Operative Treatment of Radius and Ulna Diaphyseal Nonunions
Introduction and Epidemiology
A diaphyseal forearm fracture is formally classified as a nonunion when there is a complete cessation of osteogenic healing progression over a period of three consecutive months, typically assessed at least six to nine months post-injury, or when there is no clinical or radiographic likelihood that the fracture will progress to union without intervention (e.g., in the presence of a massive segmental defect). The forearm must be conceptualized not merely as two parallel long bones, but as a complex, bi-articular ring structure. Consequently, the failure of one or both bones to unite severely compromises the biomechanical integrity, load transmission, and spatial kinematics of the entire upper extremity.
Secondary to the widespread adoption of rigid internal fixation—specifically the principles of dynamic compression plating established by the AO Foundation—the overall incidence of forearm nonunions has decreased significantly over the past several decades. Current orthopedic literature cites nonunion rates following modern open reduction and internal fixation (ORIF) of the radius at approximately 2% and the ulna at 4%. However, in the setting of high-energy trauma, severe open fractures (Gustilo-Anderson Types II and III), ballistic injuries, and significant soft tissue compromise, the incidence increases substantially. Patient-specific risk factors, including active tobacco use, uncontrolled diabetes mellitus, malnutrition, and the chronic use of nonsteroidal anti-inflammatory drugs (NSAIDs) or corticosteroids, further elevate the risk of failed osteogenesis.

The pathogenesis of diaphyseal nonunions is classically categorized into biologic failure, mechanical failure, or a synergistic combination of both.
* Atrophic Nonunions: Most diaphyseal nonunions of the forearm, particularly those resulting from high-energy mechanisms or open fractures, are atrophic. These are characterized by a profound lack of biologic activity, avascular bone ends, and an absence of callus formation. They are frequently secondary to iatrogenic periosteal stripping, loss of the initial fracture hematoma, and significant bone loss.
* Hypertrophic Nonunions: These exhibit abundant, biologically active callus formation (often termed "elephant foot" or "horse hoof" callus) but fail to bridge the fracture gap due to inadequate mechanical stability.
* Oligotrophic Nonunions: These represent an intermediate state, possessing the biological potential to heal but lacking sufficient callus due to a combination of minor biological compromise and mechanical instability.
* Infected Nonunions: Infection remains a critical and devastating factor. Comminuted open fractures with associated bone loss and environmental contamination carry the highest rates of infected nonunion, necessitating a staged reconstructive approach.
Without surgical intervention, established nonunions of the forearm do not heal. The resultant loss of stability unhinges the entire mechanism of forearm motion, leading to a profound loss of pronation and supination. Because the kinematics of the proximal radioulnar joint (PRUJ) and distal radioulnar joint (DRUJ) are intricately dependent on the normal length and rotational relationships between the radius and ulna, motion at these articulations is invariably affected. Untreated, the resulting deformity and functional deficit become permanent, often accompanied by chronic pain, grip strength weakness, and secondary degenerative changes in adjacent joints.
Surgical Anatomy and Biomechanics
The radius and ulna form a functional, bi-articular unit that relies on precise anatomical relationships to facilitate complex multi-planar motion. The axis of rotation for the forearm extends obliquely from the center of the radiocapitellar joint proximally to the fovea of the ulna at the DRUJ distally. Any alteration in the length, alignment, or rotational orientation of either bone shifts this axis, resulting in profound kinematic disturbances and obligate subluxation of the radioulnar joints.
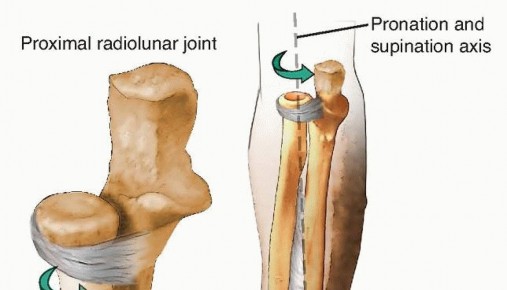
The radial bow is a critical anatomical parameter dictating the clearance of the radius around the ulna during pronation. The maximal radial bow typically measures approximately 15 millimeters (range 13-17 mm) and is located at the apex of the middle third of the radius, roughly 60% of the distance from the bicipital tuberosity to the radiocarpal joint. Restoration of this bow during nonunion surgery is paramount; failure to restore the anatomical bow directly correlates with a proportional loss of forearm rotation. Specifically, a loss of the radial bow primarily restricts pronation, while angular deformities restrict supination.
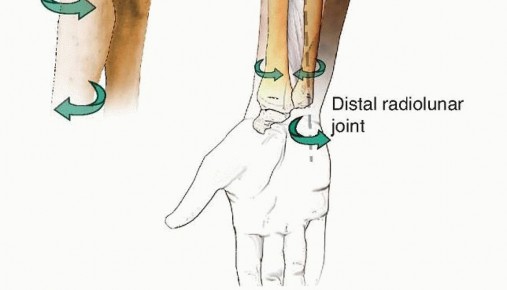
The interosseous membrane (IOM) connects the two diaphyses. Its central band, originating from the proximal radius and inserting on the distal ulna, acts as the primary stabilizer against longitudinal radioulnar dissociation. The IOM also partitions the forearm into volar and dorsal compartments. In the setting of a chronic nonunion, profound scarring often obliterates these normal tissue planes, complicating surgical dissection, tethering muscle excursions, and increasing the risk of iatrogenic injury to surrounding neurovascular structures.
Deforming muscle forces play a significant role in both the initial displacement of forearm fractures and the persistent instability seen in nonunions. For radial shaft nonunions, the deforming forces vary predictably based on the location relative to the pronator teres insertion:
* Proximal Third (Proximal to Pronator Teres): The proximal fragment is forcefully supinated by the biceps brachii and supinator muscles. The distal fragment is pronated by the pronator teres and pronator quadratus.
* Middle Third (Distal to Pronator Teres): The proximal fragment is maintained in neutral rotation (balanced by the supinating force of the supinator and the pronating force of the pronator teres), while the distal fragment is pronated by the pronator quadratus.
Extrinsic and intrinsic hand extensors and flexors originate extensively from the diaphyseal surfaces of both bones. Consequently, nonunions and the ensuing scar tissue can tether these muscle bellies, leading to secondary limitations in wrist and digital motion, necessitating meticulous tenolysis during reconstruction.
Indications and Contraindications
The management of forearm diaphyseal nonunions is almost exclusively operative. Nonoperative management is reserved only for patients who are medically unfit for surgical intervention or those with asymptomatic, stable pseudarthroses who have exceptionally low functional demands.
Operative Versus Nonoperative Management
| Parameter | Operative Management | Nonoperative Management |
|---|---|---|
| Primary Indications | Symptomatic nonunion, functional deficit, hardware failure, progressive deformity, infected nonunion | Asymptomatic nonunion in low-demand patient, absolute medical contraindications to anesthesia |
| Functional Goals | Restore length, alignment, radial bow, and pronosupination; eradicate infection; achieve bony union | Pain control, maintenance of basic activities of daily living (ADLs) |
| Bone Quality | Viable bone stock or reconstructable defects via autograft/allograft | Severe osteopenia or irradiated bone precluding stable fixation (relative) |
| Infection Status | Required for infected nonunions (staged approach: debridement/spacer followed by definitive fixation) | Contraindicated if active, systemic infection is present without source control |
| Patient Compliance | Requires adherence to postoperative rehabilitation and weight-bearing restrictions | Indicated for patients unable or unwilling to comply with postoperative protocols |
Contraindications to immediate definitive internal fixation include active, untreated deep infection or severely compromised soft tissue envelopes. In such cases, a staged protocol involving radical debridement, hardware removal, and placement of an antibiotic-impregnated polymethylmethacrylate (PMMA) spacer is mandatory prior to definitive skeletal reconstruction.
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is critical for the successful operative treatment of forearm nonunions. Standard orthogonal radiographs (anteroposterior and lateral) of the entire forearm, including the wrist and elbow, are mandatory to assess the nonunion site, hardware integrity, bone loss, and the status of the PRUJ and DRUJ.
Computed tomography (CT) with 3D reconstructions is highly recommended for evaluating the exact geometry of the nonunion, quantifying segmental bone defects, and assessing rotational malalignment. Furthermore, radiographs of the contralateral, uninjured forearm should be obtained to serve as a template for restoring the native radial bow and ulnar variance.
If an infected nonunion is suspected, a comprehensive laboratory workup including complete blood count (CBC), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) must be obtained. Preoperative aspiration or open biopsy may be necessary to identify the offending organism and guide targeted antimicrobial therapy.
Templating is essential to determine the appropriate implant size, length, and the volume of bone graft required. The surgeon must anticipate the need for extended approaches and the potential requirement for structural grafting (e.g., fibular strut autograft or allograft) versus cancellous autograft (e.g., iliac crest bone graft - ICBG, or Reamer-Irrigator-Aspirator (RIA) autograft from the femur).
Patient positioning typically involves placing the patient supine on the operating table with the affected extremity extended onto a radiolucent hand table. A sterile tourniquet is applied to the proximal arm. The ipsilateral iliac crest must be prepped and draped in the sterile field if autogenous bone grafting is anticipated. Fluoroscopy should be positioned to allow unobstructed imaging of the entire forearm in both AP and lateral planes without compromising the sterile field.
Detailed Surgical Approach and Technique
The surgical management of forearm nonunions demands meticulous soft tissue handling, anatomic reduction, rigid internal fixation, and optimization of the biological environment.
Surgical Approaches to the Forearm
The choice of surgical approach depends on the bone involved, the location of the nonunion, and the presence of prior incisions. Utilizing true internervous planes is critical to minimize denervation and preserve vascular supply.
Volar Approach to the Radius (Henry Approach):
This approach utilizes the internervous plane between the brachioradialis (innervated by the radial nerve) and the pronator teres/flexor carpi radialis (innervated by the median nerve).
1. The incision is made along a line connecting the biceps tendon proximally to the radial styloid distally.
2. Superficial dissection identifies and protects the superficial branch of the radial nerve (SRN) beneath the brachioradialis.
3. The radial artery is identified and protected medially. Ligation of the recurrent radial artery branches (the "leash of Henry") is often required to mobilize the brachioradialis laterally for proximal exposures.
4. Deep dissection involves elevating the supinator (protecting the posterior interosseous nerve - PIN) for proximal exposures, or the pronator quadratus for distal exposures.
Dorsal Approach to the Radius (Thompson Approach):
This approach utilizes the internervous plane between the extensor carpi radialis brevis (radial nerve) and the extensor digitorum communis (posterior interosseous nerve). It is generally preferred for proximal and middle third radial nonunions where dorsal plating is mechanically advantageous (tension band surface). The PIN must be explicitly identified and protected as it traverses the supinator muscle.
Approach to the Ulna:
The ulna is approached via a direct incision over its subcutaneous border, utilizing the internervous plane between the extensor carpi ulnaris (posterior interosseous nerve) and the flexor carpi ulnaris (ulnar nerve).
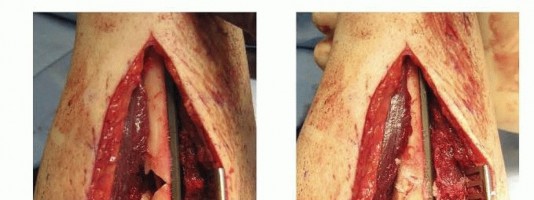
Nonunion Takedown and Preparation
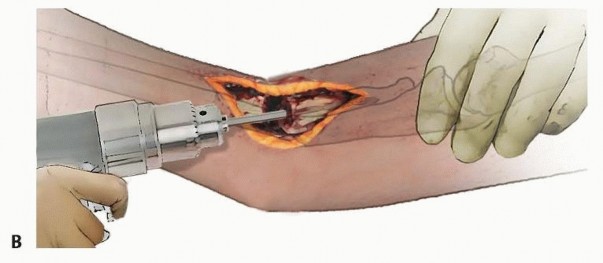
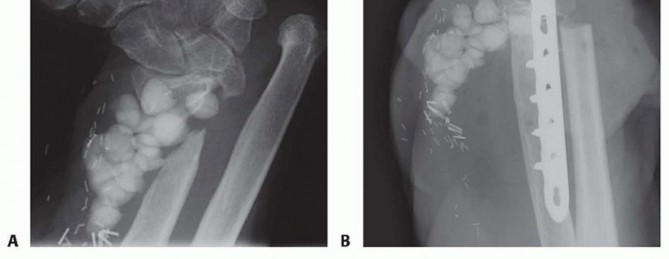
Upon exposure of the nonunion site, all previous hardware must be carefully removed. In cases of infected nonunions, multiple deep tissue cultures (minimum of 3-5 distinct samples) must be obtained before the administration of prophylactic antibiotics.
The nonunion site is meticulously debrided. All fibrous pseudarthrosis tissue, necrotic bone, and avascular scar tissue must be excised until healthy, bleeding cortical bone is encountered (the "paprika sign"). Inadequate debridement is a leading cause of recurrent nonunion.
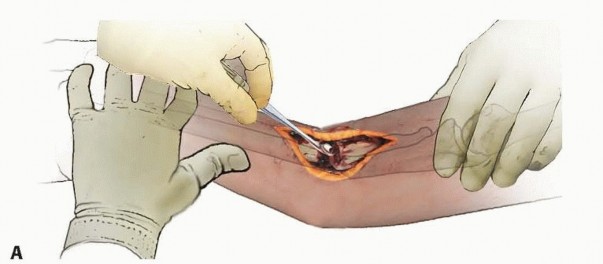
The medullary canals of both the proximal and distal fragments must be recanalized using curettes or drill bits to restore endosteal blood flow and allow for the ingress of osteoprogenitor cells. The cortical bone ends are then shingled or petalled with a sharp osteotome to increase the vascular surface area, creating a highly osteogenic bed for bone graft incorporation.
Bone Grafting Strategies
The strategy for biological augmentation depends on the size of the resultant defect after adequate debridement.
- Cancellous Autograft: For atrophic nonunions with intimate cortical contact or defects less than 1-2 cm, copious autogenous cancellous bone graft, typically harvested from the anterior or posterior iliac crest, is packed circumferentially around the nonunion site.
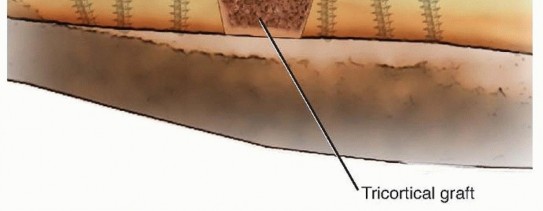
- Structural Grafting: For segmental defects ranging from 2 to 6 cm, a structural tricortical iliac crest graft or a fibular allograft strut may be utilized. The strut provides mechanical stability while cancellous autograft is packed around the host-graft junctions to promote osteogenesis.
- Masquelet Technique (Induced Membrane): For massive defects (greater than 6 cm) or in the setting of active infection, the induced membrane technique is highly effective. Stage one involves radical debridement, skeletal stabilization, and placement of a PMMA cement spacer. Stage two, performed 6-8 weeks later, involves careful incision of the induced pseudo-periosteal membrane, removal of the spacer, and packing the void with copious cancellous autograft (often combined with demineralized bone matrix or bone morphogenetic protein) before closing the biologically active membrane to contain the graft and provide vascularity.
Reduction and Fixation Techniques
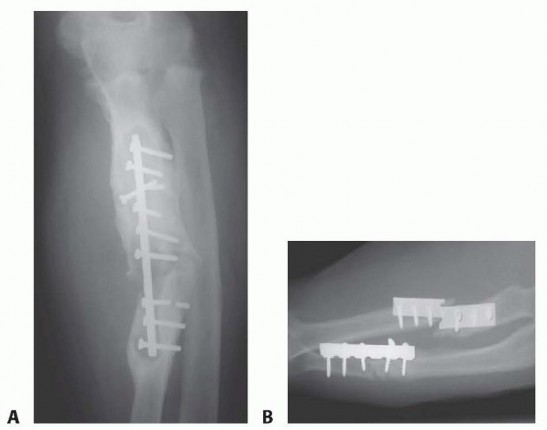
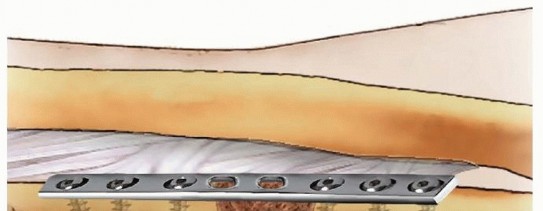
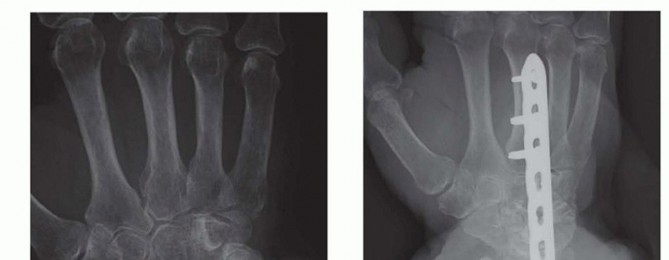
Rigid internal fixation is the cornerstone of nonunion surgery. A 3.5 mm dynamic compression plate (DCP), limited contact dynamic compression plate (LC-DCP), or locking compression plate (LCP) is the standard of care.
- Restoration of Anatomy: The radius and ulna must be restored to their anatomic length. Traction, laminar spreaders, or a push-pull screw technique can be utilized to overcome soft tissue contractures. The radial bow must be meticulously recreated, guided by preoperative templating.
- Plate Application: The plate should be applied to the tension band surface of the bone where possible (dorsal for the radius, posterior for the ulna). A minimum of 8 to 10 cortices of fixation proximal and distal to the nonunion site is strictly required to withstand the torsional forces of the forearm.
- Compression vs Bridge Plating: If the nonunion ends are transverse and well-vascularized after debridement, axial compression is applied using eccentrically placed screws. If a structural graft or segmental defect is present, the plate must function as a bridge plate. Locking screws are highly advantageous in the setting of osteopenic bone, which is frequently encountered in chronic nonunions, to prevent hardware pullout.
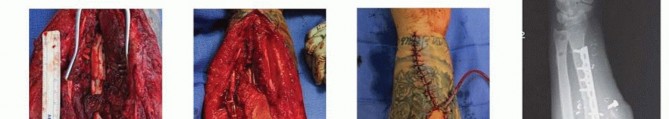
Meticulous hemostasis is achieved, and the wounds are closed in layers. The deep fascia is generally left open to prevent compartment syndrome, particularly given the extensive soft tissue dissection, prolonged retraction, and resultant swelling inherent to nonunion takedowns.
Complications and Management
The operative treatment of forearm nonunions is technically demanding and carries a significantly higher complication rate than primary fracture fixation.
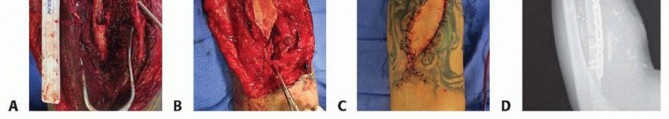
Common Complications and Salvage Strategies
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Strategy and Management |
|---|---|---|---|
| Persistent Nonunion | 5 - 10% | Inadequate fixation (insufficient plate length), persistent infection, failure to restore biology (inadequate debridement), smoking, NSAID use. | Revision ORIF with extended plating (dual plating if necessary), revision bone grafting (consider vascularized free fibula for recalcitrant cases), optimization of host metabolic factors. |
| Deep Infection | 3 - 8% | Open fractures, prolonged operative time, massive soft tissue stripping, history of prior infection. | Aggressive surgical I&D, retention of stable hardware (if union is progressing), removal of loose hardware, culture-directed IV antibiotics. May require staged Masquelet reconstruction. |
| Radioulnar Synostosis | 2 - 6% | Same-incision approaches, high-energy trauma, concurrent head injury, bone graft extravasation into the IOM. | Observation until the synostosis is radiographically mature (12-18 months). Surgical excision of the cross-union followed by prophylaxis (low-dose radiation therapy or indomethacin) to prevent recurrence. |
| Nerve Injury (PIN / SRN) | 1 - 5% | Iatrogenic injury during dissection (Thompson approach for PIN, Henry approach for SRN), excessive retractor pressure. | Intraoperative microsurgical repair if transected. If neurapraxia is suspected, observation and supportive care. Tendon transfers (e.g., pronator teres to ECRB) for permanent PIN palsy. |
| Hardware Failure | 2 - 5% | Premature weight-bearing, inadequate plate length (stress risers), failure of biological union leading to fatigue failure of the implant. | Revision surgery with longer, stronger plates (e.g., 3.5mm broad LCP), structural bone grafting to eliminate bending moments. |
| Loss of Motion | > 50% | Failure to restore radial bow, soft tissue contracture, prolonged immobilization, PRUJ/DRUJ incongruity. | Aggressive physical therapy. Surgical release (capsulectomy) is rarely indicated unless the functional deficit is severe and the bony anatomy is perfectly restored. |
Post Operative Rehabilitation Protocols
Postoperative rehabilitation must be carefully balanced between protecting the internal fixation and preventing debilitating stiffness of the elbow, forearm, and wrist. The specific protocol is dictated by the rigidity of the fixation, the quality of the host bone, and the extent of the biological reconstruction.
Phase I: Immobilization and Edema Control (0-2 Weeks)
Immediately postoperatively, the arm is placed in a bulky Jones dressing with a sugar-tong splint to control edema and immobilize the wrist and elbow. Active and passive range of motion of the digits and shoulder is initiated immediately in the recovery room to prevent distal and proximal stiffness and to encourage venous return.
Phase II: Early Motion (2-6 Weeks)
At the first postoperative visit, sutures or staples are removed. If rigid fixation was achieved and host bone quality is adequate, the splint is transitioned to a removable functional brace. Supervised active and active-assisted range of motion of the elbow, wrist, and forearm (pronation/supination) is initiated. Lifting is strictly restricted to less than 1-2 pounds (e.g., the weight of a coffee cup). If fixation is tenuous or a massive structural graft was utilized, immobilization in a Muenster or long-arm cast may be extended for 4-6 weeks to protect the construct.
Phase III: Strengthening and Maturation (6-12+ Weeks)
Serial orthogonal radiographs are obtained at 6 weeks, 12 weeks, and 6 months to assess for progressive callus formation, maintenance of alignment, and graft incorporation. Once clinical and radiographic signs of union are evident (typically around 8-12 weeks), progressive resistance exercises are introduced. Return to heavy manual labor or contact sports is strictly prohibited until complete radiographic consolidation is confirmed, which may take up to 6-9 months following complex nonunion surgery.
Summary of Key Literature and Guidelines
The evolution of forearm nonunion management is deeply rooted in the advancement of rigid internal fixation. The classic studies by Anderson et al. demonstrated the profound efficacy of compression plating, establishing the 3.5 mm DCP as the gold standard. This dramatically reduced the historical nonunion rates associated with cast immobilization or flexible intramedullary devices.
Ring and Jupiter's extensive work on forearm nonunions emphasized the critical importance of biological preparation. Their series highlighted that atrophic nonunions require meticulous decortication, opening of the medullary canals, and autogenous bone grafting, coupled with rigid plate fixation, to achieve union rates exceeding 90%.
For massive segmental defects, the literature has shifted from predominantly utilizing vascularized fibular autografts to the widespread adoption of the Masquelet (induced membrane) technique. Recent comparative studies indicate that while vascularized fibular grafting provides immediate structural integrity and biological viability, it is technically demanding, requires microsurgical expertise, and carries significant donor site morbidity. The Masquelet technique offers a highly reliable alternative, demonstrating comparable union rates for defects up to 10 cm, with the added benefit of local infection control during the first stage of reconstruction.
Current academic guidelines mandate a comprehensive, biologically and mechanically sound approach: eradication of infection, restoration of precise anatomical parameters (length, alignment, and radial bow), rigid internal fixation utilizing modern locking plate technology where appropriate, and aggressive biological augmentation. Adherence to these principles provides the highest probability of restoring functional utility to the upper extremity following a diaphyseal forearm nonunion.
You Might Also Like