Distal Humeral Tumor Resection & Prosthetic Elbow Reconstruction: An Operative Masterclass

Key Takeaway
Join us in the OR for a masterclass on distal humeral tumor resection and prosthetic reconstruction. We'll meticulously cover intricate surgical anatomy, essential preoperative planning, and a granular, real-time breakdown of every intraoperative step. Learn critical techniques for neurovascular protection, precise osteotomy, modular prosthesis implantation, and soft tissue reconstruction, alongside crucial pearls and pitfalls for optimal patient outcomes.
Introduction and Epidemiology
Nature of Distal Humerus Neoplasms
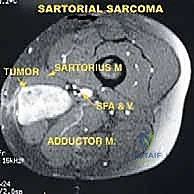
The distal humerus represents a relatively rare anatomic site for the development of primary bone sarcomas, such as osteosarcoma, Ewing sarcoma, and chondrosarcoma. More frequently, the distal humerus and the elbow joint are compromised by metastatic carcinoma (e.g., breast, lung, thyroid, renal, and prostate) or secondary invasion from soft tissue sarcomas (STS) arising in the adjacent musculature. Soft tissue sarcomas originating from the proximal flexor-pronator mass, the common extensor origin, or the distal brachialis and triceps muscles can envelop the distal humerus, necessitating en bloc resection of both the soft tissue mass and the underlying bone to achieve negative oncologic margins.
Evolution of Limb-Sparing Surgery
Historically, the complex juxtaposition of the distal humerus to critical neurovascular structures within the antecubital fossa mandated above-elbow amputation for local tumor control. However, advancements in neoadjuvant therapies, high-resolution cross-sectional imaging, and modular endoprosthetic design have made limb-sparing surgery the standard of care for most patients. The primary oncologic objective remains the complete en bloc resection of the tumor with negative margins. The reconstructive objective is the restoration of a stable, functional elbow joint through the use of a semiconstrained, hinged segmental tumor prosthesis combined with rigorous soft tissue reconstruction, including muscle rotation flaps and flexorplasty.



Surgical Anatomy and Biomechanics
Topographic and Compartmental Anatomy
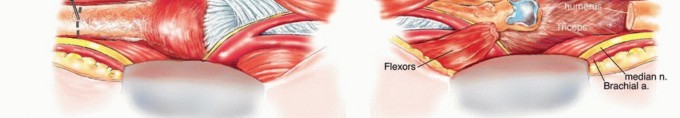
Safe oncologic resection in the distal arm requires a masterful understanding of the neurovascular topography. In the middle third of the arm, the primary neurovascular bundle is enveloped within a fibrous sheath situated in the medial bicipital groove, bordered anteriorly by the biceps brachii, posteriorly by the triceps brachii, and laterally by the brachialis muscle.
Arterial Anatomy
The brachial artery, a direct continuation of the axillary artery, courses distally along the medial aspect of the arm. At the lower border of the latissimus dorsi, it gives off the profunda brachii artery, which accompanies the radial nerve into the spiral groove. As the brachial artery enters the antecubital fossa, it rests directly on the anterior fascia of the brachialis muscle, positioned immediately lateral to the median nerve. It passes deep to the bicipital aponeurosis (lacertus fibrosus) before bifurcating into the radial and ulnar arteries. The inferior ulnar collateral artery, arising proximal to the joint line, frequently requires ligation during tumor mobilization.
Neural Anatomy
Median Nerve: The median nerve travels meticulously along the anterior aspect of the brachial artery in the proximal arm. As it descends toward the antecubital fossa, it crosses the artery anteriorly to reside on its medial side, entering the forearm between the humeral and ulnar heads of the pronator teres.
Ulnar Nerve: Positioned posteromedial to the brachial artery in the mid-arm, the ulnar nerve pierces the medial intermuscular septum in the distal third of the arm to enter the posterior compartment. It traverses the cubital tunnel posterior to the medial epicondyle, tightly tethered by the Osborne ligament, before entering the forearm deep to the flexor carpi ulnaris.
Radial Nerve: Arising from the posterior cord, the radial nerve courses with the profunda brachii artery through the triangular interval, wrapping around the posterior humerus in the spiral groove. It pierces the lateral intermuscular septum to enter the anterior compartment, descending between the brachialis and brachioradialis. Proximal to the supinator, it bifurcates into the superficial sensory radial nerve and the posterior interosseous nerve (PIN).
Medial Antebrachial Cutaneous Nerve: This sensory nerve runs deep to the fascia between the median and ulnar nerves proximally, piercing the deep fascia in the distal third of the arm to provide critical sensory innervation to the medial forearm. It should be preserved during flap elevation if oncologically feasible.
Indications and Contraindications
Indications for Resection and Reconstruction
The decision to proceed with distal humeral resection and endoprosthetic reconstruction is predicated on achieving local tumor control while preserving a functional limb. Primary indications include:
* High-grade and selected low-grade primary bone sarcomas of the distal humerus.
* Soft tissue sarcomas demonstrating direct invasion or circumferential encasement of the distal humerus.
* Solitary metastatic lesions isolated to the distal humerus.
* Destructive metastatic carcinomas where extensive cortical loss precludes standard curettage, cementation, and internal fixation.
* Salvage of severe local complications from previous treatments (e.g., irradiated nonunions, recalcitrant pathologic fractures, or massive osteomyelitis).
Absolute and Relative Contraindications
Absolute Contraindications: The primary absolute contraindication to limb salvage is the gross encasement of the main neurovascular bundle (brachial artery/vein complex) or the involvement of two or more major nerves. Note that the involvement of a single major nerve (e.g., radial nerve) is not an absolute contraindication; the nerve may be resected en bloc with the tumor, followed by appropriate tendon transfers for functional restoration.
Relative Contraindications: Extensive tumor contamination from a poorly placed prior biopsy, massive hematoma from a pathologic fracture, or active deep infection. However, pathologic fractures responding favorably to induction chemotherapy with resultant fracture stabilization and tumor necrosis may still be candidates for limb salvage, yielding local recurrence rates under 10%.
Diagnostic Imaging and Biopsy Principles
Advanced Imaging Modalities
Accurate preoperative staging and anatomic mapping are paramount.
* Plain Radiography: Essential for initial localization, differential diagnosis, and evaluating matrix mineralization. Following neoadjuvant chemotherapy, plain films can demonstrate peripheral ossification and healing of pathologic fractures, indicative of a favorable histologic response.
* Computed Tomography (CT): Highly sensitive for delineating cortical destruction and subtle periosteal reactions. Contrast-enhanced CT aids in mapping extraosseous soft tissue extension and is critical when evaluating metastatic lesions for prophylactic fixation versus en bloc resection.
* Magnetic Resonance Imaging (MRI): The gold standard for evaluating intramedullary tumor extent, skip metastases, and the precise relationship of the tumor pseudocapsule to the neurovascular structures. T1-weighted coronal and sagittal sequences dictate the level of the bone osteotomy, which must be planned 2 to 3 cm proximal to the proximal-most intramedullary signal abnormality.
* PET/CT and Thallium Scintigraphy: FDG-PET/CT is invaluable for systemic staging and detecting distant metastases. Quantitative Thallium-201 scintigraphy is utilized in specialized centers to assess tumor viability and chemotherapeutic necrosis (a ratio <4:1 compared to normal bone indicates >90% necrosis).
* Angiography: While largely supplanted by MR Angiography, traditional biplanar arteriography remains the gold standard for mapping tumor neovascularization (tumor blush) and identifying aberrant arterial anatomy prior to complex resections.
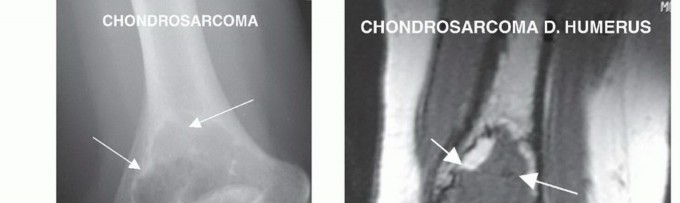
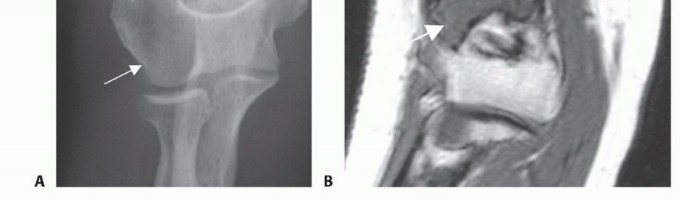
Oncologic Biopsy Techniques
The biopsy is a critical operative step. A poorly planned biopsy can convert a limb-sparing candidate into an amputee.
* Approach: The biopsy tract must be placed directly anterior, traversing the brachialis muscle in line with the planned definitive surgical incision.
* Biceps Preservation: The biopsy needle or incision must never violate the biceps brachii muscle. The biceps is the primary motor unit for postoperative elbow flexion; its contamination necessitates resection, severely compromising postoperative function.
* Soft Tissue Sarcomas: For tumors arising in the mobile wad (brachioradialis/extensors), biopsy the lateral-most aspect of the antecubital crease, meticulously avoiding the radial nerve. For flexor-pronator tumors, biopsy the medial-most extent, safeguarding the median nerve and brachial artery. All biopsy tracts must be excised en bloc during the definitive resection.
Pre Operative Planning and Patient Positioning
Surgical Planning and Templating
Preoperative templating utilizes full-length T1-weighted MRI and calibrated orthogonal radiographs. The exact level of the humeral osteotomy is calculated to ensure a minimum 2 to 3 cm margin of normal marrow. The surgeon must ensure the availability of a modular segmental distal humerus tumor prosthesis system, including multiple body segment lengths, stem diameters, and ulnar components. The ability to assemble and downsize the prosthesis intraoperatively (by 1 to 3 cm) is critical for achieving tension-free soft tissue coverage.
Patient Positioning
The patient is positioned supine on the operating table. A radiolucent bump is placed beneath the ipsilateral scapula to elevate the shoulder girdle, facilitating access to the posterior arm. The operative extremity is abducted and supported on a padded radiolucent hand table. The entire forequarter, from the sternal notch and clavicle to the fingertips, is prepped and draped free to allow circumferential access and dynamic intraoperative range of motion testing. A sterile tourniquet is generally avoided to allow continuous assessment of distal perfusion and to prevent compressive injury to neurovascular structures displaced by the tumor.
Detailed Surgical Approach and Technique
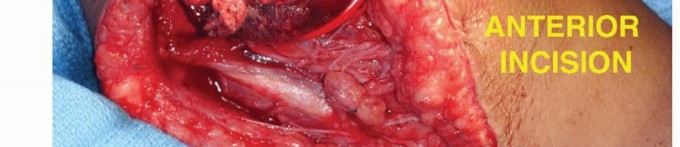
Incision and Initial Exposure
An extensile anterior S-shaped incision is utilized. The incision begins in the mid-arm along the medial border of the biceps muscle, extending distally to the antecubital crease. It incorporates the previous biopsy tract in an elliptical fashion. At the antecubital crease, the incision curves laterally across the volar aspect of the elbow toward the mobile wad, then extends distally into the proximal forearm. Thick, full-thickness fasciocutaneous flaps are raised medially and laterally to preserve the subdermal plexus and the medial/lateral antebrachial cutaneous nerves.
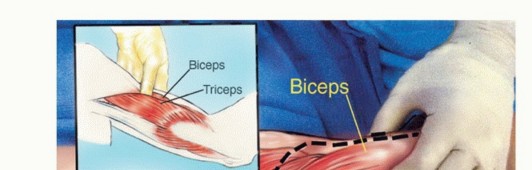
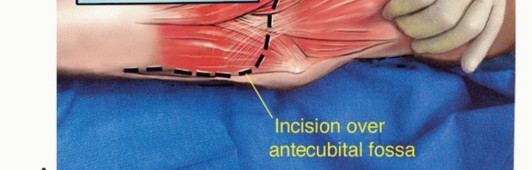

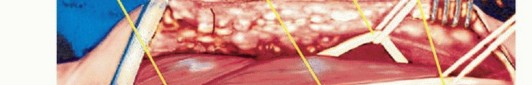
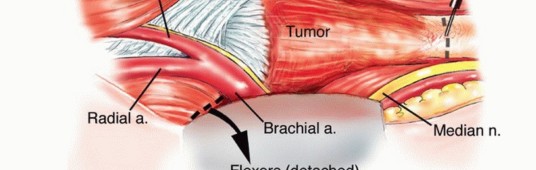
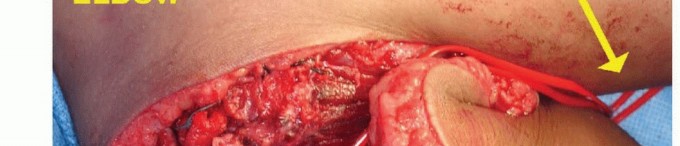
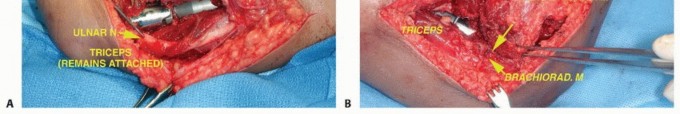
Meticulous Neurovascular Dissection
The dissection strictly proceeds from proximal (normal anatomy) to distal (distorted tumor anatomy). The deep investing fascia is incised longitudinally over the medial bicipital groove.
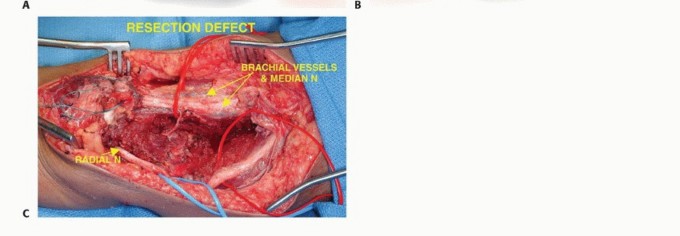
1. Vascular Isolation: The brachial artery and venae comitantes are identified, looped with vessel loops, and sharply dissected away from the tumor pseudocapsule. The bicipital aponeurosis is divided to trace the artery to its bifurcation. Muscular branches feeding the tumor or tethering the artery are ligated.
2. Median Nerve: Identified lateral to the proximal artery, traced distally as it crosses medially, and followed into the pronator teres.
3. Ulnar Nerve: Located posteromedial to the vascular bundle. The medial intermuscular septum is excised. The Osborne ligament is released, and the ulnar nerve is completely transposed anteriorly from the cubital tunnel to prevent traction injury during resection and subsequent prosthetic reconstruction.
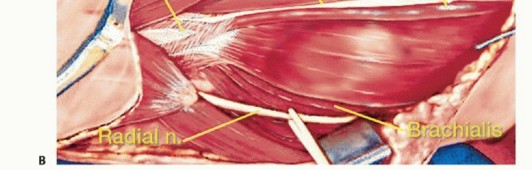
4. Radial Nerve: Identified in the interval between the brachialis and brachioradialis. The lateral intermuscular septum is opened, and the nerve is traced proximally into the spiral groove and distally to the arcade of Frohse.
Muscular Dissection and Bone Osteotomy
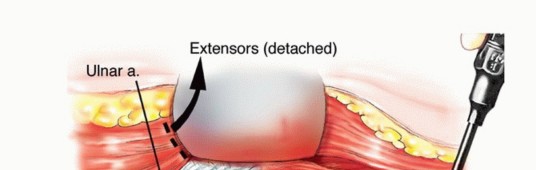
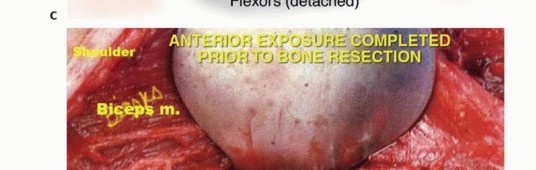
The biceps muscle is mobilized and retracted; it is rarely involved by primary bone tumors but must be protected. The brachialis muscle, lying directly over the anterior humerus, is often resected en bloc with the tumor to provide an adequate anterior soft tissue margin. The flexor-pronator origin is sharply detached from the medial epicondyle, and the mobile wad is released from the lateral epicondyle. The triceps is elevated posteriorly, preserving the lateral and long heads; the medial head may require partial resection.
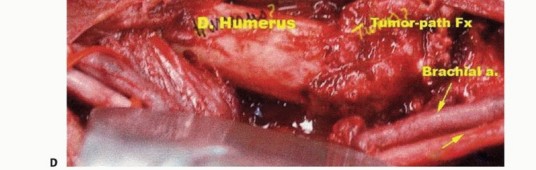
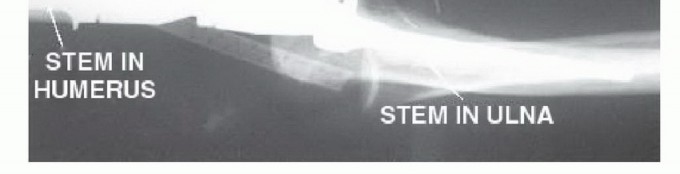
The elbow joint is entered anteriorly, and the capsule is released circumferentially. The humerus is osteotomized with an oscillating saw at the pre-calculated level (2-3 cm proximal to the MRI-defined tumor extent). The specimen is removed en bloc and sent for margin analysis.
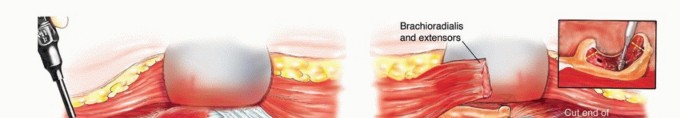
Endoprosthetic Reconstruction
Reconstruction utilizes a modular semiconstrained hinged elbow prosthesis.
1. Humeral Preparation: The proximal humeral canal is sequentially reamed using flexible reamers, over-reaming by 1-2 mm to allow for an adequate polymethylmethacrylate (PMMA) cement mantle.
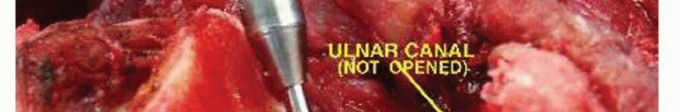
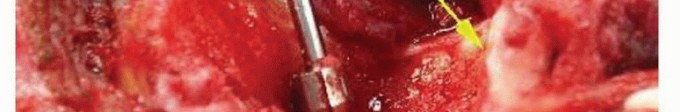
2. Ulnar Preparation: The olecranon fossa is decorticated using a high-speed burr to access the ulnar medullary canal. The proximal tip of the olecranon is contoured to allow straight-line access for the ulnar stem, preventing varus/valgus malalignment or posterior cortical perforation.
3. Cementation and Assembly: Trial components are inserted to verify length, rotation, and soft tissue tension. The prosthesis may be intentionally shortened by 1 to 3 cm to facilitate closure without excessive tension. The definitive stems are cemented. The humeral component must be meticulously rotated so the hinge axis is perfectly coronal (facing anteriorly). Once the cement cures, the hinge axle is engaged and secured.

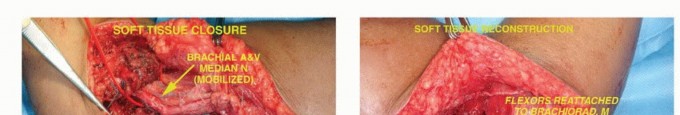
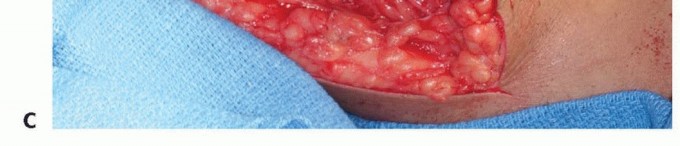
Soft Tissue Reconstruction and Flexorplasty
Robust soft tissue coverage is the primary defense against deep prosthetic infection. The elbow is positioned in 60 degrees of flexion and full supination.
* Flexorplasty: The origins of the flexor-pronator mass and the brachioradialis/extensor carpi radialis are advanced proximally and sutured directly to the medial and lateral borders of the biceps and remaining triceps using heavy (No. 5) nonabsorbable sutures.
* Biceps Retensioning: If the humerus was shortened, the biceps will be lax. It must be pulled distally under tension and sutured to the advanced forearm musculature to restore its length-tension relationship, which is critical for postoperative elbow flexion power.
* Closure: Deep closed-suction drains are placed. An epidural catheter may be placed adjacent to the brachial plexus for postoperative regional analgesia. The skin is closed meticulously without tension.


You Might Also Like