Median Nerve Decompression: Mastering Pronator and Anterior Interosseous Syndromes
Key Takeaway
This masterclass guides fellows through median nerve decompression for pronator and anterior interosseous syndromes. We cover comprehensive surgical anatomy, meticulous intraoperative techniques, and critical pearls to navigate this complex procedure. From initial incision to nerve release and closure, learn precise steps to optimize patient outcomes and manage potential complications, ensuring thorough decompression of all potential impingement sites.
Introduction and Epidemiology
Pronator syndrome and anterior interosseous syndrome represent two distinct, yet anatomically contiguous, compression neuropathies affecting the median nerve and its major motor branch, the anterior interosseous nerve (AIN), within the complex topography of the elbow and proximal forearm. While carpal tunnel syndrome (CTS) remains the most ubiquitous median nerve entrapment encountered in clinical practice, proximal compression is comparatively rare. Consequently, it frequently presents a profound diagnostic challenge, characterized by an insidious onset and clinical features that mimic or overlap with cervical radiculopathy (C6-C7), brachial plexopathy, and more distal entrapment neuropathies.
The true epidemiological incidence of proximal median nerve compression is notoriously difficult to ascertain. This ambiguity stems from the multiplicity of potential impinging anatomic structures, the transient nature of activity-related symptoms, and a high rate of clinical misdiagnosis. Pronator syndrome predominantly afflicts patients in their fourth to fifth decades of life, demonstrating a higher prevalence in individuals subjected to repetitive, forceful pronation and supination. This demographic typically encompasses manual laborers, assembly line workers, and athletes participating in racquet sports, throwing, or weightlifting. Conversely, anterior interosseous syndrome may manifest spontaneously or secondary to acute trauma. Its etiology remains a subject of intense academic debate, necessitating rigorous differentiation from neuralgic amyotrophy—specifically Parsonage-Turner syndrome—which presents with an acute, painful pre-paralytic phase followed by profound motor weakness.
Pathophysiologically, proximal median nerve compression in the forearm is frequently transient, precipitated by excessive physical exertion leading to localized muscular hypertrophy, acute fascial edema, or dynamic tethering. The mechanical compression initiates localized ischemia of the vasa nervorum, precipitating a breakdown of the blood-nerve barrier, impairment of axoplasmic transport, and subsequent endoneurial edema. If the mechanical insult remains unresolved, a cascade of chronic structural alterations ensues, including epineurial fibrosis, focal demyelination, and eventually Wallerian degeneration of the distal axons. Accurately mapping the precise site of compression is of paramount importance; surgical management mandates a comprehensive, sequential exploration and release of all potential anatomic bottlenecks to prevent persistent or recurrent symptomatology.
Surgical Anatomy and Biomechanics
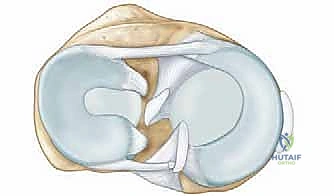
A profound, three-dimensional understanding of the regional anatomy of the antecubital fossa and proximal forearm is the unequivocal cornerstone of successful surgical decompression. The median nerve traverses a hostile anatomical landscape, navigating through rigid fascial bands, crossing vascular leashes, and intricate muscular arcades.
Median Nerve Course and Sites of Entrapment
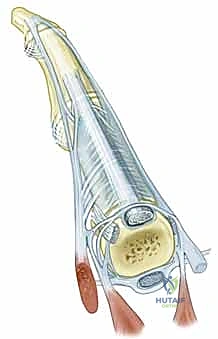
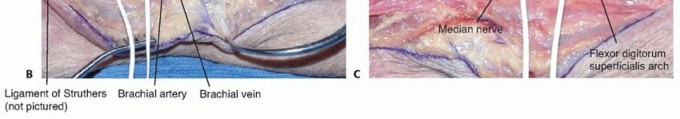
The median nerve descends through the distal arm occupying a position between the brachialis muscle and the medial intermuscular septum, with the brachial artery situated immediately lateral to the nerve. The first potential site of proximal entrapment is the ligament of Struthers. A supracondylar process—an atavistic bony spur arising from the anteromedial aspect of the distal humerus approximately five centimeters proximal to the medial epicondyle—is present in roughly 1% to 2% of the population. When present, a fibrous band (the ligament of Struthers) extends from this process to the medial epicondyle. The median nerve, and occasionally the brachial artery, can pass deep to this ligament, predisposing them to dynamic compression during elbow extension.
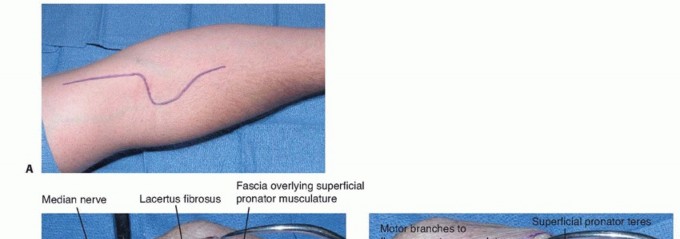
As the median nerve crosses the elbow joint, it passes deep to the lacertus fibrosus (bicipital aponeurosis). This robust fascial expansion extends from the distal biceps tendon to blend with the deep antebrachial fascia. Compression at this level is typically exacerbated during resisted elbow flexion combined with maximal forearm supination, which dynamically tightens the aponeurosis over the neurovascular bundle.
Distal to the lacertus fibrosus, the nerve enters the proximal forearm between the superficial (humeral) head and the deep (ulnar) head of the pronator teres. The superficial head originates from the medial epicondyle, while the deep head arises from the medial border of the coronoid process. In approximately 20% of individuals, the deep head is either entirely absent or exists as a diminutive, unyielding fibrous band. Paradoxically, this fibrotic variant significantly increases the risk of neural tethering. Muscular hypertrophy or intrinsic fibrotic changes within the pronator teres represent the most frequently cited etiologies for true pronator syndrome.
Motor branches supplying the palmaris longus, flexor carpi radialis, flexor digitorum superficialis (FDS), and flexor digitorum profundus (FDP) typically branch from the median nerve in an ulnar trajectory just proximal to or within the substance of the pronator teres. Progressing distally, the main trunk of the median nerve passes deep to the fibrous arcade of the flexor digitorum superficialis. This "sublimis bridge" is a dense fascial arch connecting the humeroulnar and radial origins of the FDS and serves as a primary, rigid site of compression in both pronator and anterior interosseous syndromes.
The Anterior Interosseous Nerve
Deep to the pronator teres, the anterior interosseous nerve branches radially from the main trunk of the median nerve. Surgeons must remain vigilant regarding proximal or distal branching variations of the AIN. Both the parent median nerve and the AIN pass deep to the sublimis bridge. Subsequently, the AIN descends distally along the volar aspect of the interosseous membrane, providing critical motor innervation to the flexor pollicis longus (FPL), the radial half of the flexor digitorum profundus (supplying the index and long fingers), and ultimately terminating in the pronator quadratus.
Numerous ancillary structures have been implicated in isolated AIN compression. These include an accessory bicipital aponeurosis and various anomalous muscle bellies. The most frequently encountered anomalous muscle is the accessory head of the flexor pollicis longus, eponymously known as the Gantzer muscle, present in up to 50% of anatomical specimens. Additionally, crossing vascular leashes—such as the radial recurrent vessels or a persistent median artery penetrating the median nerve trunk—can induce localized, pulsatile compression.
Cutaneous Innervation and Anatomical Variants
The operating surgeon must be acutely cognizant of the superficial cutaneous nerves traversing the antecubital and proximal forearm regions. The medial antebrachial cutaneous (MABC) nerve and the lateral antebrachial cutaneous (LABC) nerve lie within the subcutaneous tissues directly overlying the standard surgical approach. Iatrogenic injury to these nerves can precipitate profound numbness, dysesthesias, and the formation of highly symptomatic, recalcitrant neuromas that often eclipse the original compressive pathology in clinical severity.
Anomalous neural interconnections must also be anticipated, the most common being the Martin-Gruber anastomosis. Present in approximately 15% of the population, this neural crossover involves fascicular branches coursing from either the median nerve or the AIN to the ulnar nerve within the proximal forearm. Unrecognized proximal median nerve compression in the presence of a Martin-Gruber anastomosis can yield atypical clinical phenotypes, including paradoxical intrinsic hand muscle weakness traditionally attributed to isolated ulnar nerve pathology.
Indications and Contraindications
The decision algorithm for surgical decompression relies heavily upon a meticulous clinical examination, objective diagnostic adjuncts, and the systematic exhaustion of non-operative modalities. Pronator syndrome typically presents with an insidious, aching pain in the volar proximal forearm, coupled with paresthesias in the median nerve distribution of the hand. Crucially, unlike carpal tunnel syndrome, pronator syndrome often involves hypoesthesia over the thenar eminence; this occurs due to the involvement of the palmar cutaneous branch of the median nerve, which arises proximal to the transverse carpal ligament and is thus spared in isolated CTS. Provocative testing, including resisted pronation (implicating the pronator teres), resisted elbow flexion in supination (implicating the lacertus fibrosus), and resisted middle finger proximal interphalangeal joint flexion (implicating the FDS arcade), are essential diagnostic maneuvers.
Anterior interosseous syndrome, by contrast, presents as a pure motor neuropathy. It is classically demonstrated by the patient's inability to form a precise "OK" sign (Kiloh-Nevin sign), resulting instead in a flattened pinch posture due to profound weakness of the FPL and the FDP to the index finger.

Electrodiagnostic studies (EMG/NCS) are often equivocal or entirely normal in dynamic pronator syndrome, limiting their utility. However, they are highly specific and indispensable in confirming anterior interosseous syndrome, quantifying the degree of axonal loss, and definitively ruling out cervical radiculopathy.
| Clinical Scenario | Operative Indications | Non-Operative Indications |
|---|---|---|
| Pronator Syndrome | Refractory symptoms after 3 to 6 months of structured conservative management; Presence of a space-occupying lesion (e.g., lipoma, ganglion cyst); Progressive motor weakness. | Mild to moderate intermittent symptoms; Absence of motor deficits; Symptom duration less than 3 months; Equivocal provocative testing. |
| Anterior Interosseous Syndrome | Lack of clinical or electromyographic improvement after 12 weeks of serial observation; Penetrating trauma or acute organized hematoma; Clear mechanical compression visualized on MRI/US. | Acute onset preceded by severe shoulder/arm pain suggestive of Parsonage-Turner syndrome; Early presentation (<12 weeks) without structural lesions. |
| Space Occupying Lesions | Confirmed lipoma, ganglion, or soft-tissue tumor causing direct mechanical compression and neurologic deficit. | Small, asymptomatic lesions discovered incidentally without evidence of neural compromise. |
Absolute contraindications to surgery include active local infection, profound medical comorbidities precluding regional or general anesthesia, and a confirmed diagnosis of Parsonage-Turner syndrome. Parsonage-Turner syndrome (idiopathic brachial neuritis) is characterized by acute, severe shoulder or arm pain followed by patchy, often profound weakness, which can include isolated AIN palsy. Surgical decompression in the setting of Parsonage-Turner syndrome is strictly contraindicated; it provides zero therapeutic benefit, exposes the patient to unwarranted surgical morbidity, and delays appropriate medical management.
Pre Operative Planning and Patient Positioning
Preoperative planning necessitates a comprehensive review of all imaging modalities. While plain radiographs are highly specific for identifying a supracondylar process, magnetic resonance imaging (MRI) or high-resolution diagnostic ultrasonography is invaluable for detecting subtle soft tissue anomalies, space-occupying lesions, or the presence of an aberrant Gantzer muscle impinging the AIN.
The patient is positioned supine on the operating table with the operative extremity extended onto a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the proximal brachium. The arm is prepped and draped in a standard sterile fashion, ensuring full, unimpeded mobility of the elbow and wrist joints to allow for dynamic intraoperative testing. The use of loupe magnification (typically 2.5x to 3.5x) combined with a fiber-optic headlight is mandatory to ensure meticulous anatomical dissection and the preservation of the delicate microvascular supply (vasa nervorum) to the nerve.
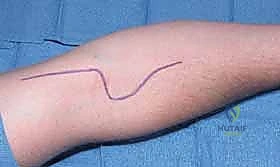
Prior to exsanguination and tourniquet inflation, key anatomical landmarks are mapped with a surgical marker. The medial epicondyle, the distal biceps tendon, and the palpable course of the brachial artery are identified. The planned incision is drawn to cross the antecubital flexion crease obliquely; a straight longitudinal incision across the flexion crease is strictly avoided, as it invariably leads to a restrictive, hypertrophic flexion contracture.
Detailed Surgical Approach and Technique
Surgical decompression of the proximal median nerve demands a systematic, stepwise approach to interrogate and release all five potential sites of compression: the ligament of Struthers, the lacertus fibrosus, the pronator teres, the flexor digitorum superficialis arcade, and any deep anomalous structures affecting the AIN.
Incision and Superficial Dissection
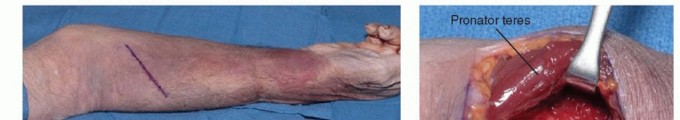
A lazy-S or curvilinear incision is utilized. The incision begins approximately five centimeters proximal to the elbow flexion crease on the medial aspect of the arm, courses obliquely across the antecubital fossa, and extends distally along the volar aspect of the forearm overlying the mobile wad.
The skin and subcutaneous tissues are carefully incised. The immediate surgical priority is the identification and protection of the superficial venous system and the regional cutaneous nerves. Branches of the medial antebrachial cutaneous nerve are typically encountered crossing the operative field in the proximal and medial aspects of the incision. The lateral antebrachial cutaneous nerve is identified laterally, emerging from beneath the lateral border of the biceps. These nerves must be mobilized with extreme care and gently retracted using vessel loops to prevent traction neuropraxia.
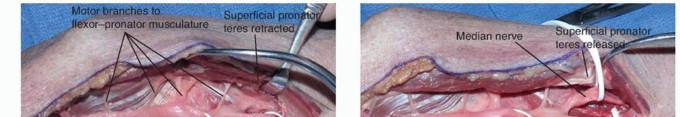
Stepwise Decompression of the Median Nerve
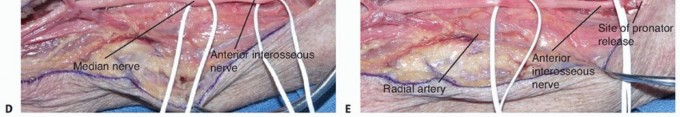
The deep antebrachial fascia is incised, and the lacertus fibrosus is identified as it extends from the biceps tendon ulnarly over the flexor-pronator mass. The lacertus fibrosus is sharply and completely divided. Deep to the lacertus, the brachial artery and the median nerve are identified. The median nerve is traced proximally into the distal third of the arm. If preoperative radiographs indicated the presence of a supracondylar process, the dissection is carried proximally to identify and excise the ligament of Struthers, along with a rongeur resection of the bony process itself, to prevent recurrent tethering.
Attention is subsequently directed distally to the pronator teres. The median nerve is tracked as it dives between the superficial and deep heads of the muscle. To adequately decompress the nerve without sacrificing pronation power, the superficial head is often managed via a fractional lengthening or a formal Z-lengthening (step-cut) tenotomy.
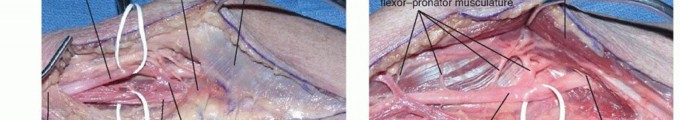
The step-cut technique allows for subsequent repair in an elongated position, preserving functional pronation strength while significantly expanding the anatomical space available for the nerve. The deep head of the pronator teres, if present and forming a tight fibrotic band over the nerve, is sharply excised. During this deep dissection, the surgeon must be meticulous in achieving hemostasis; the radial recurrent vessels frequently cross the surgical field in this vicinity and require careful bipolar coagulation or suture ligation.
Decompression of the Anterior Interosseous Nerve
Following the release of the pronator teres, the median nerve is traced further distally to the leading edge of the flexor digitorum superficialis. The fibrous arcade of the superficialis is a robust, unyielding structure that frequently indents the nerve. This arcade is completely divided under direct vision, taking immense care to protect the underlying delicate motor branches.
At this anatomical juncture, the anterior interosseous nerve is reliably identified branching from the radial aspect of the parent median nerve. The AIN is followed distally along the surface of the interosseous membrane. The surgeon must actively explore for a Gantzer muscle or an accessory bicipital aponeurosis. If a Gantzer muscle is found crossing over and compressing the AIN, its tendinous insertion or the muscle belly itself is released. The epineurium of both the main median nerve and the AIN is thoroughly inspected. If a severe hourglass constriction is noted, a careful, longitudinal epineurotomy may be performed under high magnification. However, internal neurolysis (interfascicular dissection) is strictly avoided, as it reliably induces devastating intraneural fibrosis and subsequent axonal death.
After confirming that the median nerve and the AIN are completely liberated from the distal arm to the mid-forearm, the tourniquet is deflated. Meticulous hemostasis is achieved using bipolar electrocautery to prevent postoperative hematoma. The step-cut of the pronator teres superficial head is loosely approximated with absorbable sutures. The subcutaneous layer is closed with interrupted inverted sutures, and the skin is reapproximated with a running subcuticular suture or interrupted non-absorbable monofilament.
Complications and Management
Surgical decompression in the proximal forearm is technically demanding. Complications, while infrequent in the hands of experienced peripheral nerve surgeons, can profoundly impact upper extremity function. Meticulous surgical technique and a thorough understanding of the regional anatomy are required to mitigate these risks.
| Complication | Estimated Incidence | Salvage Strategies and Management |
|---|---|---|
| Cutaneous Neuroma (MABC/LABC) | 2% to 5% | Prevention is paramount via careful superficial dissection. Established, painful neuromas require surgical excision and targeted muscle reinnervation (TMR) or burying the proximal stump deep into adjacent muscle bellies to prevent mechanical irritation. |
| Incomplete Decompression | 1% to 3% | Often results from a failure to recognize and release the deep head of the pronator teres or the distal extent of the FDS arcade. Requires revision surgery with extensive external neurolysis and confirmation of the release of all five potential compression sites. |
| Postoperative Hematoma | 1% to 2% | Minimized via tourniquet deflation and meticulous bipolar hemostasis prior to closure. Large, expanding hematomas require immediate operative evacuation to prevent secondary ischemic compression of the median nerve. |
| Pronation Weakness | Rare (<1%) | Usually transient secondary to the pronator teres step-cut. Managed with structured physical therapy. Permanent, debilitating weakness is exceptionally rare due to the compensatory action of the pronator quadratus. |
| Infection | <1% | Standard postoperative prophylactic antibiotics if indicated. Superficial infections are managed with oral antibiotics; deep space infections necessitate urgent surgical irrigation and debridement. |
Persistent pain or failure to improve neurologically postoperatively should prompt a critical re-evaluation of the initial diagnosis. Misdiagnosis of Parsonage-Turner syndrome or concurrent cervical radiculopathy (double crush syndrome) are the most common etiologies for perceived surgical failure.
Post Operative Rehabilitation Protocols
The immediate postoperative goal is to protect the surgical site, manage edema, and prevent restrictive perineural adhesions around the newly decompressed nerves. The patient is placed in a bulky soft dressing and a long-arm posterior splint with the elbow immobilized at 90 degrees of flexion and the forearm in neutral rotation.
This immobilization phase must be brief, typically lasting 7 to 10 days. Prolonged immobilization is strictly avoided as it promotes dense perineural scarring and debilitating joint stiffness. At the first postoperative clinic visit, the splint and sutures are removed.
The early range of motion phase begins immediately following splint removal. Patients are instructed by a specialized hand therapist to perform active and active-assisted elbow flexion and extension, as well as forearm pronation and supination. Specific nerve gliding exercises are introduced early to ensure the median nerve and anterior interosseous nerve mobilize freely within their newly expanded anatomical beds, preventing tethering to the healing pronator teres or FDS fascia.
By postoperative week four, the strengthening phase is initiated. Progressive resistance exercises focusing on grip strength, pronation power, and general upper extremity conditioning are systematically incorporated. Most patients can return to light duty or standard activities of daily living by week four. A full return to heavy manual labor or strenuous athletic activities is generally anticipated between eight and twelve weeks, contingent upon the complete resolution of motor deficits, the absence of pain during provocative maneuvers, and the restoration of symmetric grip strength.
Summary of Key Literature and Guidelines
The academic and surgical foundation for the management of proximal median nerve entrapments is built upon decades of rigorous anatomical studies and clinical case series. Spinner's seminal work in the 1970s definitively mapped the complex anatomical variations of the pronator teres, the flexor digitorum superficialis arcade, and the anomalous structures contributing to anterior interosseous syndrome. His detailed cadaveric dissections remain the undisputed gold standard for understanding the pathogenesis and surgical anatomy of these neuropathies.

Contemporary literature heavily emphasizes the necessity of differentiating mechanical compression from neuralgic amyotrophy. Tsairis et al. provided critical, foundational guidelines on the natural history of Parsonage-Turner syndrome, noting unequivocally that surgical intervention in these patients yields no benefit, exposes them to iatrogenic risk, and delays appropriate conservative and medical management.
Surgical outcomes for anterior interosseous syndrome with a clearly defined motor deficit and a corroborated mechanical etiology on MRI or ultrasound are generally excellent following comprehensive, stepwise decompression. Pronator syndrome outcomes, however, are inherently more variable. This variability is largely attributed to the subjective nature of the pain, the frequent absence of objective electrodiagnostic findings, and the potential for secondary gain in occupational settings. Therefore, current orthopedic guidelines strongly advocate for a minimum of three to six months of dedicated conservative therapy (including activity modification, splinting, and NSAIDs) prior to offering surgical decompression for pronator syndrome. Early operative intervention is strictly reserved for cases involving distinct space-occupying lesions or rapidly progressive, objectively measurable motor deterioration.
You Might Also Like