Surgical Management of Metastatic Bone Disease: Pelvic Lesions
Introduction and Epidemiology
Overview of Pelvic Metastatic Disease
Metastatic tumors of the pelvis represent a profound and complex challenge in the realm of orthopedic oncology. These lesions frequently precipitate intractable mechanical pain, significant structural instability, and a catastrophic loss of ambulatory function and weight-bearing capacity. Due to the substantial volume of the pelvic cavity, the elastic compliance of the resident visceral organs, and the robust surrounding musculofascial envelope, metastatic deposits at this site typically achieve considerable dimensions before manifesting clinically.
Epidemiologically, the pelvis constitutes the second most common site for skeletal metastases, superseded only by the axial spine. Primary carcinomas of the breast, prostate, lung, kidney, and thyroid—frequently remembered by the mnemonic "BLT with a Kosher Pickle"—account for the overwhelming majority of these osteolytic and osteoblastic lesions. The incidence of skeletal metastasis is rising, paradoxically driven by advancements in targeted systemic therapies and immunotherapies that significantly prolong overall patient survival. Consequently, orthopedic surgeons are increasingly tasked with providing durable reconstructions that outlast the patient's anticipated lifespan.
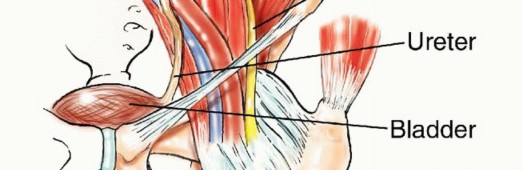
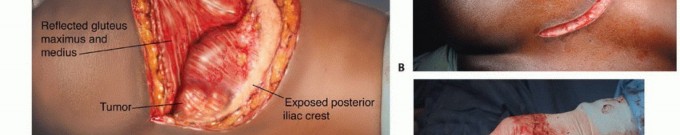
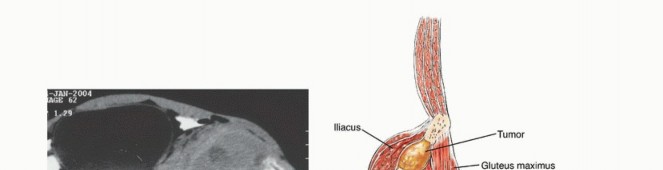
The anatomic location of the metastasis within the pelvic ring dictates its impact on global pelvic stability. Lesions isolated to the anterior ilium or the pubic rami exert minimal deleterious effects on weight-bearing kinematics. Conversely, tumors infiltrating the posterior ilium or the sacroiliac articulation pose an imminent threat to lumbosacral integrity and axial load transfer. Furthermore, neoplastic involvement of the periacetabular corridor profoundly compromises hip joint kinematics and the structural capacity of the lower extremity to support physiological loads. Both primary bone sarcomas and metastatic carcinomas frequently present with insidious, considerable extension into the adjacent extraosseous soft tissues.
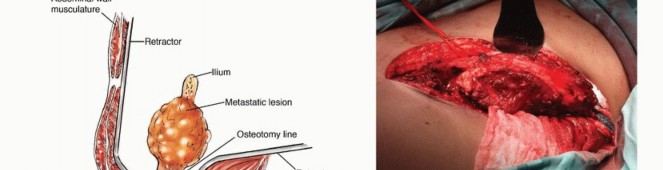
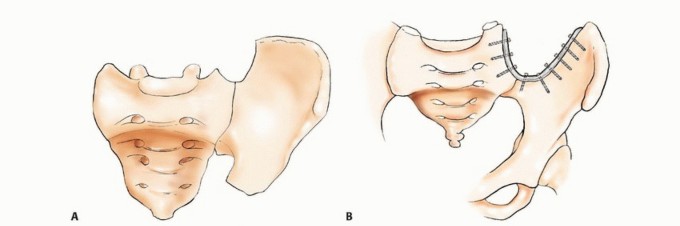
Given the inherent sensitivity of many metastatic histologies to adjuvant radiation therapy and targeted systemic treatments, the surgical management paradigm diverges significantly from that of primary bone sarcomas. En bloc resection with wide, negative margins is rarely mandated for metastatic disease, except in cases of solitary metastasis from favorable histologies (e.g., renal cell carcinoma) where a curative intent is pursued. Instead, orthopedic oncologists employ a "utilitarian approach."
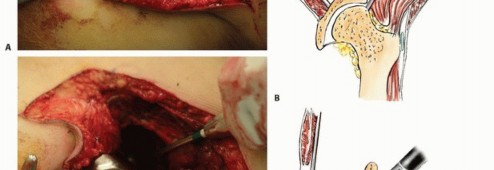
This strategy prioritizes aggressive intralesional curettage, immediate structural reconstruction, and rigid internal stabilization. Any microscopic neoplastic residua are subsequently addressed via postoperative fractionated radiotherapy. The intricate three-dimensional anatomy of the pelvic girdle necessitates exhaustive preoperative imaging, meticulous surgical planning, and precise execution of the chosen operative exposure.
Pathophysiology and Tumor Biology
The pronounced predilection for metastatic deposition within the pelvic ring is biologically driven by the extensive volume of highly vascularized red marrow within the innominate bone, coupled with the sluggish, valveless venous flow characteristic of the Batson venous plexus. Neoplastic cells disseminate hematogenously, arresting within the tortuous capillary beds of the medullary cavity.
Upon extravasation, these cells initiate a complex, cytokine-mediated cascade (frequently involving the RANK/RANKL pathway) that results in profound osteoclast-mediated bone resorption (characteristic of osteolytic lesions such as renal cell or thyroid carcinoma) or aberrant, disorganized osteoblast stimulation (characteristic of osteoblastic lesions such as prostate carcinoma). Breast carcinoma frequently presents with a mixed osteolytic and osteoblastic phenotype.
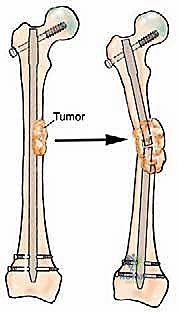
The resulting structural degradation fundamentally alters the physiological stress distribution across the pelvic ring. This biomechanical compromise precipitates microfractures, macroscopic cortical failure, and subsequent severe mechanical pain. It is imperative for the evaluating surgeon to clinically differentiate mechanical pain (exacerbated by weight-bearing and movement) from biological pain (constant, nocturnal, inflammatory). Mechanical pain dictates an impending or actual structural failure that necessitates surgical stabilization, whereas purely biological pain may be successfully palliated with radiation therapy and osteoclast inhibitors (e.g., zoledronic acid or denosumab).
Surgical Anatomy and Biomechanics
Osteology and Regional Zones
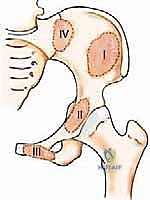
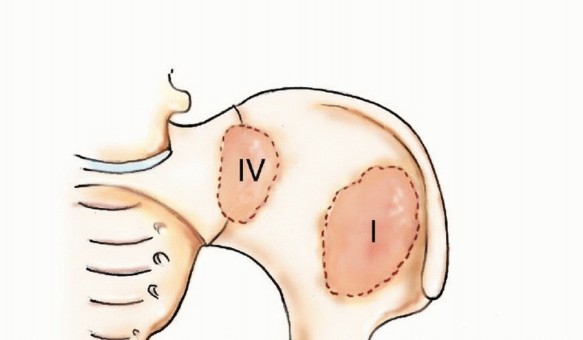
A profound mastery of pelvic osteology is the cornerstone of safe and effective oncologic reconstruction. The pelvic ring is conceptually divided into distinct anatomic zones, most notably codified by the Enneking and Dunham classification system. This classification divides the hemipelvis into four distinct regions, which dictates both the functional consequence of the metastasis and the requisite surgical approach.
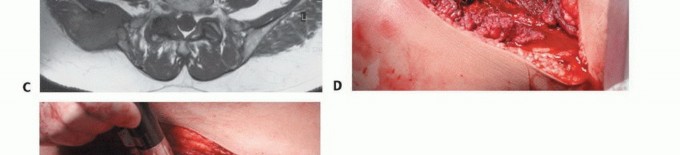
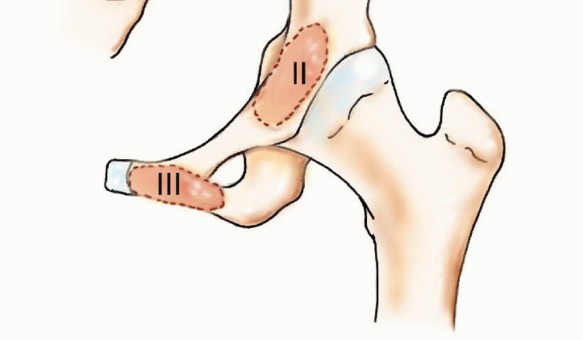
Zone I encompasses the ilium, extending from the iliac crest inferiorly to the superior border of the acetabulum. Lesions confined to Zone I, particularly those anterior to the sciatic buttress, often do not compromise the primary weight-bearing axis. However, extensive destruction of the posterior ilium threatens the integrity of the sacroiliac articulation.
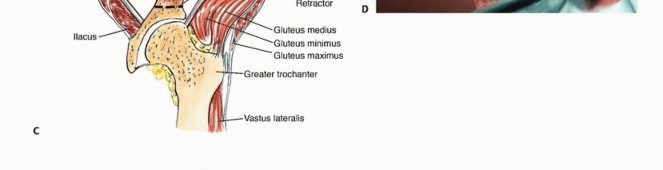
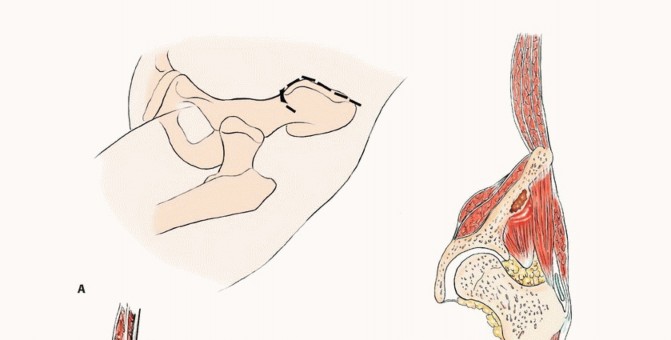
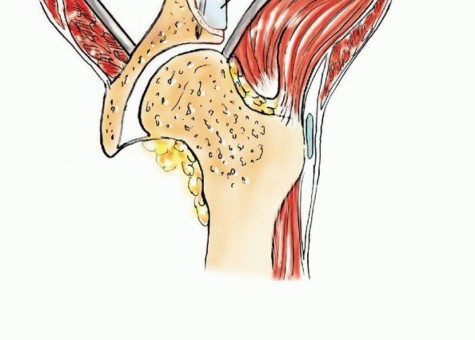
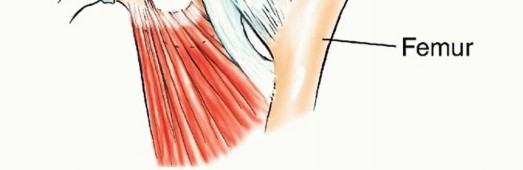
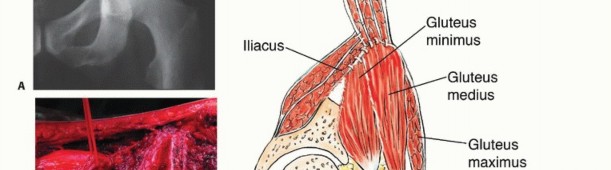
Zone II represents the periacetabular region. This is the most critical zone biomechanically, as it serves as the keystone for load transfer between the axial skeleton and the lower extremity. The acetabulum is supported by the anterior column (extending from the iliac crest to the pubic symphysis) and the posterior column (extending from the sciatic notch to the ischial tuberosity).
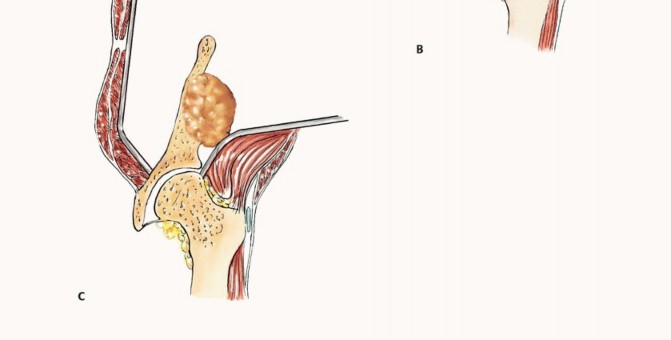
Zone III comprises the pubis and ischium. Metastatic destruction in this anterior ring segment is frequently asymptomatic or presents with localized, manageable pain. Surgical intervention in isolated Zone III lesions is exceedingly rare, as the anterior ring contributes only approximately 15-20% to the overall stability of the pelvic ring.
Zone IV involves the sacrum. Metastatic disease here threatens the neural elements of the cauda equina and the sacral nerve roots, and can precipitate spinopelvic dissociation if the alar regions are bilaterally compromised. Reconstructions in this zone require complex lumbopelvic fixation constructs.
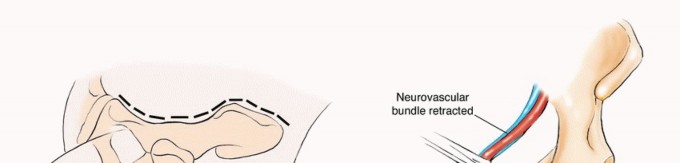
Biomechanics of Axial Load Transfer
The biomechanical imperative of the pelvis is to transmit the superimposed weight of the torso from the axial spine to the bilateral lower extremities. This load transfer follows a highly predictable trajectory: from the L5 vertebral body across the lumbosacral disc to the S1 endplate, laterally across the sacroiliac joints into the dense cortical bone of the sciatic buttress, and inferiorly into the supra-acetabular corridor and the dome of the acetabulum.
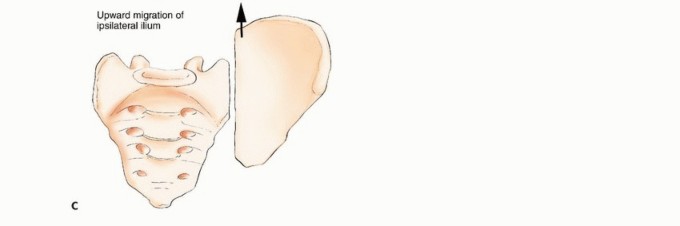
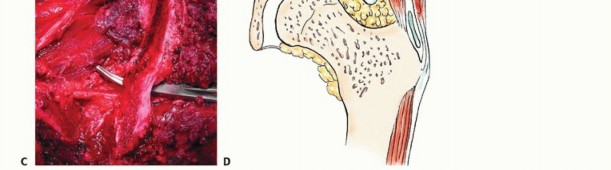
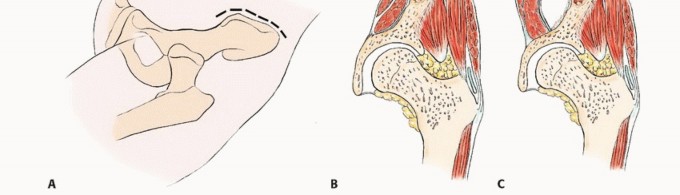
The trabecular architecture of the innominate bone aligns precisely with these principal stress trajectories. The primary compressive trabeculae extend from the SI joint to the superior acetabular dome. A secondary tensile trabecular system arches across the iliac wing. Metastatic osteolysis disrupts these trabecular networks. When the cross-sectional area of the supra-acetabular corridor is reduced by more than 50%, the risk of catastrophic mechanical failure and acetabular protrusio increases exponentially.
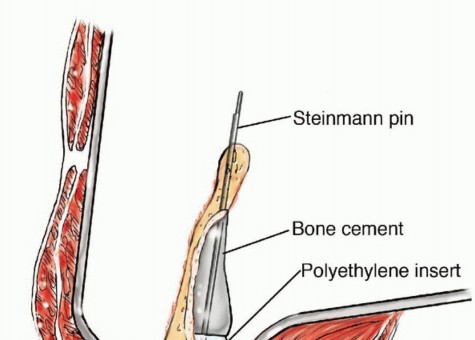
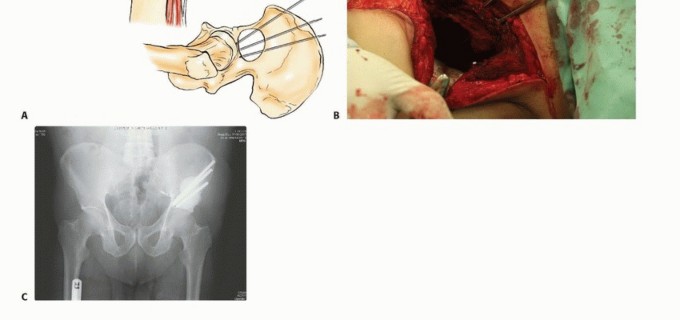
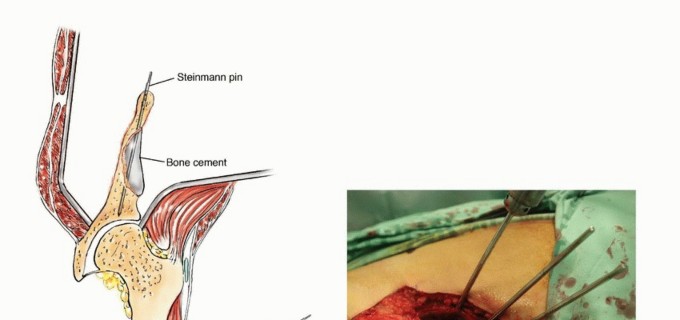
In the utilitarian approach, the surgeon must functionally bypass the area of osteolysis. If the acetabular dome is compromised, load must be transferred from the intact femoral shaft to the remaining viable bone of the superior ilium or the sacroiliac complex. This frequently necessitates the use of massive structural implants, such as anti-protrusio cages, custom triflange components, or massive polymethylmethacrylate (PMMA) constructs reinforced with threaded structural pins (the classic Harrington reconstruction).
Indications and Contraindications
Clinical Evaluation and Decision Making
The decision to proceed with major pelvic reconstruction in the setting of metastatic disease requires a delicate calculus balancing the patient's overall prognosis, systemic tumor burden, medical comorbidities, and the severity of local mechanical compromise. The primary indication for surgery is severe, intractable mechanical pain that precludes ambulation and is refractory to non-operative measures, including radiation therapy and pharmacological management.
Impending or actual pathologic fractures of the periacetabular region (Zone II) are absolute indications for surgical intervention in patients with an anticipated survival exceeding 6 to 12 weeks. The Harrington criteria for impending periacetabular failure remain a benchmark in orthopedic oncology. These criteria include:
1. Subchondral bone destruction exceeding 50% of the acetabular dome.
2. Axial migration (protrusio) of the femoral head exceeding 5 mm.
3. Medial wall destruction.
4. Intractable pain following a full course of fractionated external beam radiation therapy.
Contraindications to surgery are equally critical to recognize. Patients in extremis, those with profound coagulopathies, severe malnutrition (albumin < 2.5 g/dL), or an anticipated survival of less than 4-6 weeks are generally poor candidates for massive pelvic reconstruction. In these scenarios, the morbidity of the surgical insult outweighs the potential functional benefit, and palliative care with optimization of analgesia is the preferred pathway.
Prognostic Scoring Systems
Accurate prognostication is essential to tailor the magnitude of the surgical intervention to the patient's life expectancy. Several scoring systems assist the orthopedic oncologist in this determination. The Katagiri scoring system evaluates the primary tumor histology, the presence of visceral metastases, laboratory parameters, and the patient's performance status to predict survival.
Similarly, the modified Tokuhashi score, originally designed for spinal metastases, is frequently extrapolated to pelvic disease. Patients with highly favorable histologies (e.g., breast, prostate, thyroid) and isolated skeletal disease may survive for years, justifying complex, durable reconstructions such as custom 3D-printed implants. Conversely, patients with aggressive histologies (e.g., lung carcinoma, hepatocellular carcinoma) and widespread visceral disease require rapid, palliative stabilization techniques that permit immediate weight-bearing with minimal surgical morbidity.
| Indication Category | Operative Management | Non-Operative Management |
|---|---|---|
| Pain Characteristics | Intractable mechanical pain, pain with weight-bearing | Biological pain, nocturnal pain, responsive to XRT/analgesics |
| Structural Integrity | Impending/actual fracture, >50% cortical loss, acetabular protrusio | Intact weight-bearing axis, isolated Zone III (anterior ring) lesions |
| Patient Prognosis | Anticipated survival > 3 months, Katagiri favorable | Anticipated survival < 1 month, multiorgan failure, poor performance status |
| Neurologic Status | Progressive sacral nerve root or lumbosacral plexus compression | Intact neurology or stable, non-progressive deficits |
| Histology | Radioresistant tumors (e.g., Renal Cell, Thyroid, Melanoma) | Highly radiosensitive tumors (e.g., Myeloma, Lymphoma) without fracture |
Pre Operative Planning and Patient Positioning
Advanced Imaging Protocols
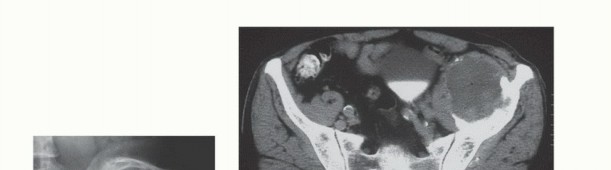
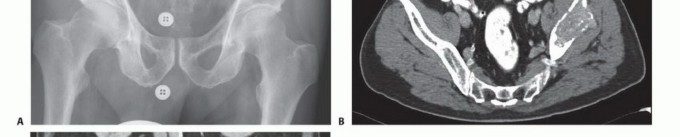
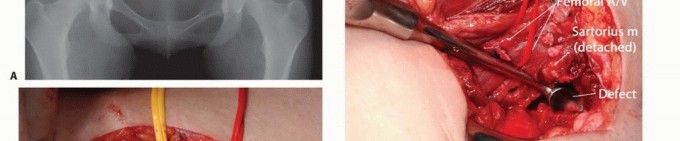
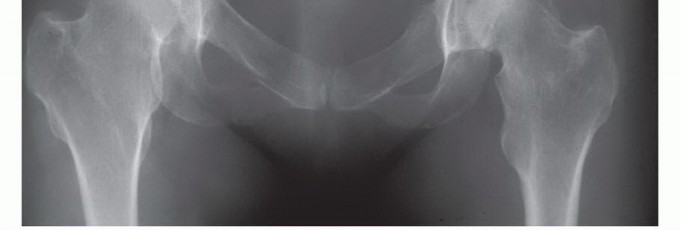
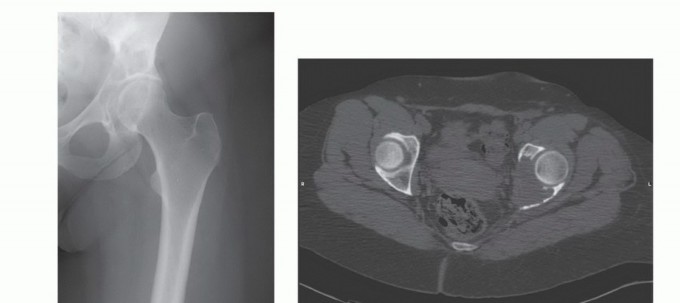
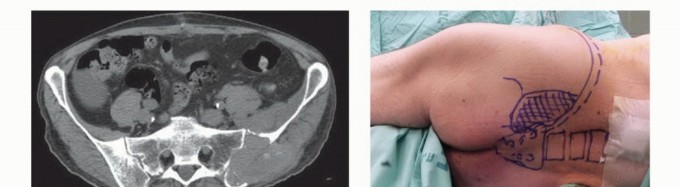
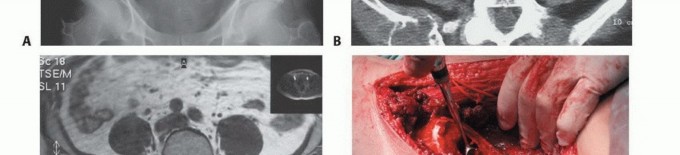
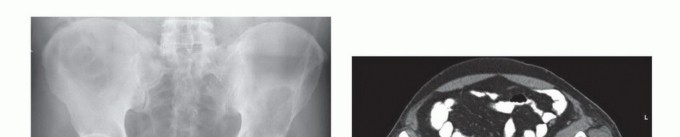
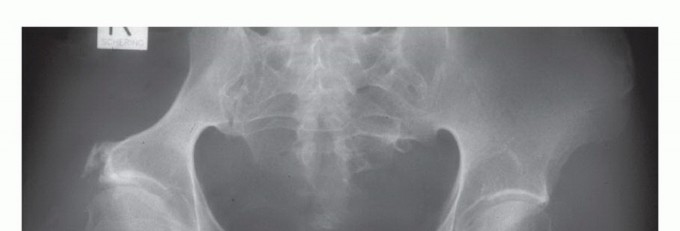
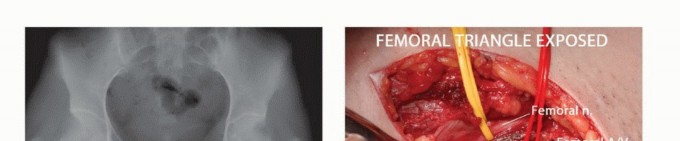
The three-dimensional complexity of the pelvis mandates an exhaustive preoperative imaging workup. Standard orthogonal radiographs (Anteroposterior, Judet obturator oblique, and Judet iliac oblique views) provide a baseline assessment of bone stock and gross deformity. However, cross-sectional imaging is non-negotiable.
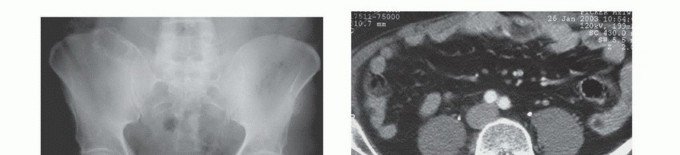
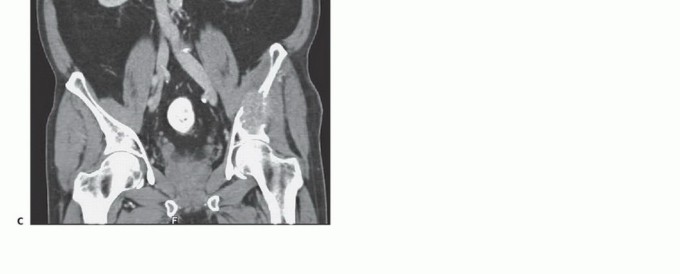
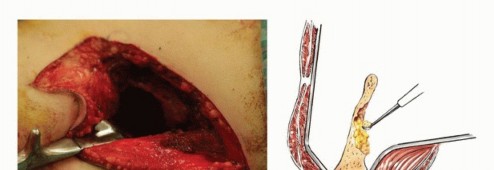
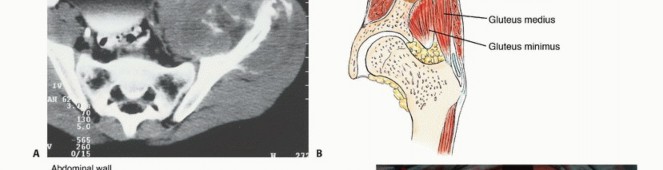
High-resolution Computed Tomography (CT) with 1mm slices and 3D reconstructions is the gold standard for evaluating cortical destruction, assessing remaining viable bone stock for implant fixation, and planning screw trajectories. The CT scan precisely delineates the extent of osteolysis within the supra-acetabular corridor and the sciatic buttress, which are critical zones for hardware purchase.
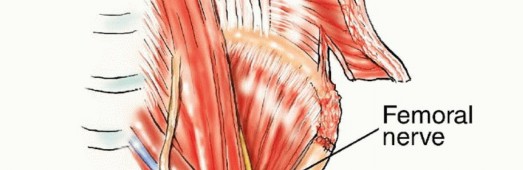
Magnetic Resonance Imaging (MRI) of the pelvis, utilizing T1, T2 fat-suppressed, and post-contrast sequences, is essential for evaluating extraosseous soft tissue extension. MRI provides unparalleled visualization of the tumor's relationship to critical neurovascular structures, including the external iliac vessels, the sciatic nerve, the lumbosacral plexus, and the obturator neurovascular bundle. Furthermore, MRI is highly sensitive for detecting skip metastases or multifocal marrow involvement that might alter the fixation strategy.
Angiography and Embolization
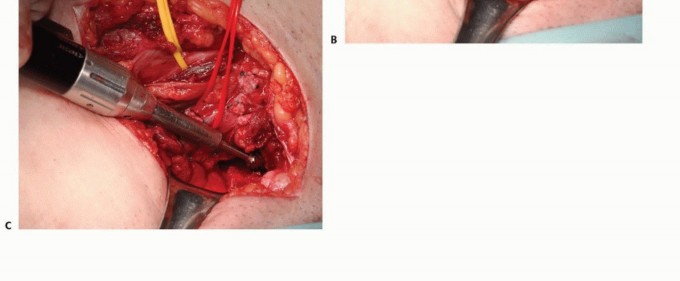
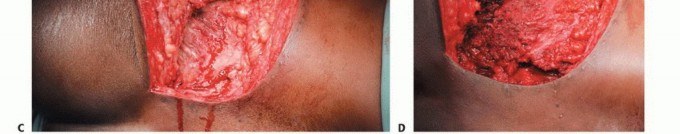
Intraoperative hemorrhage is a leading cause of morbidity and mortality in pelvic tumor surgery. Many metastatic histologies—particularly renal cell carcinoma, thyroid carcinoma, and certain breast carcinomas—are notoriously hypervascular.
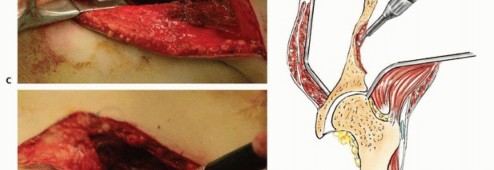
Preoperative selective arterial embolization is a critical adjunct for these lesions. Conducted by interventional radiology within 24 to 48 hours prior to the surgical procedure, embolization targets the specific feeding vessels of the tumor (frequently branches of the internal iliac artery, such as the superior gluteal, inferior gluteal, or obturator arteries). Embolization significantly reduces intraoperative blood loss, improves visualization of the surgical field, and facilitates a more thorough intralesional curettage.
Anesthesia and Patient Positioning
Pelvic reconstruction requires a coordinated multidisciplinary approach involving experienced orthopedic oncologists and specialized orthopedic anesthesiologists. Massive fluid shifts and rapid blood loss must be anticipated. Large-bore peripheral intravenous access, central venous lines, and invasive arterial blood pressure monitoring are standard. The availability of rapid infusion systems (e.g., Belmont rapid infuser) and massive transfusion protocols is mandatory. Cell salvage systems (cell saver) may be utilized; while historically controversial in oncology, modern leukodepletion filters effectively clear circulating tumor cells, making autologous blood transfusion safe and highly beneficial in these massive blood-loss cases.
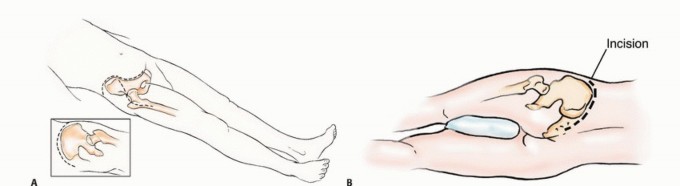
Patient positioning is dictated by the anatomic location of the lesion and the planned surgical approach.
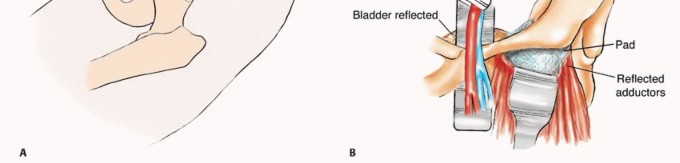
* Lateral Decubitus: The most common position for periacetabular (Zone II) reconstructions, allowing access via a Kocher-Langenbeck or modified Gibson approach. It provides excellent exposure of the posterior column and ilium.
* Supine: Utilized for anterior column (Zone III) or extensive ilioinguinal approaches. The ipsilateral bump is placed under the hemipelvis.
* Floppy Lateral / Modified Supine: Allows dynamic repositioning to access both the anterior and posterior aspects of the hemipelvis during complex, multi-zone reconstructions.
Detailed Surgical
Clinical & Radiographic Imaging




You Might Also Like