Upper Extremity Amputation: A Masterclass in Oncologic Reconstruction
Key Takeaway
Fellows, welcome to this masterclass on upper extremity amputations. We'll meticulously cover above-elbow and below-elbow techniques for advanced sarcomas, focusing on achieving wide oncologic margins while preserving maximal function. This session details preoperative planning, precise intraoperative dissection, crucial neurovascular management, robust myodesis, and comprehensive postoperative rehabilitation protocols to optimize patient outcomes.
Introduction and Epidemiology
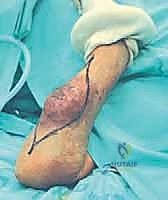
Amputation of the upper extremity, specifically at the above-elbow (transhumeral) and below-elbow (transradial) levels, constitutes a definitive and life-altering orthopaedic intervention. It is primarily indicated when limb salvage is surgically impossible, oncologically unsafe, or when the preservation of a severely compromised limb poses an imminent threat to patient survival. While lower extremity amputations are significantly more prevalent—often driven by peripheral vascular disease and diabetic neuropathy—upper extremity amputations account for a smaller percentage of all amputations but present profound challenges. The upper limb is intricately tied to fine motor function, tactile feedback, spatial manipulation, and psychosocial identity. Consequently, the loss of an upper extremity necessitates highly specialized surgical techniques to optimize the residual limb for complex prosthetic fitting and functional restoration.
Historically, the epidemiology of upper extremity amputation was dominated by battlefield trauma and industrial accidents. In contemporary orthopaedic and trauma surgery, the etiologic spectrum has evolved. High-energy trauma remains a leading cause, particularly involving crush injuries, severe avulsions, and mangled extremities where neurovascular disruption is irrecoverable. However, advanced soft tissue sarcomas and primary bone tumors (e.g., osteosarcoma, Ewing sarcoma) remain definitive indications for oncologic amputation, especially when these lesions cause extensive compartmental destruction, encase major neurovascular bundles, or demonstrate failure to respond to neoadjuvant chemoradiotherapy. While advances in limb-sparing techniques, such as endoprosthetic reconstruction and microvascular free tissue transfer, have drastically reduced the incidence of oncologic amputations, radical resection via amputation remains indispensable for achieving wide surgical margins and local tumor control in advanced or recurrent cases.
Beyond trauma and oncology, other critical indications include fulminant, life-threatening infections (e.g., necrotizing fasciitis, gas gangrene, refractory chronic osteomyelitis), intractable vascular compromise leading to irreversible ischemia, and rare congenital dysmelias that preclude functional reconstruction. The decision to proceed with an amputation requires a rigorous, multidisciplinary evaluation involving orthopaedic surgeons, vascular surgeons, oncologists, infectious disease specialists, prosthetists, and psychiatrists. The ultimate surgical objective transcends mere tissue removal; it is the meticulous creation of a durable, well-vascularized, and pain-free residual limb that maximizes the biomechanical lever arm and facilitates advanced prosthetic integration.
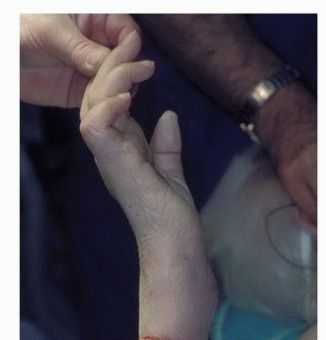
The psychosocial impact of upper extremity amputation cannot be overstated. Unlike lower extremity prostheses, which primarily restore the relatively automated function of ambulation, upper extremity prostheses must attempt to replicate the highly complex, multi-articulated prehensile functions of the human hand and arm. The rejection rate for upper extremity prostheses remains notoriously high, frequently cited between 20% and 40%. This rejection is often secondary to the weight of the prosthesis, discomfort at the socket interface, lack of sensory feedback, and the substantial cognitive load required to operate body-powered or early-generation myoelectric devices. Consequently, modern surgical epidemiology and technique have shifted toward procedures that actively prepare the residual limb for advanced bionic integration, such as Targeted Muscle Reinnervation (TMR) and osseointegration.
Surgical Anatomy and Biomechanics
A profound mastery of upper extremity surgical anatomy and biomechanics is non-negotiable for executing successful transhumeral and transradial amputations. The surgeon's goal is to construct a residual limb with robust soft tissue padding, preserved muscle integrity, and optimal bone length to facilitate prosthetic suspension, harness myoelectric signals, and maximize the mechanical lever arm.
Above Elbow Transhumeral Amputation Anatomy
Transhumeral amputations involve the transection of the humerus. The specific level is dictated by the pathology (e.g., necessary oncologic margins or extent of viable tissue in trauma). The humerus may be sectioned at the proximal metaphyseal (high), diaphyseal (mid-shaft), or supracondylar (low) levels. Each level carries distinct biomechanical implications for muscle reattachment and prosthetic socket design.
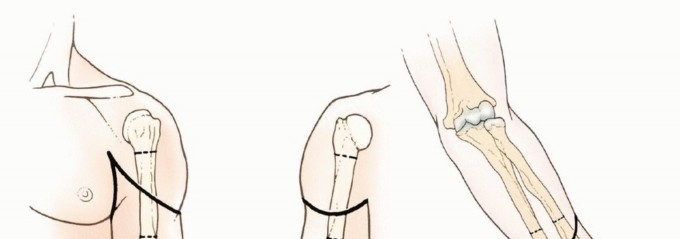
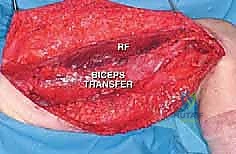
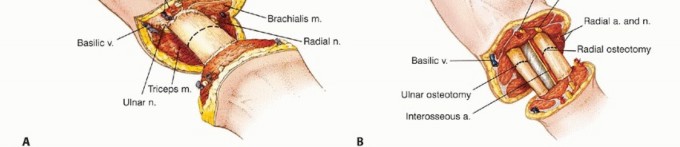
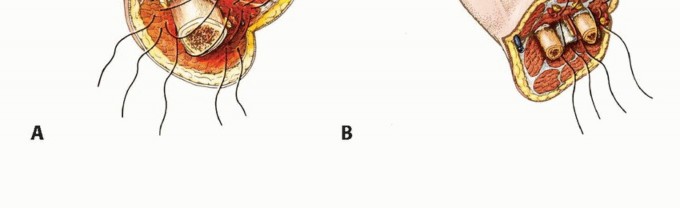
The muscular architecture of the brachium is compartmentalized into anterior and posterior fascial envelopes. The anterior compartment houses the biceps brachii, brachialis, and coracobrachialis, which are the primary flexors of the elbow, innervated predominantly by the musculocutaneous nerve. The posterior compartment contains the triceps brachii, the primary extensor of the elbow, innervated by the radial nerve. During a transhumeral amputation, these antagonistic muscle groups must be stabilized via myodesis to the distal humerus to prevent retraction, preserve resting muscle tension, and provide a stable soft-tissue envelope over the transected bone.
From a biomechanical perspective, preserving humeral length is paramount. A longer residual humerus provides a superior lever arm for shoulder flexion and abduction, which are critical for positioning a prosthetic terminal device in space. However, a minimum of 4 to 5 centimeters of clearance proximal to the anatomical elbow joint line is required to accommodate the internal mechanisms of a prosthetic elbow joint. If the amputation is performed too distally (e.g., at the epicondyles), the prosthetic elbow will project asymmetrically compared to the contralateral sound limb, leading to poor cosmesis and altered biomechanics.
Below Elbow Transradial Amputation Anatomy
Transradial amputations occur through the radius and ulna. The forearm is highly complex, containing twenty distinct muscles divided into volar (flexor/pronator) and dorsal (extensor/supinator) compartments. The preservation of the proximal radioulnar joint and the surrounding musculature is the defining biomechanical advantage of a transradial amputation over a transhumeral amputation.
The volar compartment comprises the superficial flexors (pronator teres, flexor carpi radialis, palmaris longus, flexor carpi ulnaris), the intermediate flexor (flexor digitorum superficialis), and the deep flexors (flexor digitorum profundus, flexor pollicis longus, pronator quadratus). The dorsal compartment includes the mobile wad (brachioradialis, extensor carpi radialis longus and brevis) and the superficial and deep extensors of the digits and carpus.
In transradial amputations, preserving native pronation and supination is a primary objective. The degree of preserved rotation is directly proportional to the length of the residual radius and ulna. A long transradial amputation (distal third) may preserve up to 100 degrees of rotation, whereas a short transradial amputation (proximal third) may preserve less than 20 degrees due to the loss of the pronator teres and pronator quadratus insertions, as well as the mechanical impingement of the soft tissues. The minimum functional length for a transradial amputation is generally considered to be 5 centimeters distal to the biceps tuberosity; amputations proximal to this level function biomechanically as transhumeral amputations because the residual forearm is too short to maintain a prosthetic socket during elbow flexion.

Neurovascular Considerations
Meticulous management of the neurovascular bundles is critical in both above-elbow and below-elbow amputations to prevent life-threatening hemorrhage and debilitating neuromas.
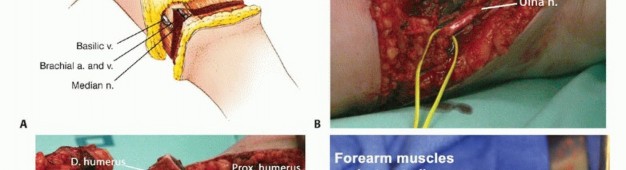
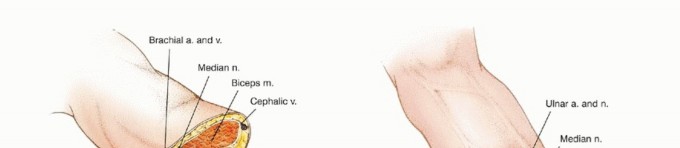
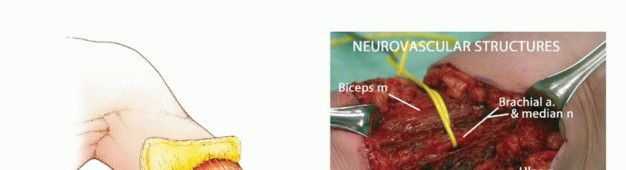
In the arm, the brachial artery travels with the median nerve in the medial bicipital groove. The ulnar nerve runs medially, piercing the medial intermuscular septum to enter the posterior compartment. The radial nerve spirals around the posterior humerus in the spiral groove, making it highly vulnerable during mid-shaft humeral transections. The musculocutaneous nerve lies between the biceps and brachialis.
In the forearm, the radial artery runs deep to the brachioradialis, while the ulnar artery runs deep to the flexor carpi ulnaris. The median nerve travels between the two heads of the pronator teres and deep to the flexor digitorum superficialis. The ulnar nerve travels with the ulnar artery. The radial nerve bifurcates into the superficial sensory branch (deep to brachioradialis) and the deep motor branch (posterior interosseous nerve, piercing the supinator). All major peripheral nerves must be identified, gently distracted, cleanly transected, and allowed to retract deep into healthy muscle beds, or preferentially managed with advanced nerve interface techniques to prevent symptomatic terminal neuromas.
Indications and Contraindications
The decision to amputate an upper extremity is rarely straightforward and requires a nuanced understanding of trauma severity, oncologic principles, and the systemic physiological status of the patient. Unlike the lower extremity, where scoring systems like the Mangled Extremity Severity Score (MESS) provide some predictive value, upper extremity scoring systems are notoriously unreliable for predicting functional outcomes following limb salvage versus amputation. The upper extremity's tolerance for ischemia and its robust collateral circulation often allow for salvage in situations that would mandate amputation in the lower limb.

Traumatic Indications
In the acute trauma setting, absolute indications for upper extremity amputation include a completely avascular limb with warm ischemia time exceeding 6 to 8 hours, particularly when massive muscle necrosis (myonecrosis) has already occurred. Attempting to revascularize a limb with established myonecrosis risks fatal reperfusion injury, characterized by myoglobinuric renal failure, hyperkalemia, and systemic inflammatory response syndrome (SIRS).
Severe crush injuries with segmental bone loss, complete avulsion of the brachial plexus, and massive soft tissue destruction also represent strong relative indications. While microvascular free flaps and nerve transfers can salvage many mangled upper extremities, the surgeon must weigh the likelihood of achieving a functional, sensate limb against the morbidity of multiple reconstructive surgeries, prolonged hospitalization, and the psychological toll on the patient. A stiff, insensate, and painful salvaged arm is functionally inferior to a well-constructed amputation with a properly fitted prosthesis.
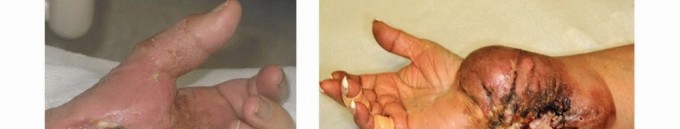
Oncologic Indications
In orthopaedic oncology, the primary goal is the eradication of the tumor with negative surgical margins. Amputation is indicated when a primary bone sarcoma (e.g., osteosarcoma, Ewing sarcoma, chondrosarcoma) or a high-grade soft tissue sarcoma extensively involves the major neurovascular bundles (e.g., brachial artery and plexus) such that limb-sparing resection would leave an avascular or completely paralyzed limb. Furthermore, amputation is indicated for massive tumors that have destroyed the adjacent joint or soft tissue compartments to an extent that functional reconstruction is impossible, or in cases of local recurrence following previous limb-salvage surgery and radiation therapy.
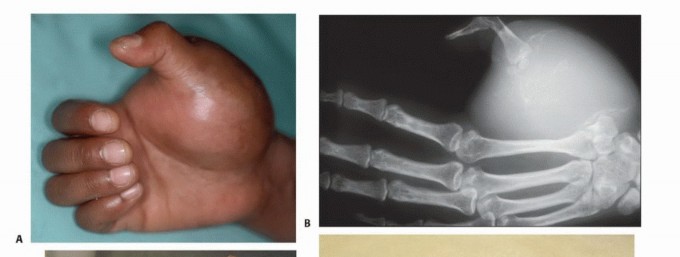
Infectious and Vascular Indications
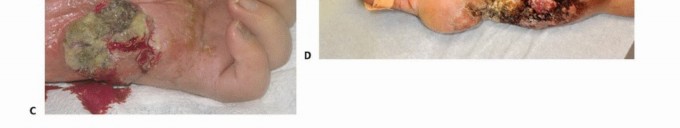
Fulminant infections, such as necrotizing fasciitis or clostridial myonecrosis (gas gangrene), require emergent, life-saving amputation if the infection cannot be controlled with radical debridement alone. Chronic, refractory osteomyelitis that has destroyed the structural integrity of the bone and overlying soft tissue envelope may also necessitate amputation, particularly in immunocompromised or medically frail patients. Irreversible ischemia secondary to arterial thrombosis, embolic events, or severe vasospastic disorders (e.g., advanced Buerger's disease) that are not amenable to vascular bypass or endovascular intervention serve as definitive indications.
Operative vs Non Operative Indications Summary
| Clinical Scenario | Indication for Amputation (Operative) | Indication for Limb Salvage / Non-Operative |
|---|---|---|
| Trauma / Mangled Limb | Warm ischemia > 6-8 hours with established myonecrosis; Complete anatomic disruption of major nerves (brachial plexus avulsion) with massive tissue loss. | Clean, sharp amputations amenable to replantation; Ischemia < 6 hours; Intact or reconstructable neurovascular bundles with adequate soft tissue coverage. |
| Oncology (Sarcoma) | Tumor encasement of critical neurovascular structures precluding negative margins; Massive compartmental destruction; Recurrence post-radiation. | Tumor resectable with wide margins while preserving critical neurovascular bundles; Amenable to endoprosthetic or allograft reconstruction. |
| Infection | Uncontrolled necrotizing fasciitis; Gas gangrene threatening systemic survival; Refractory chronic osteomyelitis with non-functional limb. | Localized soft tissue infection; Osteomyelitis amenable to radical debridement, local antibiotics, and free tissue transfer. |
| Vascular Disease | Irreversible ischemia with gangrene; Failed revascularization; Systemic toxicity from ischemic tissue. | Reversible ischemia; Amenable to embolectomy, bypass grafting, or endovascular stenting. |
Pre Operative Planning and Patient Positioning
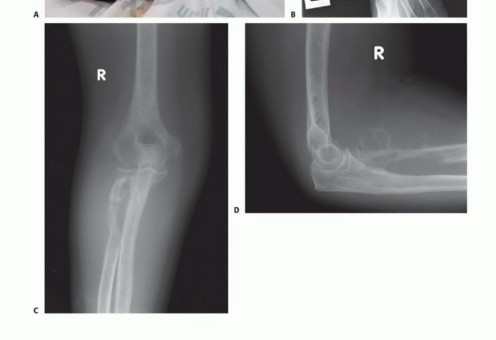
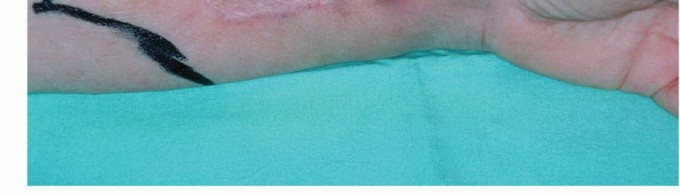
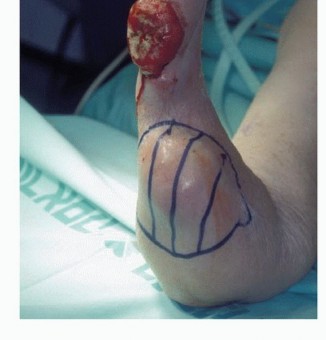
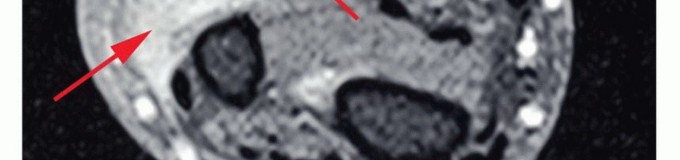
Thorough preoperative planning is essential to optimize the functional outcome of the amputation. This begins with a detailed assessment of the zone of injury or extent of disease. In trauma, plain radiographs and computed tomography (CT) angiography are critical for defining the level of viable bone and assessing the integrity of the vascular tree. In oncology, magnetic resonance imaging (MRI) of the entire involved compartment is mandatory to determine the necessary proximal extent of the bone resection and soft tissue margins.
Level Selection and Flap Design
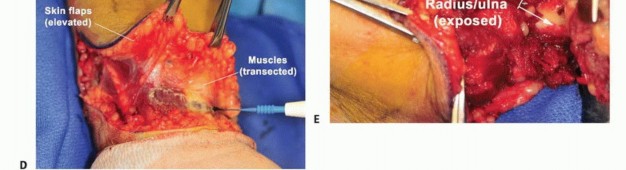
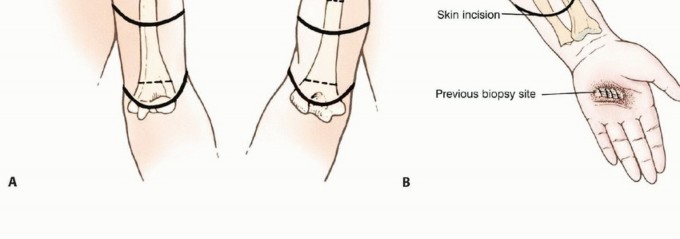
The fundamental principle of amputation surgery is the preservation of maximal functional length while ensuring adequate, healthy soft tissue coverage. The surgeon must anticipate the design of the skin flaps preoperatively.
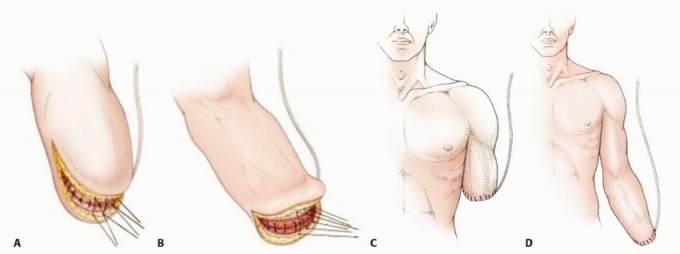
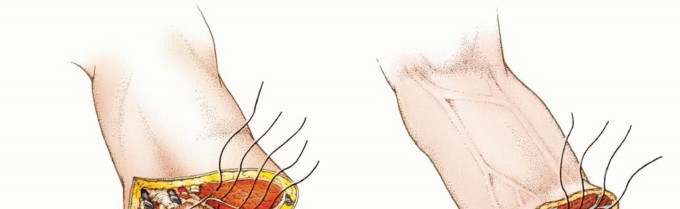
For transhumeral amputations, equal anterior and posterior fish-mouth flaps are traditionally utilized. However, atypical flaps based on available viable tissue (e.g., a long medial or lateral flap) are frequently required in trauma and oncology. The bone should be cut at a level that allows the soft tissue flaps to close without tension over the distal stump.
For transradial amputations, equal volar and
Clinical & Radiographic Imaging
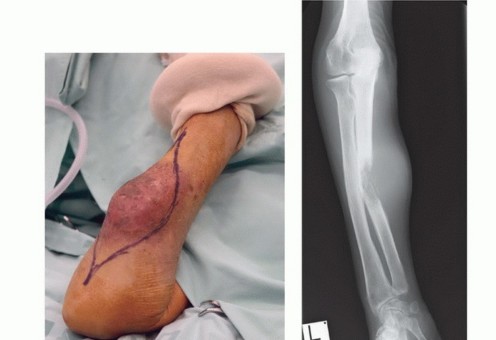
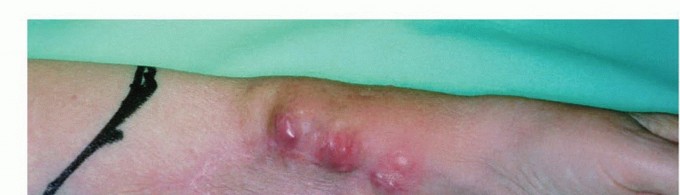
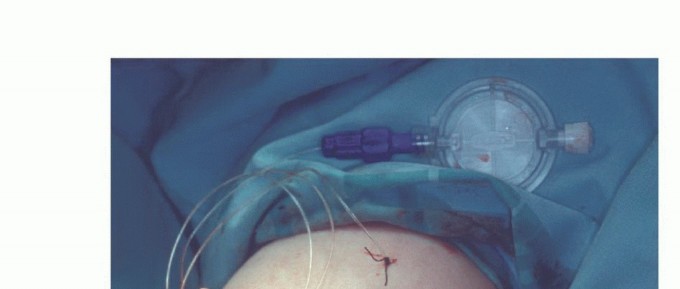
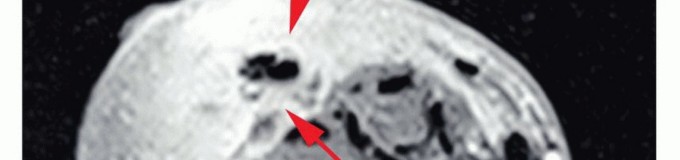




You Might Also Like