP and true lateral views of the scaphoid can be obtained purely by rotating the C-arm, minimizing the need to physically manipulate the patient's freshly reduced fracture. Establishing these perfect fluoroscopic arcs before the incision is made saves critical tourniquet time and prevents frustration during the fixation phase.
Step-by-Step Surgical Approach and Fixation Technique
Incision, Dissection, and Extensor Tendon Management
The procedure commences with the inflation of the tourniquet to 250 mmHg following exsanguination of the limb. A straight longitudinal incision, approximately 4 to 5 centimeters in length, is centered precisely over Lister's tubercle. While some surgeons advocate for lazy-S or transverse incisions for cosmetic reasons, the longitudinal incision provides the most extensile and utilitarian exposure for complex nonunions requiring concomitant distal radius bone graft harvest. The incision is carried sharply through the epidermis and dermis.
Subcutaneous dissection must proceed with extreme caution, utilizing blunt spreading with tenotomy scissors rather than aggressive electrocautery. The primary objective in this superficial layer is the identification and protection of the superficial branch of the radial nerve (SBRN). The SBRN typically courses obliquely across the radial aspect of the wrist, and its branches can inadvertently cross the surgical field. If encountered, the nerve should be gently mobilized and retracted radially with a vessel loop. The dissection is deepened to expose the glistening, transverse fibers of the extensor retinaculum.
The key to the dorsal approach is the management of the third extensor compartment. The extensor retinaculum is incised longitudinally directly over Compartment 3. The extensor pollicis longus (EPL) tendon is identified, mobilized, and transposed radially out of its tunnel. This maneuver is critical; it creates a vacant interval between the second compartment (ECRL/ECRB) and the fourth compartment (EDC/EIP). By gently retracting the EPL and the second compartment radially, and the fourth compartment ulnarly, the surgeon gains a wide, avascular window directly overlying the dorsal radiocarpal capsule and the scaphoid, without violating the muscle bellies or tenosynovium of the adjacent compartments.
Capsulotomy and Fracture Site Preparation
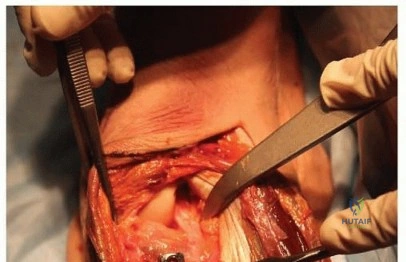
With the extensor tendons safely retracted, the dorsal wrist capsule is brought into full view. A longitudinal or ligament-sparing capsulotomy is performed. A common technique is an inverted T-shaped incision, with the vertical limb aligned with the axis of the scaphoid and the horizontal limb running parallel to the dorsal rim of the radius. During this capsular elevation, the posterior interosseous nerve (PIN), which lies directly on the capsule, is typically identified and resected to provide partial denervation of the wrist joint. The capsular flaps are elevated sharply off the dorsal carpus, taking care to preserve the dorsal radiocarpal and intercarpal ligaments to the greatest extent possible to maintain postoperative carpal stability.
Exposure of the scaphoid nonunion site usually reveals a chaotic landscape of fibrous tissue, inflamed synovium, and sclerotic bone. The humpback deformity is immediately apparent, with the scaphoid foreshortened and the distal pole flexed away from the surgeon. The absolute most critical step of the entire operation is the radical debridement of the nonunion site. Using a combination of fine curettes, small osteotomes, and a high-speed burr, the surgeon must meticulously excavate all fibrous interposition and sclerotic, avascular bone from both the proximal and distal fragments.
This debridement must continue relentlessly until healthy, punctate bleeding bone—often referred to as the "paprika sign"—is visualized on both faces of the fracture. Inadequate debridement is the leading cause of persistent nonunion following surgery. The surgeon must not be timid; it is better to create a larger defect that requires a larger bone graft than to leave sclerotic, biologically inert bone at the interface. Once debridement is complete, the true magnitude of the bone void and the extent of the humpback deformity can be accurately assessed.
Reduction Techniques and Deformity Correction
Correcting the structural collapse of the scaphoid is a biomechanical imperative. The surgeon employs the "K-wire joystick" technique to manipulate the fragments. A 0.062-inch Kirschner wire (K-wire) is driven into the proximal pole, and a second 0.062-inch K-wire is driven into the distal pole. These wires act as levers, allowing the surgeon to forcefully manipulate the fragments in three-dimensional space. The distal fragment is extended, and the proximal fragment is flexed, effectively opening the volar aspect of the nonunion site and correcting the kyphotic humpback deformity.
This maneuver simultaneously restores the overall length of the radial column and corrects the secondary DISI posture of the lunate. The reduction must be anatomically precise, akin to fitting two complex puzzle pieces together. The surgeon visually confirms the restoration of the scaphoid's dorsal anatomy and uses fluoroscopy to confirm the correction of the radiolunate angle and the scaphoid length on the true lateral view.
Once the optimal reduction is achieved, it must be provisionally stabilized. A third K-wire is driven obliquely across the fracture site, typically from the distal pole into the proximal pole. This provisional fixation wire must be placed peripherally so that it does not obstruct the central axis of the scaphoid, which is reserved for the definitive headless compression screw. The joystick K-wires can then be removed, and the reduction is rigorously interrogated under multi-planar fluoroscopy to ensure it is absolute and unyielding.
Bone Graft Harvest and Structural Interposition
With the defect prepared and the scaphoid anatomically reduced, the exact dimensions of the required bone graft are measured using a sterile caliper. Attention is then turned to the distal radius for the harvest of the corticocancellous graft. The harvest site is typically located just proximal to Lister's tubercle, within the same surgical incision. A small, rectangular cortical window is outlined using a fine osteotome or an oscillating saw, matching the templated dimensions of the scaphoid defect.
The cortical window is carefully elevated, and the underlying rich, cancellous bone of the distal radius metaphysis is harvested using a curette. The surgeon must harvest a single, contiguous block of corticocancellous bone. The structural integrity of the cortical shell is paramount, as it will act as the mechanical strut to maintain the corrected scaphoid length. The harvested graft is then meticulously contoured on the back table using a rongeur or a burr. It is typically shaped into a trapezoidal wedge, designed to fit precisely into the volar defect created by the correction of the humpback deformity.
The contoured graft is then press-fit into the nonunion site. The cortical surface of the graft is oriented either dorsally or volarly depending on the specific geometry of the defect, but it must provide rigid resistance against the compressive forces that attempt to recreate the kyphosis. The cancellous portion of the graft is packed tightly against the bleeding surfaces of the proximal and distal fragments. Additional morselized cancellous bone is packed around the structural graft to fill any remaining microscopic voids, maximizing the osteoconductive surface area and ensuring a robust biological environment for consolidation.
Definitive Fixation with Headless Compression Screw
The final phase of the intraoperative sequence is the definitive stabilization of the construct using a fully threaded, headless compression screw. The biomechanical superiority of headless screws lies in their ability to generate significant interfragmentary compression while burying the screw head beneath the articular cartilage, preventing impingement. The critical step is the placement of the guidewire. Using the dorsal approach, the guidewire is introduced at the dorsal ridge of the proximal pole, aiming distally and volarly toward the distal tubercle.
The trajectory must be perfectly aligned with the central, longitudinal axis of the scaphoid. The surgeon advances the guidewire under continuous, live fluoroscopic guidance in both the AP and lateral planes. The wire must traverse the proximal fragment, pass directly through the center of the corticocancellous graft, and achieve deep purchase into the distal fragment without breaching the articular surfaces of the radiocarpal or midcarpal joints. Once the guidewire is perfectly positioned, its length is measured using a dedicated depth gauge.
The near cortex is over-drilled, and the entire tract is prepared according to the specific screw system's protocol. The selected headless compression screw is then advanced over the guidewire. As the trailing threads of the screw engage the proximal pole, the differential pitch of the screw generates profound compression across the fracture interfaces and the interpositioned bone graft. The surgeon visually confirms the compression and ensures the trailing edge of the screw is countersunk at least 1-2 millimeters below the dorsal
