Vascularized Bone Grafts for Scaphoid Nonunion
Key Takeaway
Vascularized bone grafts are a critical surgical intervention for recalcitrant scaphoid nonunions, particularly those complicated by proximal pole avascular necrosis or failed prior reconstructions. By restoring local blood supply alongside structural osteoconduction, pedicled grafts—such as the pronator quadratus flap—significantly enhance union rates. This guide details the indications, biomechanical principles, and step-by-step surgical techniques required to optimize outcomes in complex carpal nonunions and naviculocapitate fracture syndromes.
Comprehensive Introduction and Patho-Epidemiology
The treatment of scaphoid nonunions remains one of the most formidable and technically demanding challenges in hand and wrist surgery. The scaphoid bone occupies a critical anatomical and biomechanical position, serving as the essential mechanical link between the proximal and distal carpal rows. Because of the scaphoid’s unique retrograde intraosseous vascular anatomy—where the primary blood supply enters distally and flows proximally—fractures through the waist or proximal pole frequently disrupt perfusion to the proximal fragment. This ischemic environment heavily predisposes the patient to avascular necrosis (AVN) and recalcitrant nonunion, leading to a cascade of carpal instability, progressive collapse, and ultimately, degenerative arthrosis.
While conventional non-vascularized autografts, such as the classic Matti-Russe technique or structural iliac crest wedge grafts, are highly successful for well-perfused nonunions with minimal structural collapse, their efficacy drops precipitously in the presence of proximal pole ischemia or in the revision surgical setting. In these complex scenarios, the ischemic proximal pole is entirely dependent on the slow, unpredictable process of creeping substitution for graft incorporation. The use of vascularized bone grafts (VBGs) has proven to be a highly effective, biologically superior method to circumvent this limitation. By transferring living, perfused osteocytes, osteoblasts, and osteoprogenitor cells directly into the hostile nonunion site, VBGs bypass the prolonged phase of creeping substitution, immediately re-establishing local vascularity, accelerating osteogenesis, and maximizing the probability of bony union.
Since Braun’s seminal 1983 report detailing the success of a pronator quadratus pedicled graft harvested from the distal radius, the surgical armamentarium for vascularized carpal reconstruction has expanded significantly. Today, multiple sources of pedicled flaps from the distal radius, ulna, and metacarpals have been described and refined. Prominent examples include the 1,2 intercompartmental supraretinacular artery (1,2 ICSRA) graft popularized by Zaidemberg, distal dorsolateral radius grafts, and volar carpal artery pedicled grafts. Furthermore, for the most challenging cases involving severe bone loss or complete proximal pole necrosis, microvascular free flaps such as the medial femoral condyle (MFC) or iliac crest free flap have revolutionized the salvage of the ischemic scaphoid.
Although vascularized pedicled grafts are highly effective for promoting biological healing, meticulous patient selection and rigorous preoperative assessment are paramount to achieving optimal outcomes. As extensively noted in the literature by Kawai, Yamamoto, and others, the presence of established radiocarpal arthrosis—specifically Scaphoid Nonunion Advanced Collapse (SNAC)—may severely compromise the functional outcome, even if radiographic union of the scaphoid is successfully achieved. In such advanced stages of carpal derangement, biological reconstruction of the scaphoid is generally contraindicated, and salvage procedures such as proximal row carpectomy or partial wrist arthrodesis are preferred to provide a durable, pain-free wrist.
Detailed Surgical Anatomy and Biomechanics
Vascular Anatomy of the Scaphoid
A profound understanding of the scaphoid's extraosseous and intraosseous vascularity is the foundational prerequisite for any surgeon undertaking carpal reconstruction. The scaphoid derives its primary arterial supply from the radial artery, which provides distinct dorsal and volar branches. The predominant blood supply—accounting for 70% to 80% of the intraosseous perfusion—arises from the dorsal carpal branch of the radial artery. These vessels enter the scaphoid through a narrow ligamentous ridge located on the dorsal surface of the distal half of the bone. From this entry point, the intraosseous vessels arborize and course in a strictly retrograde fashion toward the proximal pole.
Conversely, the volar extraosseous blood supply, which accounts for the remaining 20% to 30% of the scaphoid's perfusion, arises from the superficial palmar branch of the radial artery or the radial artery proper. These volar vessels enter the scaphoid at the region of the distal tuberosity and exclusively supply the distal third of the bone. Consequently, the proximal pole is entirely devoid of direct extraosseous vascular penetrators. When a fracture occurs at the scaphoid waist or proximal third, the retrograde intraosseous vessels are sheared, instantly rendering the proximal pole ischemic. This precarious vascular watershed phenomenon dictates the high incidence of avascular necrosis and nonunion observed in proximal pole fractures.
Biomechanics and Carpal Kinematics
Biomechanically, the scaphoid functions as an oblique tie-rod that physically bridges the proximal and distal carpal rows, coordinating their complex kinematics during wrist motion. The proximal carpal row (scaphoid, lunate, triquetrum) functions as an intercalated segment with no direct tendinous insertions; its movement is entirely dictated by mechanical forces transmitted from the distal row and the surrounding capsuloligamentous constraints. The normal scaphoid naturally tends to flex under axial loading due to its articulation with the trapezium and trapezoid, while the triquetrum naturally tends to extend. The lunate, situated between them, is balanced by these opposing forces via the intact scapholunate and lunotriquetral interosseous ligaments.
In the setting of a scaphoid waist nonunion, this critical mechanical linkage is uncoupled. The distal scaphoid fragment flexes volarly, while the proximal scaphoid fragment and the lunate extend dorsally, driven by the uninhibited extension forces of the triquetrum. This uncoupling results in a characteristic "humpback" deformity of the scaphoid and a Dorsal Intercalated Segment Instability (DISI) pattern of the carpus. Failure to anatomically correct this humpback deformity during surgical reconstruction leaves the carpus in a state of chronic kinematic dysfunction, leading to altered contact stresses across the radiocarpal and midcarpal joints, ultimately culminating in rapid-onset SNAC arthropathy.
Anatomy of the Pronator Quadratus Pedicle
The pronator quadratus (PQ) pedicled bone graft relies on the robust and predictable vascular anatomy of the anterior interosseous artery (AIA). The AIA descends along the volar aspect of the interosseous membrane and terminates by providing multiple muscular branches to the deep surface of the pronator quadratus muscle. This rich intramuscular vascular plexus extends distally to supply the volar cortex and underlying cancellous bone of the distal radius via numerous small periosteal and intraosseous perforators.
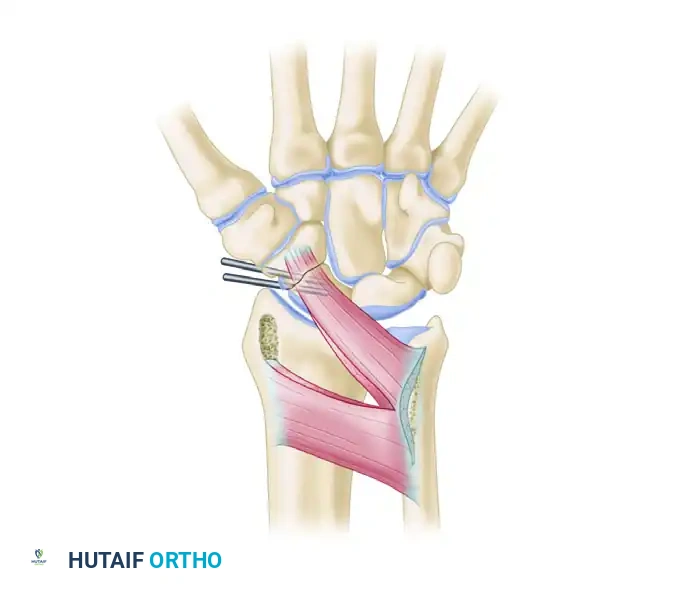
When harvesting the PQ pedicled graft, a block of corticocancellous bone is elevated from the volar distal radius along with its attached pronator quadratus muscle belly. The viability of the transferred osteocytes is entirely dependent on preserving the structural integrity of this musculoperiosteal attachment. The venous drainage of the graft is equally critical and is facilitated by the venae comitantes accompanying the AIA branches within the muscle pedicle. The arc of rotation of the PQ pedicle is generally sufficient to reach the volar aspect of the scaphoid, making it an ideal choice for addressing volar defects and humpback deformities where an anterior structural wedge is required to restore scaphoid length and correct DISI alignment.
Pathomechanics of Naviculocapitate Syndrome
Historically referred to as Fenton's syndrome, the naviculocapitate (scaphocapitate) fracture syndrome is a rare but catastrophic complex carpal injury that shares biomechanical overlap with severe scaphoid fractures. The mechanism of injury is highly specific, involving a massive axial load applied to a forcefully dorsiflexed wrist. As extreme dorsiflexion occurs, the scaphoid fractures first, typically at the waist. As the deforming force continues, the dorsal lip of the distal radius forcefully impacts the head of the capitate, creating a shearing force that fractures the capitate neck.
As the carpus is driven into further extreme dorsiflexion, the proximal head of the capitate rotates 90 degrees dorsally. When the deforming force dissipates and the hand rebounds to a neutral position, the proximal fragment of the capitate is dragged further by the intact capsular attachments, resulting in a full 180 degrees of rotation. The articular surface of the capitate head now faces distally, while the fracture surface faces proximally toward the radius. This syndrome represents a massive disruption of the carpal arcs and is frequently associated with dorsal perilunate dislocations. Understanding this extreme pathomechanics is crucial, as missed naviculocapitate injuries inevitably lead to profound midcarpal arthrosis if not acutely recognized and anatomically reconstructed.
Exhaustive Indications and Contraindications
The decision to proceed with a vascularized bone graft for a scaphoid nonunion must be based on a rigorous assessment of the patient's clinical presentation, radiographic parameters, and intraoperative findings. Vascularized grafting is a technically demanding procedure that carries inherent donor site morbidity; therefore, it should be reserved for cases where conventional non-vascularized grafting has a high probability of failure.
The primary indication for a VBG is the presence of established avascular necrosis of the proximal pole. This is typically diagnosed preoperatively using Magnetic Resonance Imaging (MRI), where a lack of gadolinium enhancement in the proximal fragment indicates profound ischemia. Intraoperatively, AVN is confirmed by the absence of punctate bleeding from the proximal pole after tourniquet deflation and meticulous debridement of sclerotic bone. Furthermore, recalcitrant nonunions that have previously failed surgical intervention (e.g., failed Herbert screw fixation with iliac crest bone grafting) are prime candidates for vascularized reconstruction, as the local biological environment has been compromised by previous scar tissue and altered vascularity.
Conversely, absolute contraindications to scaphoid reconstruction—vascularized or otherwise—include advanced carpal collapse and established arthrosis. If the patient presents with Stage II or Stage III Scaphoid Nonunion Advanced Collapse (SNAC), characterized by degenerative changes at the radioscaphoid and midcarpal (capitolunate) joints, achieving union of the scaphoid will not alleviate the patient's pain or restore normal kinematics. Active infection, such as acute or chronic osteomyelitis of the carpus, strictly precludes the use of any bone grafting technique until the infection is definitively eradicated.
| Clinical Parameter | Primary Indications for VBG | Relative Contraindications | Absolute Contraindications |
|---|---|---|---|
| Vascular Status | MRI-confirmed AVN (no gadolinium uptake); absent intraoperative punctate bleeding. | Questionable viability with massive cavitary defects (may require combined structural/VBG). | Normal vascularity (conventional non-vascularized graft preferred). |
| Fracture Location | Proximal pole fractures; proximal third waist fractures. | Distal third fractures (rarely ischemic). | N/A |
| Surgical History | Recalcitrant nonunions; failed previous internal fixation and grafting. | First-time surgery for well-aligned, non-ischemic waist nonunions. | N/A |
| Carpal Arthrosis | Minimal to no arthrosis (SNAC Stage 0 or early Stage I). | Mild radial styloid beaking (requires concurrent styloidectomy). | Established SNAC Stage II/III; midcarpal or pancarpal arthrosis. |
| Bone Stock | Sufficient proximal pole bone stock to accept fixation hardware. | Severe comminution of the proximal pole. | Complete resorption or fragmentation of proximal pole (requires free tissue transfer or salvage). |
| Infection Status | Aseptic nonunion. | History of treated superficial surgical site infection. | Active osteomyelitis; untreated deep space infection. |
Pre-Operative Planning, Templating, and Patient Positioning
Advanced Imaging and Templating
Meticulous preoperative planning is the cornerstone of successful vascularized scaphoid reconstruction. Standard posteroanterior, lateral, and scaphoid-view radiographs are initially obtained to assess the overall carpal alignment, the presence of a humpback deformity, and the extent of radiocarpal arthrosis. However, plain radiography is insufficient for three-dimensional surgical planning. High-resolution Computed Tomography (CT) scanning, reformatted along the true longitudinal axis of the scaphoid (sagittal and coronal planes relative to the scaphoid), is mandatory. The CT scan accurately quantifies the volume of bone loss, the degree of cystic resorption, and the exact angle of the humpback deformity.
Magnetic Resonance Imaging (MRI) without and with intravenous gadolinium contrast is the gold standard for evaluating the vascular status of the proximal pole. A T1-weighted hypointense signal combined with a lack of contrast enhancement on T1 fat-suppressed sequences strongly correlates with avascular necrosis. Based on the CT volumetric data, the surgeon must template the required dimensions of the bone graft. For a pronator quadratus pedicled graft, the anticipated bone block typically measures 10 to 20 mm in length and 5 to 10 mm in width. If the templated defect exceeds these dimensions, alternative vascularized grafts, such as the medial femoral condyle free flap, should be considered to provide adequate structural support.
Patient Positioning and Operating Room Setup
The patient is brought to the operating room and placed in the supine position on the operating table. The operative extremity is extended onto a radiolucent hand table, ensuring that the entire arm is supported and easily accessible for fluoroscopic imaging. A well-padded pneumatic tourniquet is applied high on the proximal arm to provide a bloodless surgical field during the critical stages of dissection and debridement.
The fluoroscopy unit (C-arm) is positioned either parallel to the hand table or coming in from the foot of the table, depending on the surgeon's preference and the specific design of the operating room. It is imperative that the C-arm can freely arc to obtain true posteroanterior and lateral views of the wrist without compromising the sterile field. The surgeon typically sits in the axilla, with the surgical assistant seated opposite. Loupe magnification (typically 3.5x to 4.5x) and a sterile headlamp are highly recommended to facilitate the meticulous identification of the vascular pedicle and the precise debridement of the nonunion site.
Anesthesia and Preoperative Optimization
Regional anesthesia, specifically an axillary or supraclavicular brachial plexus block, is the preferred modality for scaphoid reconstruction. Regional blocks not only provide excellent intraoperative anesthesia but also induce a profound sympathectomy. This sympathetic blockade causes maximal vasodilation of the peripheral vasculature, which is highly beneficial for optimizing blood flow through the delicate pedicle of the vascularized bone graft once the tourniquet is deflated.
Prior to the induction of anesthesia, a thorough review of the patient's medical history is conducted, with particular attention to smoking status. Nicotine is a potent vasoconstrictor and significantly impairs microvascular perfusion and osteogenesis. Patients must be strictly counseled on smoking cessation for a minimum of 4 to 6 weeks preoperatively and 12 weeks postoperatively. Failure to abstain from nicotine drastically increases the risk of pedicle thrombosis and subsequent failure of the vascularized bone graft.
Step-by-Step Surgical Approach and Fixation Technique
1. Surgical Approach and Exposure
The surgical approach for a pronator quadratus (PQ) pedicled bone graft requires extensive exposure of both the volar scaphoid and the distal radius. A volar zigzag (Bruner-type) or a longitudinal incision is made over the scaphoid tuberosity, extending proximally over the volar aspect of the distal radius for approximately 8 to 10 centimeters. The subcutaneous tissues are sharply divided, and meticulous hemostasis is achieved. The flexor carpi radialis (FCR) tendon sheath is incised, and the tendon is retracted ulnarly. The radial artery is identified, carefully mobilized, and retracted radially to protect it throughout the procedure.
Deep dissection reveals the volar wrist capsule and the radioscaphocapitate (RSC) ligament complex. The RSC ligament is a critical secondary stabilizer of the scaphoid and the entire carpus. It must be sharply divided to expose the underlying scaphoid nonunion. Crucial Step: The surgeon must retain robust, taggable ends of the divided RSC ligament; it must be meticulously repaired with heavy non-absorbable or slow-absorbing sutures during closure. Failure to anatomically repair the volar capsule and RSC ligament will inevitably result in severe postoperative iatrogenic carpal instability.
2. Preparation of the Nonunion Site
Once the scaphoid is exposed, the nonunion site is identified. The pseudoarthrosis is often filled with dense, avascular fibrous tissue. Using a combination of sharp curettes and a high-speed power burr under continuous cold saline irrigation, all fibrous tissue, necrotic debris, and sclerotic bone ends are meticulously excised. The debridement must continue proximally and distally until healthy, bleeding, cancellous bone is encountered. If necessary, the tourniquet can be temporarily deflated at this stage to assess for punctate bleeding in the proximal pole, confirming its vascular status.
The debridement process is designed to create an oval, trough-like cavity on the volar aspect of the scaphoid, typically measuring 10 to 20 mm in length. This cavity must be oriented parallel to the longitudinal axis of the scaphoid. The volar approach is particularly advantageous for waist nonunions with a humpback deformity, as the excavation of the volar cortex allows for the insertion of a structural wedge graft that will simultaneously restore length and correct the angular deformity.
3. Correction of Carpal Deformity (DISI)
Scaphoid waist nonunions frequently collapse into a humpback deformity, leading to Dorsal Intercalated Segment Instability (DISI). Correction of this deformity is absolutely critical before graft inset. Longitudinal traction is applied to the thumb to distract the radiocarpal joint. Simultaneously, the surgeon uses a fine periosteal elevator or inserts 0.045-inch joystick K-wires into the proximal and distal scaphoid poles.
Using the joysticks, the distal scaphoid pole is extended, and the proximal pole is flexed. This maneuver corrects the intercalated segment instability, restores the scaphoid's anatomical length and intrascaphoid angle, and opens the volar cavity to allow the vascularized bone graft to be inserted snugly. The corrected alignment is temporarily pinned with a fine K-wire and verified under orthogonal fluoroscopy before proceeding to graft harvest.
4. Graft Harvest and Pedicle Mobilization
Attention is then turned to the proximal extent of the incision to harvest the pronator quadratus graft. The PQ muscle is identified on the volar aspect of the distal radius. A block of corticocancellous bone measuring 15 to 20 mm in length and 5 to 10 mm in width is outlined at the distal insertion of the PQ, in close proximity to the abductor pollicis longus (APL) tendon.
Surgical Tip: To prevent catastrophic propagation of a cortical fracture into the radiocarpal joint, outline the margin of the intended graft by drilling multiple contiguous holes with a 0.035-inch K-wire. This creates a precise perforation line. A sharp, curved osteotome is then used to gently elevate the bone block. Surgical Warning: The surgeon must ensure that the pronator quadratus muscle fibers are not detached from the harvested bone graft. The viability of the graft depends entirely on this musculoperiosteal attachment.
The PQ muscle is then dissected proximally and toward the ulna to secure a robust muscle pedicle approximately 20 mm thick. The main anterior interosseous vessels do not need to be formally skeletonized; the microvascular network within the muscle belly is sufficient. If the pedicle lacks adequate excursion to reach the scaphoid tension-free, the ulnar origin of the PQ can be subperiosteally released from the ulna, sometimes requiring a small accessory ulnar incision.
5. Graft Inset and Rigid Fixation
Prior to placing the vascularized graft, pure autologous cancellous bone (harvested from the distal radius metaphysis or iliac crest) is tightly packed into the deepest dorsal portions of the scaphoid cavity to maximize osteoconduction and fill any dead space. The vascularized PQ bone block is then rotated distally and inserted snugly into the volar window of the scaphoid. The cortical surface of the graft is oriented volarly to act as a structural strut, maintaining the correction of the humpback deformity.
Rigid internal fixation is achieved using either multiple Kirschner wires or a headless compression screw. If K-wires are chosen, two 0.045-inch (1.16-mm) wires are driven across the nonunion site, ensuring they capture the proximal pole, the interposed vascularized graft, and the distal pole.

The trajectory and depth of the hardware must be meticulously verified under multiplanar fluoroscopy to ensure that neither the radiocarpal nor the midcarpal articular surfaces are violated. If K-wires are used, they are typically cut beneath the skin to prevent pin-tract infections and allow for prolonged casting. Following fixation, the tourniquet is deflated, and active bleeding from the muscle pedicle and the margins of the bone graft is visually confirmed to ensure vascular patency.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique and advanced biological grafting, vascularized scaphoid reconstruction is associated with a specific profile of complications. The most devastating complication is persistent nonunion, which occurs in 10% to 30% of cases, heavily dependent on patient selection, smoking status, and the severity of the initial proximal pole ischemia. Failure of the graft is typically secondary to pedicle thrombosis, inadequate rigid fixation leading to micromotion, or failure to correct the underlying DISI deformity, which subjects the healing graft to excessive shear forces.
Hardware-related complications are also prevalent. When Kirschner wires are utilized, migration or breakage can occur, necessitating premature removal and potentially compromising the stability of the reconstruction. If headless compression screws are used, inaccurate sizing or placement can lead to articular penetration, causing rapid iatrogenic chondrolysis of the radioscaphoid or capitolunate joints. Furthermore, failure to meticulously repair the radioscaphocapitate ligament during the surgical closure can lead to chronic volar carpal instability and progressive ulnar translation of the carpus.
When a vascularized bone graft fails, or if the patient presents with advanced, uncorrectable radiocarpal traumatic arthritis (SNAC wrist), further attempts at biological reconstruction of the scaphoid are universally contraindicated. In these advanced salvage scenarios, the surgical goal shifts from restoring normal anatomy to providing a stable, pain-free, and functional wrist through arthrodesis or excisional arthroplasty.
| Complication / Pathology | Estimated Incidence | Salvage Management Strategy |
|---|---|---|
| Persistent Nonunion (Aseptic) | 10% - 30% | Revision with Microvascular Free Flap (MFC) if no arthrosis; otherwise, salvage procedure. |
| Hardware Prominence/Migration | 5% - 15% | Hardware removal once union is achieved; immediate removal if articular penetration occurs. |
| Iatrogenic Carpal Instability | 2% - 5% | Capsuloligamentous repair/reconstruction; partial wrist fusion if chronic. |
| SNAC Stage I (Radial Styloid Arthrosis) | Variable | Radial Styloidectomy (often performed concurrently with VBG if mild). |
| SNAC Stage II/III (Midcarpal Arthrosis) | Progression over time | Proximal Row Carpectomy (PRC) OR Scaphoid Excision and Four-Corner Fusion. |
| Pan-Carpal Arthrosis (SNAC Stage IV) | End-stage | Total Wrist Arthrodesis (TWA) using a dorsal spanning plate. |
| Naviculocapitate Syndrome (Necrotic Capitate) | Rare (High-energy) | Primary excision of the proximal capitate pole; midcarpal fusion if severely symptomatic. |
Salvage of Naviculocapitate Syndrome
In the context of complex carpal trauma, the naviculocapitate fracture syndrome presents a unique salvage challenge. Conservative management is universally destined to fail due to the severe displacement and 180-degree rotation of the capitate head, which rapidly leads to avascular necrosis and catastrophic midcarpal arthrosis. Urgent open reduction is mandatory. The surgeon must carefully expose the midcarpal joint, identify the inverted proximal capitate fragment, and manually derotate it back into its anatomical position within the lunate fossa. Rigid internal fixation with headless screws or K-wires is performed. However, in cases of delayed presentation where the capitate head is entirely devoid of soft tissue attachments and grossly necrotic, primary excision of the proximal capitate pole may be required, eventually necessitating a midcarpal fusion to stabilize the central column.
Phased Post-Operative Rehabilitation Protocols
Strict adherence to a phased, highly controlled postoperative rehabilitation protocol is critical to protect the delicate vascular pedicle, maintain the integrity of the internal fixation, and ensure successful bony union. The rehabilitation timeline is dictated by the biological reality of bone healing and the mechanical vulnerability of the reconstructed scaphoid during the early phases of incorporation.
Phase I: Immediate Postoperative Immobilization (Weeks 0 - 2)
Immediately following the surgical procedure, the operative extremity is immobilized in a rigid, well-padded long-arm thumb spica splint. The positioning of the limb is crucial: the forearm must be placed in neutral to slight supination. Supination is specifically chosen to relax the pronator quadratus muscle and its vascular pedicle, preventing any stretching or kinking of the anterior interosseous vessels. The wrist is positioned in neutral extension, and the thumb is immobilized in palmar abduction to neutralize the deforming forces of the abductor pollicis brevis and longus. Strict elevation and active digital range of motion (fingers only) are encouraged to minimize postoperative edema and prevent intrinsic contractures.
Phase II: Intermediate Casting (Weeks 2 - 8)
At the two-week postoperative mark, the patient is seen in the clinic for the first wound check. The surgical sutures are removed, and the incision is assessed for any signs of superficial infection or delayed healing. Provided the wound is stable, the patient is transitioned into a custom-molded, fiberglass long-arm thumb spica cast. The forearm remains in neutral/supination. This rigid immobilization is maintained continuously. The patient is instructed to strictly avoid any lifting, pushing, or pulling with the operative extremity. Cardiovascular exercise is permitted provided it does not involve the upper extremities.
Phase III: Radiographic Assessment and Mobilization (Weeks 8 - 12)
At approximately eight weeks postoperatively, the cast is removed, and a clinical and radiographic evaluation is performed. Standard radiographs are often insufficient to definitively confirm union across the complex geometry of the scaphoid and the graft interfaces. Therefore, a fine-cut (1 mm slice thickness) Computed Tomography (CT) scan reformatted in the sagittal and coronal planes of the scaphoid is highly recommended. The surgeon evaluates the CT scan for the presence of bridging trabeculae across at least 50% of the cross-sectional area of both the proximal and distal graft
