Arixtra (Fondaparinux): A Comprehensive Medical SEO Guide
As an expert Medical SEO Copywriter and Orthopedic Specialist, we understand the critical role of effective anticoagulation in preventing and treating venous thromboembolism (VTE). Arixtra, with its active ingredient fondaparinux, represents a significant advancement in this field. This exhaustive guide provides an authoritative overview of Arixtra, delving into its mechanism, indications, pharmacokinetics, dosage, and important safety considerations.
1. Comprehensive Introduction & Overview
Arixtra (fondaparinux sodium) is a synthetic, selective Factor Xa inhibitor, a class of anticoagulant medications designed to prevent and treat blood clots. Unlike traditional heparins, fondaparinux is a pentasaccharide, a small, highly specific molecule that indirectly inhibits Factor Xa via antithrombin. This specificity contributes to its predictable anticoagulant response and a reduced risk of certain heparin-associated complications, such as heparin-induced thrombocytopenia (HIT).
Developed for its efficacy and safety profile, Arixtra has become a cornerstone in the management of thrombotic disorders, particularly in high-risk orthopedic surgical settings (e.g., hip fracture surgery, major knee surgery, hip replacement surgery) where the risk of deep vein thrombosis (DVT) and pulmonary embolism (PE) is substantial. Its fixed-dose regimen and lack of a need for routine coagulation monitoring simplify its use, making it a valuable tool for healthcare professionals and patients alike. This guide aims to equip both clinicians and patients with a thorough understanding of Arixtra's clinical utility and safety profile.
2. Deep-Dive into Technical Specifications & Mechanisms
Mechanism of Action: The Selective Factor Xa Inhibition
Fondaparinux exerts its anticoagulant effect by selectively inhibiting Factor Xa, a crucial enzyme in the coagulation cascade. It achieves this indirectly through its high affinity for antithrombin III (ATIII). When fondaparinux binds to ATIII, it causes a conformational change that markedly enhances ATIII's natural ability to neutralize Factor Xa.
- Indirect Inhibition: Fondaparinux itself does not directly inactivate Factor Xa. Instead, it acts as a catalytic template, accelerating the ATIII-mediated inhibition of Factor Xa by approximately 300-fold.
- Specificity: This mechanism is highly specific to Factor Xa. Unlike unfractionated heparin (UFH) and low molecular weight heparins (LMWHs), fondaparinux does not directly inhibit thrombin (Factor IIa) and does not bind to platelet Factor 4. This specificity is a key differentiator, contributing to a lower risk of HIT.
- Prevention of Thrombin Generation: By inhibiting Factor Xa, fondaparinux effectively prevents the conversion of prothrombin to thrombin, thereby disrupting the intrinsic and extrinsic pathways of the coagulation cascade and ultimately preventing clot formation and propagation.
Pharmacokinetics: Absorption, Distribution, Metabolism, and Excretion
Understanding the pharmacokinetics of fondaparinux is essential for appropriate dosing and patient management, especially in populations with altered renal function.
- Absorption:
- Administered via subcutaneous (SC) injection.
- Rapid and complete absorption.
- Peak plasma concentrations are typically reached within 2-3 hours post-dose.
- Absolute bioavailability is 100%.
- Distribution:
- Small volume of distribution (7-11 liters), primarily confined to the blood.
- Highly bound to antithrombin III, but not to other plasma proteins or cells, including platelets.
- Metabolism:
- Fondaparinux is not metabolized by cytochrome P450 (CYP450) enzymes.
- It does not undergo hepatic metabolism and does not produce active metabolites. This minimizes the potential for drug-drug interactions related to hepatic enzyme systems.
- Elimination:
- Primarily excreted unchanged by the kidneys.
- The elimination half-life is approximately 17-21 hours in individuals with normal renal function, allowing for once-daily dosing.
- Renal impairment significantly prolongs the half-life, necessitating dose adjustments or contraindication in severe cases.
Pharmacokinetic Profile Summary Table:
| Parameter | Description | Clinical Relevance |
|---|---|---|
| Administration | Subcutaneous injection | Convenient, high bioavailability. |
| Bioavailability | 100% | Predictable drug levels. |
| Peak Plasma Conc. | 2-3 hours | Rapid onset of action. |
| Metabolism | None (not via CYP450) | Low risk of hepatic drug interactions. |
| Elimination | Renal excretion (unchanged) | Requires dose adjustment/contraindication in renal impairment. |
| Half-life | 17-21 hours (normal renal function) | Once-daily dosing. |
3. Extensive Clinical Indications & Usage
Arixtra's potent and specific anticoagulant properties make it suitable for a range of indications, primarily focused on the prevention and treatment of venous thromboembolism.
Detailed Indications
- Prophylaxis of Venous Thromboembolism (VTE):
- Orthopedic Surgery:
- Hip Fracture Surgery: Reducing the risk of DVT and PE in patients undergoing hip fracture surgery.
- Major Knee Surgery: Prophylaxis in patients undergoing major knee replacement surgery.
- Hip Replacement Surgery: Prevention of DVT and PE following elective hip replacement surgery.
- Abdominal Surgery: Prophylaxis in patients undergoing abdominal surgery who are at high risk of thromboembolic complications (e.g., those with malignancy, advanced age, or a history of VTE).
- Medical Patients at Risk: Prevention of VTE in acutely ill medical patients who are at risk for thromboembolic complications due to restricted mobility during acute illness.
- Orthopedic Surgery:
- Treatment of Acute Deep Vein Thrombosis (DVT):
- Used in conjunction with warfarin or other oral anticoagulants (OACs) for the initial treatment of acute DVT. Arixtra provides rapid anticoagulation while the OAC reaches therapeutic levels.
- Treatment of Acute Pulmonary Embolism (PE):
- Used in conjunction with warfarin or other OACs for the initial treatment of acute PE, including cases without hemodynamic instability.
Dosage Guidelines
Dosage of Arixtra varies depending on the indication, patient weight, and renal function. It is administered via subcutaneous injection.
- VTE Prophylaxis (Orthopedic & Abdominal Surgery, Medical Patients):
- Standard Dose: 2.5 mg once daily, administered subcutaneously.
- Initiation: First dose typically given 6 to 8 hours post-surgery, provided hemostasis has been established. For medical patients, initiation can be at any time.
- Duration: Usually 5 to 9 days, but may be extended for up to 24-35 days in certain orthopedic high-risk patients.
- Treatment of Acute DVT and PE:
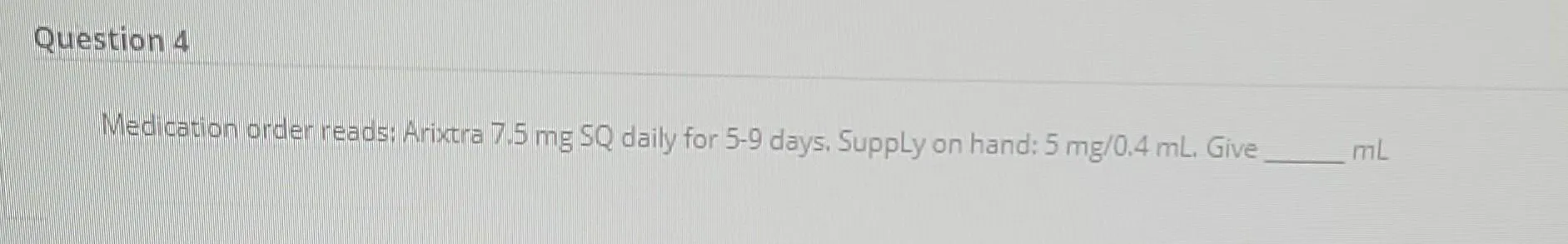
- Weight-Based Dosing:
- Body weight < 50 kg: 5 mg once daily, subcutaneously.
- Body weight 50-100 kg: 7.5 mg once daily, subcutaneously.
- Body weight > 100 kg: 10 mg once daily, subcutaneously.
- Duration: Typically continued for at least 5 days or until a stable therapeutic INR (usually 2.0-3.0) is achieved with an overlapping oral anticoagulant.
- Weight-Based Dosing:
Dosage Adjustment in Renal Impairment:
| Creatinine Clearance (CrCl) | Prophylaxis Dose (2.5 mg) | Treatment Dose (5 mg, 7.5 mg, 10 mg) |
|---|---|---|
| > 50 mL/min | No dose adjustment. | No dose adjustment. |
| 30-50 mL/min | Use with caution; consider reduced dose. | Use with caution; consider reduced dose. |
| < 30 mL/min | Contraindicated. | Contraindicated. |
- Administration Technique: Arixtra is administered by deep subcutaneous injection, alternating injection sites (e.g., abdominal wall, thigh). It should not be administered intramuscularly.
4. Risks, Side Effects, and Contraindications
While Arixtra is a highly effective anticoagulant, it carries inherent risks, primarily related to bleeding. Careful patient selection and monitoring are crucial.
Contraindications
Arixtra is contraindicated in patients with:
- Severe Renal Impairment: Creatinine clearance < 30 mL/min for VTE prophylaxis and < 20 mL/min for DVT/PE treatment.
- Active Major Bleeding: Any ongoing major bleeding episode.
- Bacterial Endocarditis: Due to the risk of intracranial hemorrhage.
- Hypersensitivity: Known hypersensitivity to fondaparinux or any excipients.
- Body Weight < 50 kg: For the treatment of acute DVT/PE, as this increases the risk of bleeding.
Common Side Effects
The most frequently reported adverse effects are related to bleeding.
- Bleeding:
- Injection site bleeding, ecchymosis, hematoma.
- Gastrointestinal bleeding (e.g., melena, hematemesis).
- Genitourinary bleeding (e.g., hematuria).
- Epistaxis (nosebleeds).
- Anemia: Often secondary to blood loss.
- Injection Site Reactions: Pain, redness, irritation.
- Other: Nausea, constipation, rash, fever, peripheral edema.
Serious Side Effects
- Major Hemorrhage: Intracranial hemorrhage, retroperitoneal hemorrhage, or other life-threatening bleeding episodes. This is the most serious complication.
- Thrombocytopenia: While rare and non-heparin induced (unlike HIT), a decrease in platelet count can occur.
- Anaphylactic Reactions: Though uncommon, severe allergic reactions requiring immediate medical attention are possible.
Drug Interactions
Concomitant use of Arixtra with other medications that affect hemostasis can significantly increase the risk of bleeding.
- Other Anticoagulants: Warfarin, other direct oral anticoagulants (DOACs), unfractionated heparin, low molecular weight heparins (LMWHs) – concurrent use should be managed with extreme caution or avoided unless clinically justified for bridging.
- Antiplatelet Agents: Aspirin, NSAIDs (non-steroidal anti-inflammatory drugs), clopidogrel, prasugrel, ticagrelor, dipyridamole – these increase the risk of bleeding.
- Thrombolytic Agents: Streptokinase, alteplase – significantly increase bleeding risk.
- SSRIs/SNRIs: Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) may increase bleeding risk.
Pregnancy and Lactation Warnings
- Pregnancy: Arixtra is classified as Pregnancy Category B. Animal studies have shown no evidence of harm to the fetus, but there are no adequate and well-controlled studies in pregnant women. It should be used during pregnancy only if clearly needed and the potential benefit justifies the potential risk to the fetus. The drug does not cross the placenta in significant amounts based on animal data, but human data is limited.
- Lactation: It is unknown whether fondaparinux is excreted in human milk. Caution should be exercised when Arixtra is administered to a nursing mother. The decision to discontinue nursing or discontinue the drug should consider the importance of the drug to the mother.
Overdose Management
There is no specific antidote for fondaparinux. Management of an overdose or excessive bleeding involves:
- Discontinuation: Immediately discontinue Arixtra.
- Supportive Care: Provide supportive measures, including pressure on bleeding sites, surgical hemostasis if necessary, and fluid replacement for hypovolemia.
- Blood Product Transfusion: Transfusion of red blood cells or fresh frozen plasma may be considered for significant blood loss.
- Recombinant Factor VIIa (rFVIIa) or Prothrombin Complex Concentrates (PCCs): These agents have been used in severe, life-threatening bleeding events associated with fondaparinux, but evidence of their efficacy as reversal agents is limited and not officially endorsed. They act by bypassing the coagulation cascade inhibition.
- Dialysis: Fondaparinux is highly protein-bound (to ATIII) and has a relatively small volume of distribution, making it unlikely to be effectively removed by hemodialysis.
5. Massive FAQ Section
Q1: What is Arixtra used for?
A1: Arixtra (fondaparinux) is primarily used to prevent blood clots (deep vein thrombosis and pulmonary embolism) after certain orthopedic surgeries (hip fracture, knee replacement, hip replacement) and in high-risk abdominal surgery patients or acutely ill medical patients. It is also used to treat existing acute DVT and PE, often in conjunction with other anticoagulants.
Q2: How is Arixtra administered?
A2: Arixtra is administered as a subcutaneous (SC) injection, typically once daily. It is usually injected into the fatty tissue just under the skin, often in the abdomen or thigh. Your healthcare provider will show you the correct technique if you need to administer it yourself.
Q3: Does Arixtra require routine blood monitoring like warfarin?
A3: No, one of the advantages of Arixtra is that it does not require routine coagulation monitoring (e.g., INR or aPTT) because it provides a predictable anticoagulant response at fixed doses.
Q4: What should I do if I miss a dose of Arixtra?
A4: If you miss a dose, take it as soon as you remember on the same day. Do not take a double dose to make up for a missed one. If it's almost time for your next dose, skip the missed dose and continue with your regular schedule. Contact your doctor or pharmacist if you are unsure.
Q5: Can I take Arixtra with other medications?
A5: You must inform your doctor about all medications you are taking, including over-the-counter drugs, supplements, and herbal remedies. Arixtra can interact with other anticoagulants, antiplatelet agents (like aspirin or NSAIDs), and certain other drugs, significantly increasing the risk of bleeding.
Q6: What are the signs of bleeding I should watch for while on Arixtra?
A6: You should immediately report any signs of unusual bleeding to your doctor, such as:
* Easy bruising or prolonged bleeding from cuts.
* Nosebleeds or bleeding gums.
* Red or dark brown urine (hematuria).
* Red or black, tarry stools (melena).
* Vomiting blood or material that looks like coffee grounds.
* Severe headache, dizziness, or weakness (could indicate internal bleeding).
Q7: Is Arixtra safe during pregnancy or while breastfeeding?
A7: Arixtra is classified as Pregnancy Category B, meaning animal studies have not shown harm, but human data is limited. It should only be used during pregnancy if clearly needed and the potential benefits outweigh the risks. It is unknown if Arixtra passes into breast milk, so caution is advised for nursing mothers. Discuss with your doctor.
Q8: How long will I need to take Arixtra?
A8: The duration of Arixtra treatment depends on the specific indication. For VTE prophylaxis after surgery, it's typically 5 to 9 days, but can be extended up to 24-35 days for high-risk orthopedic patients. For the treatment of DVT or PE, it's usually for at least 5 days, or until an oral anticoagulant reaches therapeutic levels. Your doctor will determine the appropriate duration for your condition.
Q9: What is the main difference between Arixtra and heparin?
A9: Arixtra (fondaparinux) is a synthetic, selective Factor Xa inhibitor, which means it specifically targets Factor Xa in the clotting cascade. Heparin (unfractionated heparin or low molecular weight heparins) inhibits both Factor Xa and thrombin (Factor IIa). Arixtra's specificity contributes to its predictable effect and a significantly lower risk of heparin-induced thrombocytopenia (HIT).
Q10: Who should not take Arixtra?
A10: Arixtra is contraindicated in individuals with severe kidney impairment (CrCl < 30 mL/min for prophylaxis, < 20 mL/min for treatment), active major bleeding, bacterial endocarditis, or a known hypersensitivity to fondaparinux. It is also not recommended for the treatment of DVT/PE in patients weighing less than 50 kg.
Q11: Can Arixtra cause hair loss?
A11: Hair loss is not a commonly reported side effect or a known adverse drug reaction specifically associated with Arixtra (fondaparinux) in its clinical trials or post-marketing surveillance. If you experience hair loss while on Arixtra, it would be important to discuss this with your doctor to explore other potential causes.
Q12: How quickly does Arixtra start working?
A12: Arixtra is rapidly absorbed after subcutaneous injection, with peak plasma concentrations typically reached within 2-3 hours. This allows for a relatively quick onset of its anticoagulant effect, making it suitable for immediate prophylaxis or initial treatment of thrombotic events.