Fracture Healing Understanding: How Bones Repair & What Treatments Help
Key Takeaway
In this comprehensive guide, we discuss everything you need to know about Fracture Healing Understanding: How Bones Repair & What Treatments Help. Fracture healing understanding involves a complex, sequential process restoring injured bone. It progresses through stages: inflammation (hematoma formation, cytokine release from immune cells like macrophages, neutrophils, and platelets), followed by reparative phases. Healing type (primary via Haversian remodeling or secondary via endochondral ossification) is dictated by mechanical stability and strain at the fracture site.
Introduction and Epidemiology
Fracture healing is a highly complex, orchestrated physiological process that, unlike most tissue repair mechanisms in the human body, results in true biological regeneration rather than fibrous scar formation. The ultimate goal of fracture management is to restore the anatomical alignment and biomechanical function of the skeletal system while minimizing the risk of complications. Understanding the intricate balance between the mechanical environment and the biological cascade is paramount for the orthopedic surgeon, as every operative and non-operative intervention directly alters this microenvironment.
Epidemiologically, fractures represent a significant global healthcare burden. Millions of fractures occur annually due to high-energy trauma, low-energy fragility mechanisms in the aging population, and repetitive stress injuries. While the vast majority of these fractures heal predictably with appropriate conservative or surgical management, approximately five to ten percent progress to delayed union or nonunion. This subset of patients experiences significant morbidity, prolonged functional impairment, and requires complex, resource-intensive reconstructive procedures.
The evolution of orthopedic traumatology has shifted from a purely mechanical approach—prioritizing absolute rigidity at all costs—to a more nuanced biological approach. Modern fracture management emphasizes the preservation of the soft tissue envelope, maintenance of the periosteal blood supply, and the application of fixation constructs that provide the optimal mechanical strain to stimulate osteogenesis. This paradigm shift necessitates a profound understanding of the cellular, molecular, and biomechanical principles that govern osseous repair.
Surgical Anatomy and Biomechanics
The skeletal system is a composite material exhibiting anisotropic and viscoelastic properties, meaning its biomechanical behavior depends on both the direction of the applied load and the rate of loading. To understand how bones repair, one must first comprehend the baseline structural anatomy and the vascular supply, which serve as the foundation for the regenerative cascade.
Osseous Architecture and Vascular Supply
Bone is structurally categorized into cortical (compact) and cancellous (trabecular) bone. Cortical bone, found primarily in the diaphysis of long bones, is dense and provides the majority of the mechanical strength against bending and torsional forces. It is organized into Haversian systems (osteons), which contain central canals housing blood vessels and nerves, interconnected by Volkmann canals. Cancellous bone, located in the metaphyses and epiphyses, consists of a porous, three-dimensional lattice of trabeculae that dissipates compressive loads and houses the osteoprogenitor-rich bone marrow.
The vascular supply to a long bone is tripartite, consisting of the nutrient artery system, the metaphyseal-epiphyseal system, and the periosteal system. The nutrient artery pierces the diaphysis and divides into ascending and descending branches, supplying the inner two-thirds of the mature cortex via the endosteal circulation. The periosteal vessels supply the outer one-third of the cortex. However, in the setting of a fracture, this physiological centripetal blood flow is disrupted. The endosteal supply is often compromised by the injury and subsequent intramedullary instrumentation, causing the periosteal system to transiently become the dominant blood supply to the healing callus. This physiological reversal underscores the critical importance of preserving periosteal attachments during surgical dissection.
Biomechanics of Fracture Healing and Strain Theory
The mechanical environment at the fracture site dictates the histological pathway of bone healing. This concept is best described by Perren's Strain Theory. Strain is defined as the relative change in the fracture gap length divided by the original gap length under physiological loading. The magnitude of this interfragmentary strain determines which type of tissue can survive and differentiate within the fracture gap.
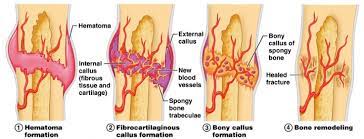
Granulation tissue can tolerate high strain environments (up to one hundred percent), whereas cartilage can tolerate intermediate strain (up to ten percent), and mature lamellar bone can only tolerate low strain (less than two percent). Therefore, if a fracture gap is subjected to high strain, bone cannot form directly. The body must first produce a voluminous soft callus (granulation tissue and cartilage) to increase the cross-sectional area of the healing tissue, which exponentially increases the area moment of inertia and decreases the strain at the fracture site. Once the strain drops below the critical threshold of two percent, hard bony callus can replace the cartilaginous scaffold.
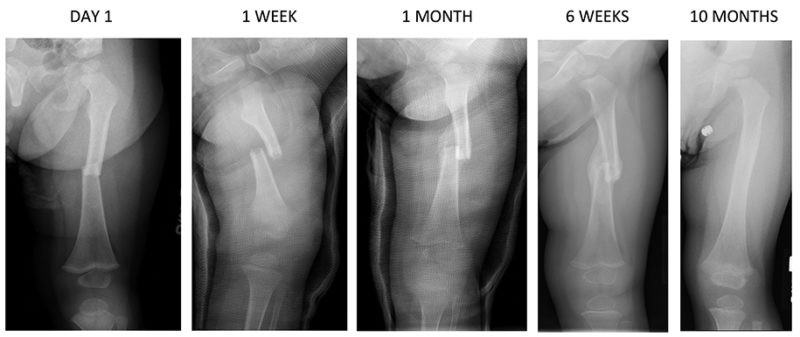
Primary vs Secondary Bone Healing
Based on the mechanical environment provided by the surgeon, bone heals via two distinct pathways.
Primary (direct) bone healing occurs under conditions of absolute stability and anatomical reduction, where interfragmentary strain is virtually zero (less than two percent). This requires rigid surgical fixation, typically achieved with interfragmentary lag screws and compression plating. Because there is no motion, no external callus forms. Healing occurs via direct osteonal remodeling. Cutting cones, led by osteoclasts, tunnel across the fracture line, followed closely by osteoblasts that lay down new lamellar bone. While mechanically rigid, this process is biologically slow and relies entirely on intact intramedullary and periosteal vascularity.
Secondary (indirect) bone healing is the natural physiological pathway and occurs under conditions of relative stability (strain between two and ten percent). This is seen with conservative management (casting, bracing) or flexible surgical constructs (intramedullary nails, bridge plates, external fixators). Secondary healing progresses through four overlapping phases. The initial inflammatory phase involves hematoma formation, macrophage infiltration, and the release of osteoinductive cytokines (Interleukin-1, Interleukin-6, Tumor Necrosis Factor-alpha). This is followed by the soft callus phase, characterized by chondrogenesis and the formation of a cartilaginous scaffold. The hard callus phase involves endochondral ossification, where the cartilage is mineralized and replaced by woven bone. Finally, the remodeling phase, governed by Wolff's Law, converts the woven bone into mature lamellar bone oriented along the lines of mechanical stress.
Indications and Contraindications
The decision to proceed with operative intervention versus non-operative management is multifactorial. The surgeon must evaluate the mechanical instability of the fracture, the biological capacity of the host, the condition of the soft tissue envelope, and the functional demands of the patient. Operative treatments are designed to either restore absolute stability for articular congruence or provide relative stability to align the mechanical axis while promoting secondary healing.
| Management Strategy | Indications for Use | Contraindications and Limitations |
|---|---|---|
| Non-Operative Management (Casting, Functional Bracing) | Undisplaced or minimally displaced fractures; Stable fracture patterns; High surgical risk patients; Pediatric diaphyseal fractures with high remodeling potential. | Open fractures; Displaced intra-articular fractures; Polytrauma requiring early mobilization; Fractures with associated vascular compromise; Pathological fractures. |
| Absolute Stability (Lag Screws, Compression Plating) | Displaced intra-articular fractures; Simple diaphyseal fractures (e.g., forearm) requiring exact anatomical restoration; Nonunion surgery requiring rigid compression. | Severe soft tissue compromise (e.g., Tscherne Grade III); Highly comminuted diaphyseal fractures where anatomical reduction would strip the periosteum; Active local infection. |
| Relative Stability (Intramedullary Nailing) | Diaphyseal fractures of the femur and tibia; Selected metaphyseal fractures; Pathological fractures requiring load-sharing constructs. | Open physes in pediatric patients (standard rigid nails); Narrow medullary canals; Active intramedullary infection. |
| Relative Stability (Bridge Plating / MIPO) | Comminuted metaphyseal and diaphyseal fractures; Periarticular fractures where the articular block is reconstructed but the metaphysis is bridged. | Simple transverse fractures where a gap is left (risk of high strain and nonunion); Severe skin compromise over the plate insertion sites. |
| External Fixation | Damage control orthopedics in polytrauma; Severe open fractures with massive soft tissue loss; Periarticular fractures with severe swelling (spanning constructs); Infected nonunions. | Definitive fixation in non-compliant patients; Situations where pin track infections would compromise planned definitive internal fixation incisions. |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is the cornerstone of successful orthopedic trauma surgery. The objective is to anticipate the biological and mechanical requirements of the fracture and formulate a strategic roadmap for reduction and fixation.
Optimization of the Biological Envelope
Before addressing the mechanical construct, the surgeon must evaluate the host's biological envelope. Patient optimization involves identifying and mitigating modifiable risk factors that impair the cellular and vascular responses necessary for osteogenesis. Smoking cessation is critical, as nicotine causes peripheral vasoconstriction, diminishes tissue oxygen tension, and inhibits osteoblast differentiation, significantly increasing the risk of nonunion. Glycemic control in diabetic patients must be strictly managed to prevent advanced glycation end-products from interfering with collagen cross-linking and to mitigate the risk of surgical site infections. Furthermore, the use of non-steroidal anti-inflammatory drugs (NSAIDs) should be carefully considered; while they provide excellent analgesia, cyclooxygenase-2 (COX-2) inhibition has been shown in animal models to delay endochondral ossification and suppress the inflammatory phase of secondary healing.
Imaging and Templating
High-quality orthogonal radiographs are mandatory. In complex periarticular fractures, computed tomography (CT) with three-dimensional reconstructions is essential for understanding the fracture morphology, identifying articular step-offs, and planning screw trajectories. Digital templating allows the surgeon to select the appropriate implant size, determine the working length of the construct, and anticipate the required reduction maneuvers.
Patient Positioning and Fluoroscopy
Positioning must facilitate both the surgical approach and unobstructed fluoroscopic imaging. For intramedullary nailing of the femur, the patient may be placed supine on a fracture table with traction applied, or in a lateral decubitus position on a radiolucent flat table, depending on the fracture level and surgeon preference. The fluoroscopy unit (C-arm) must be positioned to easily obtain true anteroposterior and lateral views without compromising the sterile field. Proper positioning ensures that gravity and muscle tension assist, rather than hinder, the reduction process.
Detailed Surgical Approach and Technique
The execution of the surgical plan must balance the mechanical need for reduction and fixation with the biological imperative to preserve tissue vitality. The evolution of minimally invasive techniques highlights the orthopedic community's recognition that surgical trauma can iatrogenically impair fracture healing.
Soft Tissue Management and Internervous Planes
Surgical approaches should utilize established internervous and intermuscular planes to minimize denervation and devascularization. For example, the anterior approach to the humerus utilizes the plane between the deltoid (axillary nerve) and the pectoralis major (pectoral nerves) proximally, and the brachialis (musculocutaneous nerve) and brachioradialis (radial nerve) distally. Meticulous soft tissue handling is required. Retractors should be placed carefully to avoid crushing the periosteum or neurovascular bundles. The periosteum, containing the highly osteogenic cambium layer, must never be aggressively stripped from the bone fragments.
Achieving Absolute Stability
When primary bone healing is the goal—such as in a displaced intra-articular fracture of the distal humerus or a simple transverse fracture of the radius—absolute stability is required. The technique involves open anatomical reduction, clearing the fracture site of interposed hematoma and soft tissue. Interfragmentary compression is achieved using lag screws. A lag screw is not a specific type of screw, but a biomechanical technique where the near cortex is overdrilled (gliding hole) and the far cortex is engaged (thread hole). As the screw head engages the near cortex, it compresses the two fragments together, eliminating interfragmentary strain.
This construct is typically protected by a neutralization plate, which resists torsional, bending, and shear forces that would otherwise cause the lag screw to fail. Alternatively, dynamic compression plates (DCP) or locking compression plates (LCP) can be used to generate axial compression across a transverse fracture utilizing the eccentric geometry of the plate holes. Because this technique devascularizes the bone ends to some degree, meticulous anatomical reduction is mandatory; any gap left under absolute stability will result in a nonunion, as primary healing cannot cross a macroscopic void.
Achieving Relative Stability
For multi-fragmentary diaphyseal or metaphyseal fractures, attempting to anatomically reduce every butterfly fragment requires extensive soft tissue stripping, which destroys the local blood supply and invariably leads to atrophic nonunion. In these scenarios, relative stability is employed to promote secondary healing via callus formation. The goal is to restore length, alignment, and rotation without disturbing the fracture hematoma.

Intramedullary nailing is the gold standard for diaphyseal fractures of the femur and tibia. It is a load-sharing device inserted via a remote entry portal (e.g., the piriformis fossa or greater trochanter for the femur). Reaming the medullary canal generates autologous bone graft that is deposited at the fracture site, stimulating osteogenesis. The nail acts as an internal splint, allowing controlled axial micromotion while resisting bending and torsion. This micromotion generates the ideal strain environment (two to ten percent) to stimulate robust soft and hard callus formation.
Minimally Invasive Plate Osteosynthesis (MIPO) is another application of relative stability. A long bridge plate is slid submuscularly and extraperiosteally across the comminuted fracture zone. Screws are placed only in the proximal and distal main fragments. By leaving the fracture zone entirely untouched, the fracture hematoma and periosteal blood supply are preserved. The working length of the plate (the distance between the two closest screws across the fracture) is intentionally kept long to increase the flexibility of the construct, thereby promoting secondary healing.
Complications and Management
Despite optimal surgical technique, complications in fracture healing can arise due to severe initial trauma, host factors, or mechanical failures. Recognizing these complications early and understanding their pathophysiology is critical for effective salvage.
| Complication | Pathophysiology and Incidence | Management and Salvage Strategies |
|---|---|---|
| Hypertrophic Nonunion | Adequate biology but inadequate mechanical stability. Characterized by abundant callus formation ("elephant foot") that fails to bridge the gap due to excessive strain. Incidence: 2-5% of fractures. | Improve mechanical stability. Typically requires exchange nailing (larger diameter nail) or the addition of compression plating. Bone grafting is rarely necessary as the biological potential is intact. |
| Atrophic Nonunion | Inadequate biology and vascularity. Often due to severe soft tissue stripping, smoking, or excessive rigid fixation with a gap. Characterized by no callus formation and sclerotic bone ends. Incidence: 3-7% of fractures. | Requires biological stimulation and stable fixation. Management involves debridement of sclerotic bone to bleeding edges, rigid internal fixation, and the application of autologous bone graft (e.g., iliac crest or RIA) or orthobiologics (BMP-2). |
| Oligotrophic Nonunion | Intermediate state with no obvious callus on radiograph, but biologically active tissue present. Often due to major displacement or minor biological impairment. | Requires a combination of improved stability and biological augmentation depending on the exact clinical presentation. |
| Infected Nonunion | Presence of active pyogenic infection at the fracture site, leading to bone necrosis, osteolysis, and implant colonization (biofilm formation). Incidence: 1-5% in closed fractures, up to 30% in severe open fractures. | Radical surgical debridement of all necrotic bone and hardware removal. Placement of antibiotic-impregnated cement spacers (Masquelet technique). Once infection is eradicated, definitive reconstruction with bone transport (Ilizarov) or massive grafting. |
| Malunion | Healing of the fracture in an unacceptable anatomical position (angulation, rotation, or shortening), leading to altered joint biomechanics and early post-traumatic osteoarthritis. | Corrective osteotomy. Preoperative planning with 3D modeling is used to design closing-wedge, opening-wedge, or derotational osteotomies, followed by rigid internal fixation. |
| Implant Failure | Mechanical fatigue of the plate, nail, or screws prior to fracture consolidation. Often a consequence of delayed union or nonunion where the implant is subjected to prolonged cyclical loading. | Revision surgery. Removal of broken hardware, addressing the underlying cause of delayed healing (biology vs stability), and revision with a more robust construct and biological augmentation. |
Post Operative Rehabilitation Protocols
The biological process of fracture healing does not end when the patient leaves the operating room; it is heavily influenced by the postoperative mechanical environment. Rehabilitation protocols must be carefully tailored to the specific fracture pattern, the type of surgical fixation, and the biological phase of healing.
Mechanotransduction and Wolff's Law
The foundation of postoperative rehabilitation is based on mechanotransduction—the process by which bone cells convert mechanical stimuli into biochemical signals. Osteocytes, embedded within the mineralized matrix, act as the primary mechanosensors. When physiological loads are applied, fluid flow within the canalicular network exerts shear stress on the osteocytes, downregulating the production of sclerostin (an inhibitor of Wnt/beta-catenin signaling) and upregulating osteoblastic bone formation. This is the cellular basis of Wolff's Law, which states that bone will adapt to the loads under which it is placed.
Weight-Bearing and Mobilization Strategies
For fractures treated with absolute stability (e.g., articular fractures fixed with lag screws and plates), the construct relies entirely on the implant to withstand physiological loads until primary healing occurs. Because primary healing is slow and there is no protective callus, early aggressive weight-bearing can lead to catastrophic implant failure. However, early active range of motion of the adjacent joints is mandatory to prevent capsular contracture and promote cartilage nutrition. Therefore, these patients typically undergo early mobilization with restricted weight-bearing for eight to twelve weeks.
Conversely, fractures treated with relative stability (e.g., diaphyseal fractures treated with intramedullary nails) benefit significantly from controlled mechanical loading. Intramedullary nails are load-sharing devices. Early weight-bearing induces axial micromotion at the fracture site, which stimulates the formation of a robust secondary callus. For stable diaphyseal femur or tibia fractures treated with locked intramedullary nails, weight-bearing as tolerated is often encouraged immediately postoperatively.
Dynamization
In cases where a diaphyseal fracture treated with a statically locked intramedullary nail shows signs of delayed secondary healing (sluggish callus formation at six to twelve weeks), the surgeon may opt for dynamization. This involves removing the interlocking screws from the longer segment of the bone (usually the dynamic slots). This converts the static construct into a dynamic one, allowing the bone fragments to axially compress under physiological load during weight-bearing, thereby increasing the mechanical stimulus for callus maturation.
Summary of Key Literature and Guidelines
The modern understanding of fracture healing and surgical intervention is built upon decades of foundational research and evolving clinical guidelines. Academic orthopedic surgeons must remain intimately familiar with the seminal literature that dictates current evidence-based practices.
Foundational Biomechanical Concepts
Stephan Perren's introduction of the Strain Theory in 1979 revolutionized the understanding of how the mechanical environment dictates tissue differentiation. His work established that the goal of fracture surgery is not always absolute rigidity, but rather the creation of an optimal strain environment tailored to the desired healing pathway. This led directly to the development of biological osteosynthesis and the design of modern locking plates, which function as internal fixators rather than friction-based compression plates.
The Diamond Concept
In the realm of biological augmentation, Giannoudis et al. introduced the "Diamond Concept" for fracture healing and nonunion management. This framework posits that successful bone regeneration requires the simultaneous presence of four key elements:
1. Osteogenic Cells: Mesenchymal stem cells and osteoprogenitors capable of differentiating into bone-forming cells.
2. Osteoconductive Scaffold: A structural matrix (e.g., autograft, allograft, or synthetic ceramics) that allows for cellular attachment and vascular ingrowth.
3. Osteoinductive Growth Factors: Biochemical signals, such as Bone Morphogenetic Proteins (BMP-2, BMP-7) and Platelet-Derived Growth Factor (PDGF), that direct cellular differentiation.
4. Mechanical Stability: An appropriate mechanical environment (absolute or relative stability) to allow the biological cascade to proceed without disruption.
Current Guidelines and Orthobiologics
Current guidelines from orthopedic trauma societies emphasize the judicious use of orthobiologics in high-risk fractures. The Reamer-Irrigator-Aspirator (RIA) system has emerged as a gold standard for harvesting large volumes of highly osteogenic autograft from the femoral or tibial canal with less donor-site morbidity than traditional iliac crest bone grafting. Furthermore, the use of recombinant human BMP-2 (rhBMP-2) is supported by extensive literature for the treatment of acute open tibial shaft fractures and recalcitrant nonunions, acting as a potent osteoinductive catalyst when the biological envelope is compromised.
In conclusion, the successful management of fractures requires the surgeon to act as both a biomechanical engineer and a cellular biologist. By meticulously planning the surgical approach, selecting the appropriate fixation construct, and manipulating the postoperative mechanical environment, the orthopedic surgeon can reliably guide the physiological cascade of fracture healing toward complete osseous regeneration.
You Might Also Like

