Mastering Kyphectomy in Myelomeningocele: An Intraoperative Guide
Key Takeaway
This intraoperative masterclass guides fellows through kyphectomy for myelomeningocele kyphosis. We cover comprehensive preoperative planning, meticulous surgical anatomy, and granular, real-time execution from incision to rod placement. Learn critical pearls, pitfalls, and post-operative care, ensuring optimal outcomes for these complex pediatric spinal deformities.
Introduction and Epidemiology
Kyphosis in the patient with myelomeningocele represents one of the most challenging spinal deformities encountered by the pediatric orthopedic surgeon. The deformity typically manifests at the thoracolumbar junction, the midlumbar spine, or the lumbosacral junction. While the origin of the deformity may be classified as congenital, developmental, or paralytic, the clinical consequences of a neglected gibbus deformity are universally devastating. Approximately 15% to 20% of patients born with myelomeningocele will develop a severe, progressive kyphosis that ultimately mandates surgical intervention.
Embryologically, the notochord is covered dorsally by the closure of the ectoderm, which progresses in a cephalad to caudal direction. In myelomeningocele, this closure is incomplete, most frequently at the caudal end. This lack of posterior migration of the ectoderm surrounding the notochord leaves the neural placode in a vulnerable position, resulting in the exposed myelomeningocele sac observed at birth.

As the child grows and assumes an upright sitting posture, the biomechanical disadvantages of the dysraphic spine become clinically apparent. The severe forward-flexed posture shifts the sagittal vertical axis far anterior to the pelvis. The C7 lateral plumb line demonstrates a profound sagittal imbalance. To maintain an upright posture and interact with their environment, these children often resort to the "marionette maneuver." This compensatory mechanism involves pivoting on extended upper extremities to unweight the diaphragm and support the torso. Consequently, the child is rendered functionally quadriplegic, as the upper extremities are entirely occupied with maintaining balance and facilitating respiration rather than engaging in functional tasks.

The life-threatening complications of this deformity are multifold. Recurrent skin breakdown over the apex of the kyphosis is ubiquitous in severe cases, leading to chronic ulcerations, deep wound infections, osteomyelitis, and potentially fatal central nervous system infections (meningitis). Furthermore, the severe abdominal crowding pushes the viscera cephalad into the thoracic cavity, severely compromising pulmonary capacity and leading to thoracic insufficiency syndrome. A compensatory thoracic lordosis cephalad to the kyphosis further exacerbates this respiratory compromise. Diminished gastrointestinal absorption and genitourinary stasis, often leading to renal calculi, are frequent secondary effects of the compressed abdominal cavity.
Surgical Anatomy and Biomechanics
The surgical anatomy of the myelomeningocele patient with severe kyphosis is highly distorted, presenting unique challenges for achieving stabilization and fusion. The kyphotic angle may present as a gradual slope or an acute, rigid gibbus.

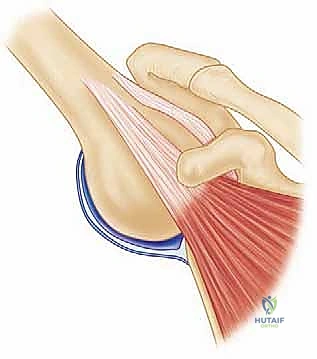
A fundamental biomechanical driver of this deformity is the anomalous position of the paraspinal musculature. Because of the embryologic failure of posterior element formation and the lack of posterior migration, the paraspinal muscles are displaced laterally and anteriorly relative to the axis of rotation of the spinal column. These muscles remain segmentally innervated and partially active. In their anomalous anterior position, they act as primary flexors of the spine rather than extensors, actively contributing to the progression of the kyphosis. Over time, gravity and active muscle contracture exacerbate the forward flexion of the spinal column.

The posterior bony elements in the area of the diastasis are widely splayed, leaving sparse, dysplastic bone available for a posterior fusion mass. The midline defect is characteristically covered by a fragile, adherent dura separated from the overlying skin by only a thin, poorly vascularized layer of scarred subcutaneous tissue.
Crucially, the vascular anatomy anterior to the spine dictates the surgical approach to reduction. The great vessels (the aorta and the inferior vena cava) do not follow the contours of the kyphotic apex. Instead, they span the bow of the deformity. Any surgical maneuver that lengthens the anterior column—such as a purely posterior opening wedge osteotomy or aggressive distraction—risks catastrophic stretching, tearing, or thrombosis of these great vessels. Therefore, spinal shortening via apical resection is a mandatory biomechanical principle in these procedures.
Distally, the sacral ala represents one of the most reliably formed vertebral structures in this patient population, providing a critical anchor point for pelvic fixation, which is almost universally required to counteract the massive cantilever forces exerted by the upper torso.
Indications and Contraindications
The decision to proceed with a kyphectomy in a patient with spina bifida must be carefully weighed against the formidable complication profile of the procedure. Bracing is universally ineffective for myelomeningocele kyphosis; it exacerbates skin pressure over the insensate gibbus and fails to halt curve progression.

Surgical intervention is generally indicated when the deformity threatens the patient's life or functional independence. The primary indications include recurrent or recalcitrant skin breakdown over the apex, progressive thoracic insufficiency syndrome, severe abdominal crowding compromising visceral function, and loss of sitting balance necessitating the marionette maneuver.
| Clinical Scenario | Operative Indication | Non Operative Management |
|---|---|---|
| Skin Integrity | Recurrent full-thickness ulceration over apex, chronic infection | Intact skin, mild erythema manageable with seating modifications |
| Pulmonary Status | Thoracic insufficiency syndrome, restrictive lung disease | Stable pulmonary function tests, no recurrent pneumonias |
| Sitting Balance | Loss of hands-free sitting, reliance on marionette maneuver | Independent sitting balance maintained |
| Curve Progression | Documented rapid progression, acute rigid gibbus | Slow progression in a medically fragile patient |
| Medical Stability | Optimized nutritional and pulmonary status | Uncorrectable coagulopathy, active systemic sepsis |
Contraindications include active systemic infection (though localized osteomyelitis at the apex may be resected during the procedure), profound malnutrition (albumin < 3.0 g/dL), uncorrectable coagulopathy, and extreme medical instability precluding a prolonged anesthetic course.
Pre Operative Planning and Patient Positioning
Thorough preoperative planning requires a multidisciplinary approach involving orthopedic surgery, neurosurgery, plastic surgery, urology, and pulmonology. Optimization of the patient's nutritional status and pulmonary function is paramount.
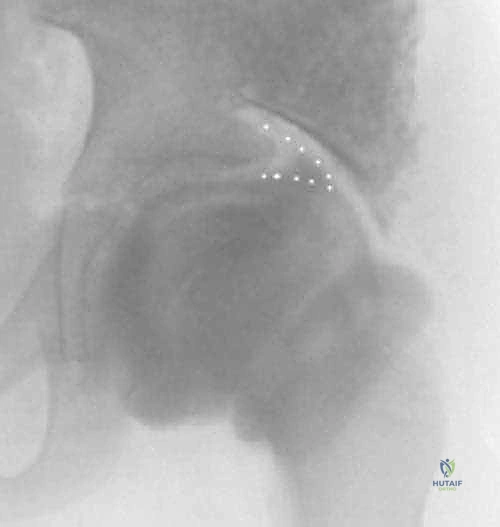
Imaging protocols must include full-length standing or sitting orthogonal radiographs to assess global sagittal and coronal balance. Fine-cut computed tomography (CT) with 3D reconstruction is critical for evaluating the dysplastic bony anatomy, measuring pedicle diameters for instrumentation, and assessing the sacral ala and ilium for pelvic fixation trajectories.

Magnetic resonance imaging (MRI) of the entire neural axis is mandatory. The surgeon must evaluate for hydrocephalus, Chiari malformations, syringomyelia, and the exact level of the functional neural placode. The presence of a tethered cord must be documented, as the kyphectomy procedure will inherently involve transection or manipulation of the distal thecal sac.

Patient Positioning Considerations
The patient is typically positioned prone on a Jackson spinal table or a specialized pediatric frame. Given the insensate nature of the patient's lower extremities and pelvis, meticulous attention must be paid to padding all bony prominences to prevent iatrogenic pressure necrosis.

The abdomen must hang completely free to prevent increased intra-abdominal pressure, which would subsequently increase central venous pressure and exacerbate epidural venous bleeding during the exposure. The hips are typically extended to maximize the lordosis of the pelvis, aiding in the eventual sagittal reduction of the spine.
Detailed Surgical Approach and Technique
The kyphectomy procedure is a massive undertaking that combines elements of neurosurgical placode management, aggressive spinal osteotomy, and complex pelvic reconstruction.
Incision and Soft Tissue Exposure
The surgical incision typically involves an elliptical excision of the scarred, poorly vascularized skin overlying the gibbus deformity.

Dissection is carried down through the subcutaneous tissues. The surgeon must be acutely aware that the dura is often immediately deep to the skin, lacking standard myofascial coverage. The dysplastic paraspinal muscles, located laterally, are mobilized subperiosteally to expose the lateral aspects of the vertebral bodies and the transverse processes.

Neural Placode Management and Thecal Sac Ligation
Coordination with neurosurgery is essential during this phase. In patients with no distal motor function, the thecal sac is identified proximal to the apex of the deformity. The dura is carefully opened, and the neural elements are inspected.

To facilitate the resection of the apical vertebrae and allow for profound spinal shortening, the thecal sac and the non-functional distal spinal cord are ligated and transected. This is typically performed using heavy non-absorbable sutures. Meticulous dural closure is imperative to prevent postoperative cerebrospinal fluid (CSF) fistulas, which are a leading cause of deep wound infections in this population.
Apical Vertebral Resection
Once the neural elements are managed, the orthopedic surgeon proceeds with the kyphectomy. The goal is to resect the apical vertebral bodies (typically 1 to 3 segments) to create a void that allows for spinal shortening and extension.

Subperiosteal dissection is carried circumferentially around the apical vertebrae. Segmental vessels are meticulously identified, ligated, and divided. Retractors are placed anterior to the vertebral bodies to protect the great vessels and abdominal viscera. The discs above and below the planned resection are excised, and the vertebral bodies are removed using a combination of osteotomes, rongeurs, and high-speed burrs. The resection must be wide enough to allow for complete closure of the osteotomy without hinging on the anterior cortex, which would dangerously stretch the great vessels.

Spinal Instrumentation and Reduction
Fixation in the osteopenic bone of a myelomeningocele patient requires multiple points of fixation. Proximally, pedicle screws are placed into the thoracic and upper lumbar vertebrae. Depending on bone quality, sublaminar wires or hooks may be used as adjuncts, though pedicle screws remain the gold standard for biomechanical purchase.
Distally, robust pelvic fixation is non-negotiable. The sacral ala, as noted, is generally well-formed. Fixation techniques have evolved from the classic Galveston rod technique to modern S2-alar-iliac (S2AI) screws or traditional iliac screws. S2AI screws offer the advantage of remaining in line with the cranial instrumentation, minimizing the need for complex rod contouring and reducing hardware prominence.

Once the anchors are placed, pre-contoured rods are introduced. The reduction is achieved by closing the osteotomy defect.

This maneuver must shorten the spinal column. The surgeon applies compression across the osteotomy site, bringing the proximal and distal spinal segments together. This corrects the kyphosis while relaxing the anterior soft tissues and great vessels.

Arthrodesis and Soft Tissue Closure
Extensive decortication of all exposed bony surfaces is performed. The resected apical bone is milled and used as autograft. Given the meager local bone stock, allograft or orthobiologics (such as bone morphogenetic protein, used off-label with caution due to the open dura) are frequently utilized to augment the fusion mass.
Soft tissue closure is arguably the most critical step in preventing postoperative morbidity. Because the native skin is often deficient and scarred, primary closure is frequently impossible or under excessive tension. Plastic surgery colleagues often perform complex myocutaneous advancements. Bilateral latissimus dorsi turnover flaps, gluteal fascial advancements, or keystone flaps are utilized to provide robust, vascularized soft tissue coverage over the prominent hardware.

Complications and Management
The complication rate following kyphectomy in spina bifida is notoriously high, with historical literature reporting complication rates exceeding 50%. The surgeon must be prepared to manage these complex issues aggressively.
One of the most frequent complications is hardware prominence and subsequent failure, particularly in patients with profound osteopenia.

Wound dehiscence and deep infection are life-threatening complications that require immediate operative debridement. The presence of a CSF leak exponentially increases the risk of meningitis and deep hardware infection.

| Complication | Estimated Incidence | Prevention and Salvage Strategies |
|---|---|---|
| Wound Dehiscence / Infection | 15% - 30% | Prophylactic plastic surgery flaps, meticulous dural closure. Salvage: Serial debridement, vacuum-assisted closure, hardware retention if stable. |
| Pseudarthrosis / Hardware Failure | 10% - 25% | Multi-point pelvic fixation (S2AI), robust bone grafting. Salvage: Revision instrumentation, anterior column support if feasible. |
| CSF Fistula | 5% - 15% | Watertight thecal sac ligation. Salvage: Over-sewing, lumbar drain placement, fibrin glue augmentation. |
| Great Vessel Injury / Thrombosis | 1% - 5% | Mandatory spinal shortening during reduction; avoid anterior distraction. Salvage: Immediate vascular surgery intervention. |
| Pressure Ulcers (Iatrogenic) | 5% - 10% | Meticulous intraoperative padding of insensate areas. Salvage: Local wound care, offloading, potential flap coverage. |
Post Operative Rehabilitation Protocols
The immediate postoperative period requires intensive care unit monitoring. Patients are typically kept on flat bed rest for 3 to 5 days to minimize tension on the dural closure and the complex soft tissue flaps. Hemodynamic monitoring is critical, as third-spacing and delayed bleeding can occur.
Once cleared by plastic surgery, mobilization is initiated gradually. The patient is transitioned to a custom-molded thoracolumbosacral orthosis (TLSO) to protect the instrumentation during the early phases of bony fusion.

Rehabilitation focuses on maximizing upper extremity strength and adapting to the new center of gravity. Custom wheelchair modifications are essential. The seating system must be re-evaluated and modified to accommodate the corrected posture, ensuring that pressure is distributed evenly across the ischial tuberosities and posterior thighs, avoiding any point loading on the newly reconstructed spine.
Summary of Key Literature and Guidelines
The surgical management of myelomeningocele kyphosis has evolved significantly over the past several decades. Early literature by Shurtleff and Lindseth highlighted the natural history of the disease, demonstrating that non-operative management inevitably leads to severe morbidity and early mortality due to cardiopulmonary compromise and sepsis.
The concept of the "kyphectomy" was popularized by Dunn and later refined by Warner, who emphasized the absolute necessity of spinal shortening. Their biomechanical studies demonstrated that the great vessels span the concavity of the deformity, and any attempt at lengthening the spine leads to catastrophic vascular injury.
Current academic consensus suggests delaying the kyphectomy until the child is at least 3 to 4 years of age, if clinically feasible. Operating on neonates, while advocated by some to facilitate initial skin closure, is associated with unacceptably high rates of pseudarthrosis, hardware pullout due to cartilaginous bone, and the need for multiple revision surgeries as the child grows. In the older child, the ossific nucleus of the pedicles and the sacral ala are better formed, allowing for more rigid internal fixation.
Recent advancements in pelvic fixation, particularly the transition from Galveston rods to S2-alar-iliac (S2AI) screws, have significantly decreased the rates of distal hardware failure and prominence. Furthermore, the routine integration of plastic surgery for primary flap coverage has drastically reduced the incidence of catastrophic wound dehiscence, representing the current standard of care in modern pediatric spinal deformity surgery.
Clinical & Radiographic Imaging




















You Might Also Like