Surgical Masterclass: Advanced Management of Metastatic Bone Disease
Key Takeaway
This masterclass guides fellows through the comprehensive surgical management of metastatic bone disease. We cover critical preoperative planning, precise intraoperative techniques for pathological fractures and impending lesions, including meticulous tumor debulking and robust fixation with cement. Emphasis is placed on neurovascular preservation, optimal implant selection, and managing complex reconstructions, ensuring immediate stability and restoring function for cancer patients.
Introduction and Epidemiology
Metastatic bone disease (MBD) represents a profound oncologic and orthopedic challenge, necessitating a multidisciplinary approach to optimize patient outcomes. The human skeleton is the third most common site of metastatic disease dissemination, superseded only by the pulmonary and hepatic systems. The vast majority of skeletal metastases—approximately 80%—originate from carcinomas of the prostate, breast, lung, kidney, and thyroid (frequently remembered by the mnemonic "Lead Kettle" or PB KTL). The continuous, rapid advancement in systemic targeted therapies, immune checkpoint inhibitors, and advanced chemoradiation protocols has significantly prolonged the overall survival of oncology patients. Consequently, the incidence of patients presenting with MBD and surviving long enough to experience skeletal-related events (SREs) has grown exponentially. While the exact incidence of bone metastases remains difficult to quantify precisely due to subclinical presentations, epidemiological data suggest that in the United States alone, over 350,000 patients die annually with skeletal metastases originating from primary carcinomas.
Metastatic bone disease is a primary driver of morbidity, contributing directly to the deterioration of the quality of life in patients with disseminated cancer. These patients frequently require orthopedic surgical intervention for the management of impending or completed pathologic fractures, or for the alleviation of intractable pain associated with locally progressive, mechanically destabilizing lesions. SREs are clinically defined by the occurrence of pathologic fractures, the requirement for radiation or surgery to bone, spinal cord compression, and tumor-induced hypercalcemia. These events are associated with a severe loss of function, profound pain, and significant impairment of mobility, leading to a cascade of systemic deconditioning.
It is critical to recognize that surgical interventions in this highly comorbid population are predominantly palliative, with the exception of solitary metastases in specific histologies (e.g., renal cell carcinoma, thyroid carcinoma) where wide resection may offer a survival advantage. The overarching surgical goals are to achieve local tumor control, establish immediate and durable structural stability of the compromised skeletal site, and restore normal ambulatory or upper extremity function as rapidly as possible. Failure to achieve immediate and durable stability usually necessitates revision surgery, which carries an unacceptably high morbidity and mortality rate in an already physiologically compromised patient. Historical literature demonstrates failure rates of surgeries performed for MBD reaching as high as 40%. These catastrophic failures typically result from poor initial mechanical fixation, underestimation of the biomechanical forces, improper implant selection, or relentless progression of the osteolytic disease within the operative field.

Surgical Anatomy and Biomechanics
A profound understanding of the biomechanical alterations in bone infiltrated by metastatic disease is the fundamental prerequisite for meticulous surgical planning. Normal diaphyseal bone relies on its tubular cortical structure to resist bending, axial, and torsional forces. The presence of an osteolytic lesion creates a significant stress riser, fundamentally altering the load-bearing capacity of the bone. According to basic biomechanical principles, the stress concentrated at the periphery of a cortical defect is magnified by a factor of three compared to intact bone (an open-section defect). Furthermore, when a lesion destroys more than 50% of the cortical circumference, the torsional strength of the bone is reduced by over 70%, drastically increasing the risk of a spiral pathologic fracture during normal, low-demand physiologic loading.
The healing potential of a pathologic fracture is fundamentally different from that of a traumatic fracture. The underlying neoplastic process actively impedes the normal biologic cascade of fracture healing. Tumor cells secrete factors such as parathyroid hormone-related protein (PTHrP), interleukins (IL-1, IL-6), and tumor necrosis factor (TNF), which stimulate osteoclastogenesis via the RANK/RANKL/OPG pathway. This leads to continuous, aggressive bone resorption that vastly outpaces osteoblastic bone formation. Consequently, the prognosis for the osseous union of a pathologic fracture is poor and heavily dependent on the histology of the primary tumor and its response to adjuvant therapies.
Lesions associated with metastatic adenocarcinomas of the breast and prostate, multiple myeloma, and lymphoma exhibit a higher propensity for union following stabilization, provided they are responsive to systemic therapy and local radiation. Conversely, metastases from the lung, kidney, and gastrointestinal tract rarely achieve functional osseous union. Even when healing does occur, it requires a prolonged period, and the resulting callus is mechanically inferior and disorganized. Therefore, reduction and immobilization techniques that rely on biologic healing (such as standard non-locking plates without cement, or cast immobilization) are entirely inappropriate for MBD. The surgical construct must be designed to bear the full physiologic load of the extremity for the remainder of the patient's life, functioning as a permanent load-bearing, rather than load-sharing, device.
Indications and Contraindications
The decision to proceed with operative intervention requires a meticulous, multidisciplinary assessment of the patient's systemic disease burden, life expectancy, and the mechanical integrity of the affected bone. The primary indications for surgical intervention include an existing completed pathologic fracture, an impending pathologic fracture, intractable pain associated with locally progressive disease unresponsive to non-operative modalities (radiation, bisphosphonates, denosumab), and a solitary bone metastasis in highly selected tumor types where wide en bloc resection may confer a distinct survival benefit.
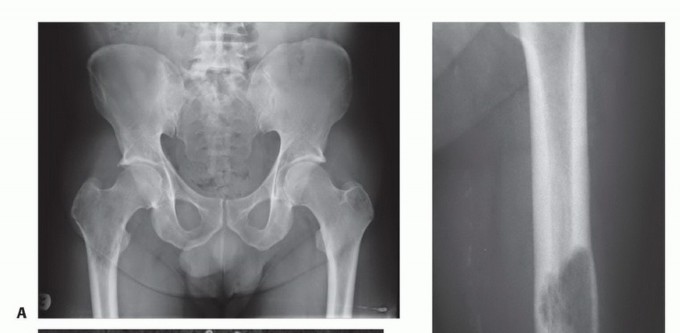
The assessment of an impending pathologic fracture relies heavily on Mirels' criteria, a validated scoring system that evaluates four parameters: the site of the lesion (upper extremity, lower extremity, peritrochanteric), the nature of the lesion (blastic, mixed, lytic), the size of the lesion (<1/3, 1/3-2/3, >2/3 of cortical diameter), and the degree of pain (mild, moderate, functional). A Mirels score of 9 or greater carries a fracture risk of >33% and is a strong indication for prophylactic operative stabilization. Prophylactic fixation is technically less demanding, associated with significantly lower intraoperative blood loss, shorter hospital stays, and results in faster rehabilitation compared to the stabilization of a completed pathologic fracture.

Patient selection is paramount and requires honest prognostication. It is generally accepted that surgical intervention is appropriate for patients with an expected survival of greater than six to twelve weeks. Patients with a life expectancy of less than six weeks are unlikely to benefit from the physiological insult of major orthopedic surgery, as they may spend their remaining time recovering from the procedure, confined to a hospital bed, rather than enjoying an improved quality of life. In such cases, non-operative management, including palliative radiation therapy, pharmacological pain control, and hospice care, is indicated.
Operative vs Non Operative Indications
| Clinical Scenario | Recommended Management | Rationale |
|---|---|---|
| Completed Pathologic Long Bone Fracture | Operative Fixation (IMN or EPR) | Immediate pain relief, restoration of mobility, facilitates nursing care, prevents further soft tissue compromise. |
| Impending Fracture (Mirels Score $\ge$ 9) | Prophylactic Operative Fixation | Prevents catastrophic mechanical failure, lower morbidity, lower blood loss than post-fracture surgery. |
| Solitary Renal Cell or Thyroid Metastasis | Wide Resection and Reconstruction | Potential for curative intent or significantly prolonged disease-free survival; these tumors are highly radioresistant. |
| Life Expectancy Less Than 6 Weeks | Non-Operative Management | Surgical morbidity and recovery time outweigh potential functional benefits. Focus on palliative comfort. |
| Diffuse Spinal Metastases without Instability | Radiation and Antiresorptives | Systemic control and pain palliation without surgical risk. SINS score utilized to assess mechanical instability. |
| Intractable Pain Unresponsive to Radiation | Palliative Operative Stabilization | Mechanical stabilization eliminates micro-motion at the tumor-bone interface, providing dramatic pain relief. |
Pre Operative Planning and Patient Positioning
Thorough, exhaustive preoperative planning is the absolute cornerstone of preventing construct failure in metastatic bone disease. The evaluation must begin with high-quality, full-length orthogonal plain radiographs of the entire affected bone, including the joints above and below. It is a catastrophic error to stabilize a proximal lesion with a short construct, only to have the bone fracture through a distal, unrecognized skip lesion weeks later. The surgical implant must span the entire bone to protect it from future metastatic deposits.
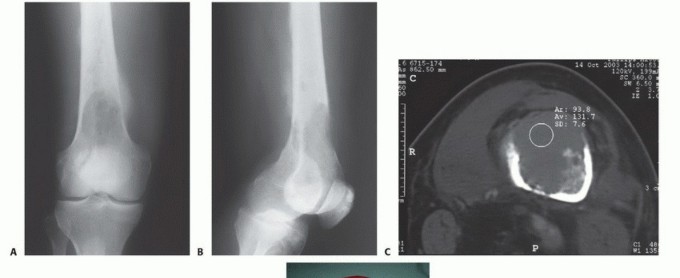
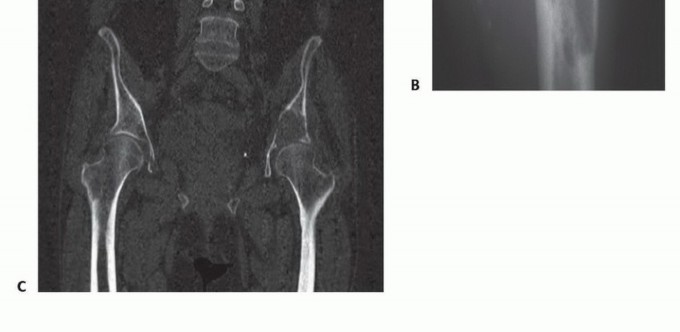
Advanced cross-sectional imaging is mandatory. Computed tomography (CT) provides precise delineation of cortical destruction, aiding in the assessment of fracture risk, the planning of screw trajectories, and the identification of optimal osteotomy sites. Magnetic resonance imaging (MRI) of the entire anatomic compartment is essential for evaluating the intraosseous extent of the tumor, skip metastases within the marrow cavity, and any extraosseous soft tissue extension that might dictate the surgical approach, neurovascular involvement, or the necessity for a more radical resection.
In cases where the primary tumor is unknown (metastatic carcinoma of unknown primary), a biopsy is absolutely required prior to definitive fixation. Proceeding with prophylactic stabilization without a tissue diagnosis can compromise future curative resections if the lesion is ultimately identified as a primary bone sarcoma (e.g., osteosarcoma, Ewing sarcoma) rather than a metastasis. The biopsy tract must be meticulously planned, typically utilizing a longitudinal incision in line with the definitive surgical approach, ensuring that the entire tract can be excised during the definitive surgery.
Preoperative embolization is a critical adjunct, particularly for highly vascular tumors such as metastatic renal cell carcinoma, thyroid carcinoma, and occasionally multiple myeloma. Embolization performed 24 to 48 hours prior to surgery significantly reduces intraoperative hemorrhage, improves surgical field visualization, and decreases perioperative morbidity. Failure to embolize these specific metastases can result in massive, life-threatening intraoperative blood loss, coagulopathy, and hemodynamic collapse.
Patient positioning depends entirely on the chosen surgical approach, the planned implant, and the patient's pulmonary status. For femoral diaphyseal lesions planned for intramedullary nailing, the patient may be positioned supine on a fracture table or in the lateral decubitus position on a radiolucent flat table. The lateral position often facilitates easier access to the piriformis fossa or greater trochanter entry points, particularly in obese patients. Furthermore, the flat table allows for simultaneous access to the anterior and posterior aspects of the extremity if open tumor debulking, cementation, or conversion to an open approach is required.
Detailed Surgical Approach and Technique
The surgical technique for metastatic bone disease must prioritize immediate, rigid, and durable stability. Because biologic healing is unreliable and often non-existent, reconstruction techniques that rely on autologous bone grafts, allografts, or allograft-prosthetic composites are generally inappropriate in the metastatic setting. Instead, load-sharing devices such as full-length intramedullary nails, or load-bearing devices such as modular endoprostheses and cement-augmented plates, are the mainstays of operative treatment.
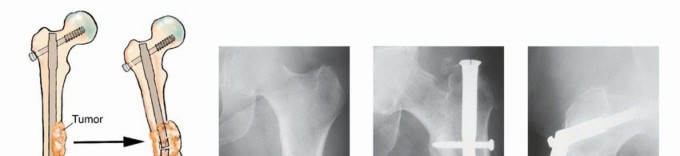
Intramedullary Nailing
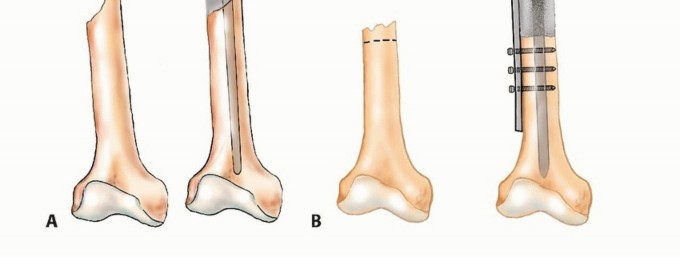
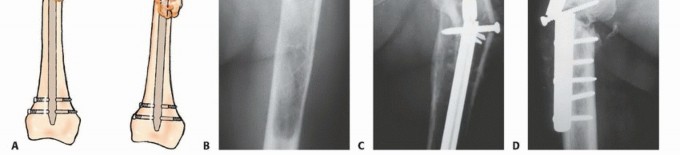
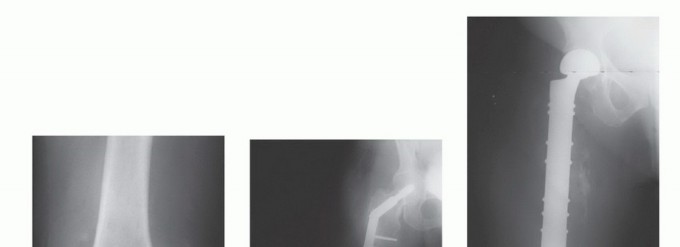
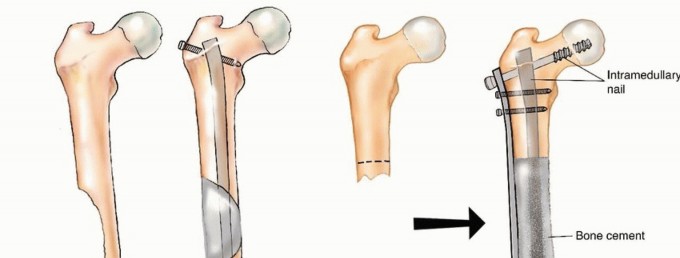
Intramedullary (IM) nailing is the workhorse for diaphyseal metastases of the femur, tibia, and humerus. The procedure is biomechanically superior to plating as the implant is located at the mechanical axis of the bone, significantly reducing bending moments. The surgical approach involves establishing a proximal entry portal. For the femur, a cephalomedullary reconstruction nail is strongly preferred to protect the femoral neck from future metastatic involvement, even if the current lesion is isolated entirely to the diaphysis.
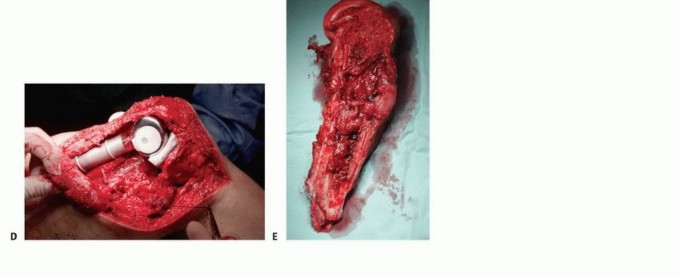
The canal is sequentially reamed to accommodate the largest possible nail diameter, maximizing the area moment of inertia. Reamings should routinely be sent for pathologic evaluation if the diagnosis is in question or to confirm the primary histology. The nail must span the entire length of the bone, and static interlocking screws must be placed both proximally and distally to control rotation and axial length. In cases of massive cortical destruction where the nail lacks adequate cortical contact (the "stovepipe" canal), open curettage of the tumor and augmentation with polymethylmethacrylate (PMMA) bone cement around the nail is necessary. This provides immediate rotational stability, reinforces the thinned cortices, and prevents implant fatigue failure.
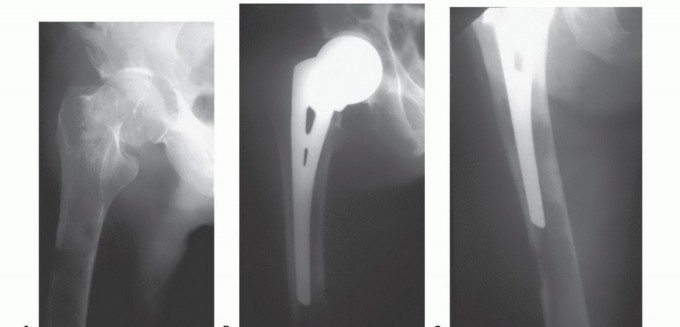
Plate Osteosynthesis with Cement Augmentation
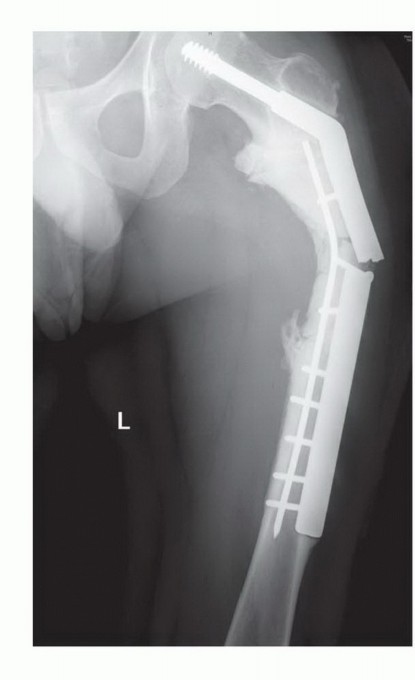
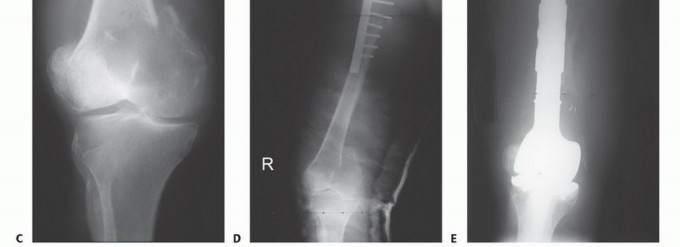
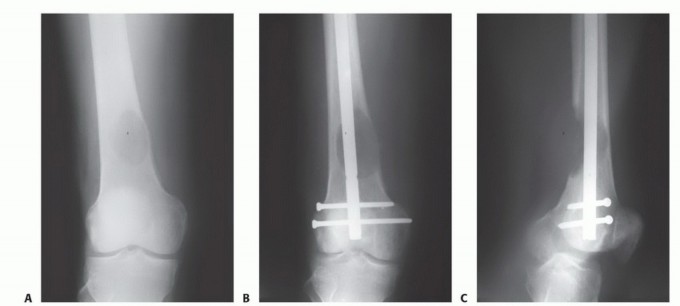
When lesions are located in the metaphyseal regions or metaphyseal-diaphyseal junctions where intramedullary nails cannot achieve adequate purchase or multi-planar stability, plate osteosynthesis combined with tumor curettage and cement augmentation is indicated. The surgical approach involves direct exposure of the tumor cavity. A thorough intralesional curettage is performed, often utilizing a high-speed burr, to remove all gross tumor tissue and expose healthy, bleeding cortical bone.
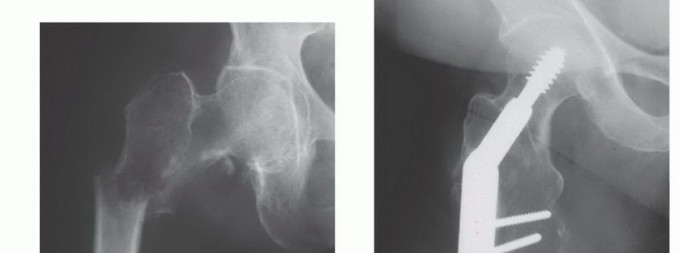
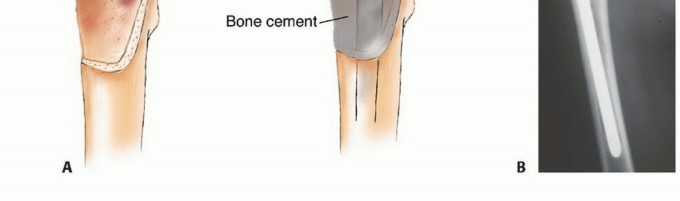
The resulting cavitary defect is then filled with PMMA. The cement provides immediate compressive strength and acts as an excellent void filler. A broad, heavy-duty locking plate is then applied across the defect, spanning the entire bone if possible. Screws are placed through the plate and directly into the curing cement. The exothermic reaction of the cement provides local adjuvant tumor necrosis (thermal cytotoxicity), while the composite construct of plate, cement, and bone provides immediate structural integrity, a biomechanical concept frequently referred to as the "rebar in concrete" technique.
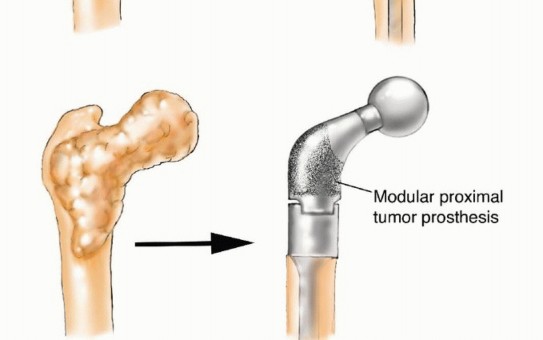
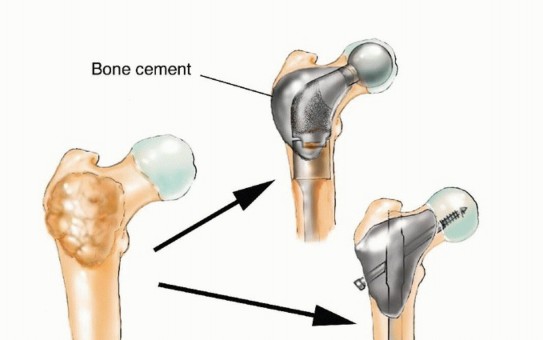
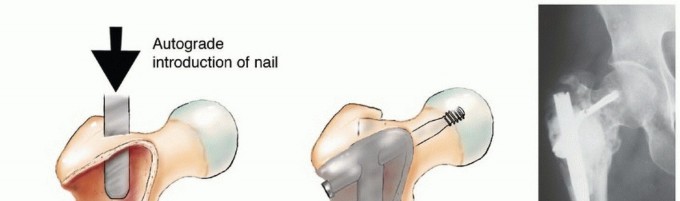
Endoprosthetic Reconstruction
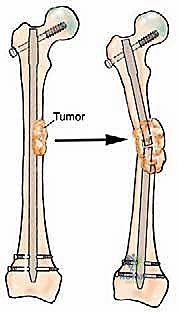
For extensive periarticular destruction, massive segmental bone loss, or failed previous osteosynthesis—particularly in the proximal femur, proximal humerus, or distal femur—internal fixation is doomed to fail. In these scenarios, segmental resection of the involved bone and reconstruction with a modular tumor endoprosthesis (EPR) is required.
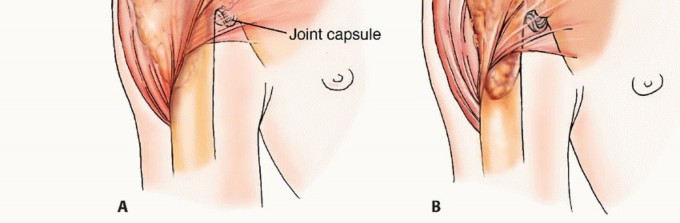
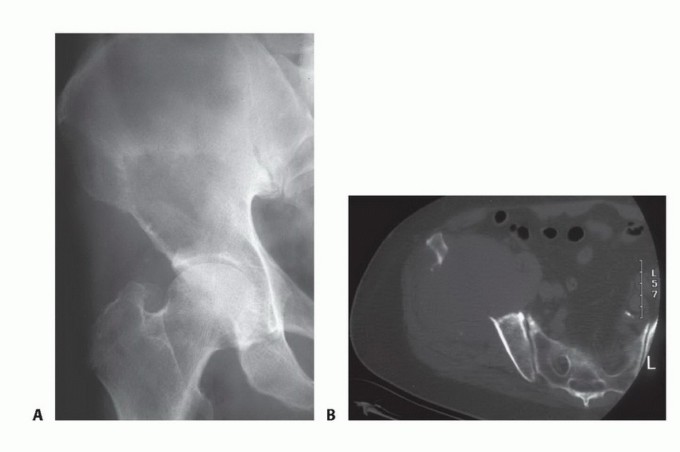
For a proximal femoral metastasis with severe neck, head, or intertrochanteric involvement (e.g., Mirels $\ge$ 9 in the peritrochanteric region), a cemented bipolar hemiarthroplasty (for limited disease) or a modular proximal femoral replacement (for extensive disease) is the procedure of choice. The surgical approach is typically a standard posterolateral or direct lateral approach to the hip. The femoral neck or proximal diaphysis is osteotomized distal to the tumor extent, ensuring a margin of healthy bone.
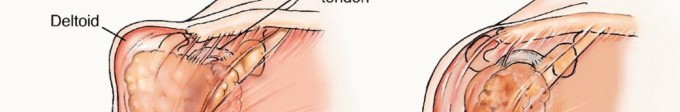
The diaphyseal canal is prepared, and a long-stemmed prosthesis is cemented in place. Cemented fixation is mandatory in the metastatic setting to allow for immediate, unrestricted full weight-bearing and to bypass the reliance on biologic osteointegration, which will be profoundly inhibited by postoperative radiation therapy and systemic chemotherapy. Soft tissue reconstruction, particularly the meticulous reattachment of the abductor mechanism to the prosthesis (often utilizing Trevira tubes or heavy non-absorbable sutures through specialized trochanteric claw plates), is critical for postoperative function, ambulation, and the prevention of dislocation.
Complications and Management
Surgical intervention for metastatic bone disease carries a high complication profile due to the systemic frailty of the patient population, the compromised local tissue envelope, the hypercoagulable state of malignancy, and the aggressive nature of the underlying disease. Anticipation, preoperative optimization, and aggressive postoperative management of these complications are essential for optimizing outcomes.
Construct failure is the most devastating orthopedic complication. It typically occurs due to underestimating the mechanical demand, utilizing an implant that is too short (failing to span the entire bone), or failing to augment massive cortical defects with PMMA cement. When a construct fails, salvage usually requires revision to a massive modular endoprosthesis, which carries significant surgical morbidity, massive blood loss, and a high risk of perioperative mortality.
Infection is another major concern, exacerbated by cancer cachexia, malnutrition, prior local radiation, and systemic immunosuppressive chemotherapy. Deep surgical site infections in the presence of massive metallic hardware and bone cement are incredibly difficult to eradicate and often require suppressive antibiotic therapy for the remainder of the patient's life. Venous thromboembolism (VTE) is highly prevalent given the hypercoagulable state of malignancy combined with the endothelial injury and stasis associated with major orthopedic surgery.
Common Complications and Salvage Strategies
| Complication | Estimated Incidence | Prevention and Salvage Strategy |
|---|---|---|
| Hardware Failure / Nonunion | 10% - 15% | Span entire bone, use cement augmentation, avoid load-sharing in massive defects. Salvage: Revision to modular endoprosthesis. |
| Intraoperative Hemorrhage | 5% - 10% | Preoperative embolization (RCC, Thyroid, Myeloma). Salvage: Rapid volume resuscitation, damage control packing, massive transfusion protocol. |
| Deep Surgical Site Infection | 5% - 12% | Meticulous soft tissue handling, optimize nutrition/albumin, delay XRT. Salvage: Serial debridement, suppressive antibiotics, hardware retention if stable. |
| Venous Thromboembolism | 10% - 20% | Aggressive chemical (LMWH/DOAC) and mechanical prophylaxis. Salvage: Therapeutic anticoagulation, IVC filter if anticoagulation is strictly contraindicated. |
| Tumor Progression at Surgical Site | 15% - 20% | Adjuvant postoperative external beam radiation therapy (EBRT). Salvage: Repeat resection, revision reconstruction, escalation of systemic therapy. |
Post Operative Rehabilitation Protocols
The primary objective of the postoperative rehabilitation protocol is the immediate restoration of patient mobility and independence. Because the surgical constructs are explicitly designed to be load-bearing and entirely independent of biologic healing, patients should be permitted immediate full weight-bearing on the affected lower extremity, or immediate functional use of the affected upper extremity, as tolerated by pain. Restricted weight-bearing protocols defeat the palliative purpose of the surgery and contribute directly to rapid systemic deconditioning, deep vein thrombosis, decubitus ulcers, and pulmonary complications (atelectasis, pneumonia).
Coordination with the radiation oncology team is a critical component of the postoperative pathway. Postoperative external beam radiation therapy (EBRT) is the standard of care to achieve local tumor control, prevent disease progression around the implant, and palliate residual bone pain. However, radiation must be carefully timed. Administering radiation too early can severely compromise surgical wound healing, leading to wound dehiscence, flap necrosis, and deep periprosthetic infection. It is generally recommended to delay radiation therapy for two to three weeks postoperatively to ensure adequate soft tissue healing and suture removal, while not delaying so long as to allow aggressive local tumor recurrence.
Summary of Key Literature and Guidelines
The surgical management of metastatic bone disease is guided by several foundational orthopedic studies and evolving oncologic guidelines. The seminal work by Mirels (1989) established the predictive scoring system for impending pathologic fractures, shifting the orthopedic paradigm towards prophylactic stabilization. This approach has been repeatedly proven to yield superior functional outcomes, shorter operative times, and lower perioperative morbidity compared to the fixation of completed fractures.
Gainor and Buchert's analysis of pathologic fracture healing remains highly relevant, emphasizing that reliance on biologic union is flawed, particularly in non-osteoblastic tumors (lung, renal, GI). Their findings cemented the absolute
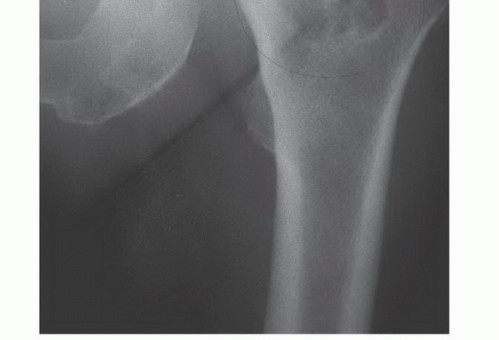
Clinical & Radiographic Imaging
You Might Also Like