Masterclass: Dorsal Vascularized Bone Graft for Scaphoid Nonunion
Key Takeaway
This masterclass guides orthopaedic fellows through the intricate dorsal vascularized bone graft procedure for scaphoid nonunions. We cover essential anatomy, preoperative planning, meticulous intraoperative steps from incision to fixation, and critical pearls to ensure successful outcomes. Learn to manage common pitfalls and optimize postoperative rehabilitation for this challenging wrist pathology.
Comprehensive Introduction and Patho-Epidemiology
The Burden of Scaphoid Nonunion
Scaphoid fractures represent the most frequently encountered carpal bone fractures in clinical orthopedic practice, accounting for approximately 60% to 70% of all carpal fractures. Despite advancements in diagnostic modalities and internal fixation techniques, scaphoid nonunion remains a formidable clinical challenge, occurring in up to 15% of all scaphoid fractures. This high rate of nonunion is multifactorial, frequently stemming from delayed initial clinical presentation, inadequate or abbreviated periods of immobilization, subtle fracture displacement unrecognized on initial radiographs, or involvement of the proximal pole. The proximal pole is particularly susceptible due to its precarious retrograde blood supply, which predisposes the fragment to ischemia and subsequent avascular necrosis (AVN).
The natural history of an untreated scaphoid nonunion is predictable and devastating to wrist biomechanics. The scaphoid acts as a critical stabilizing link between the proximal and distal carpal rows. When its structural integrity is compromised by nonunion, the wrist undergoes a predictable sequence of kinematic alterations. The distal scaphoid fragment tends to flex along with the distal carpal row, while the proximal fragment extends with the lunate, leading to a dorsal intercalated segment instability (DISI) deformity. Over time, this uncoupled motion alters the contact mechanics of the radiocarpal and midcarpal joints, inevitably culminating in Scaphoid Nonunion Advanced Collapse (SNAC). This degenerative cascade inexorably leads to debilitating osteoarthritis, profound weakness, and severe restriction of motion, severely impacting the patient's functional independence and quality of life.
Addressing a scaphoid nonunion is not merely an exercise in achieving radiographic osseous union; it is a complex reconstructive endeavor aimed at restoring carpal kinematics, preventing the progression of the SNAC sequence, and alleviating chronic pain. Traditional non-vascularized bone grafting techniques, such as the Matti-Russe procedure, have historically yielded acceptable results for scaphoid waist nonunions with adequate vascularity. However, in the setting of proximal pole ischemia, avascular necrosis, or longstanding nonunions with significant fibrous interposition and sclerosis, non-vascularized grafts frequently fail. In these high-risk scenarios, the dorsal vascularized bone graft, specifically utilizing the 1,2-intercompartmental supraretinacular artery (1,2 IC SRA), has emerged as an indispensable tool in the hand surgeon's armamentarium, providing both structural osteoconduction and immediate osteogenic vascularity.
Pathogenesis of Avascular Necrosis and Nonunion
The pathogenesis of scaphoid nonunion is intrinsically linked to the unique intraosseous vascular anatomy of the bone. When a fracture transects the scaphoid waist or proximal pole, the retrograde blood supply to the proximal fragment is frequently disrupted. Without adequate perfusion, the normal cascade of secondary bone healing—hematoma formation, inflammatory response, soft callus formation, hard callus formation, and remodeling—is arrested. The hypoxic environment at the fracture site favors the differentiation of mesenchymal stem cells into fibroblasts rather than osteoblasts, resulting in the formation of a dense, avascular fibrous nonunion rather than a robust osseous bridge.
In cases where the ischemia is profound and sustained, the proximal pole undergoes avascular necrosis. AVN is characterized histologically by osteocyte death, empty lacunae, and microtrabecular collapse. Macroscopically, the bone becomes dense, sclerotic, and fragmented. The presence of AVN drastically alters the mechanical and biological environment of the nonunion site. The necrotic bone cannot participate in the remodeling process and acts as a mechanical barrier to creeping substitution from a non-vascularized graft. The structural collapse of the necrotic proximal pole further exacerbates the carpal instability, driving the progression of the DISI deformity and the "humpback" scaphoid morphology.
The introduction of a vascularized bone graft directly addresses the biological deficit inherent in scaphoid nonunion with AVN. By transferring a corticocancellous block of bone along with its dedicated arterial pedicle and venous drainage, the surgeon bypasses the slow and unpredictable process of creeping substitution. The vascularized graft introduces living osteocytes, osteoblasts, and a functional microcirculation directly into the ischemic bed. This immediate restoration of blood flow not only facilitates the incorporation of the graft but also actively promotes the revascularization and revitalization of the native necrotic proximal pole, significantly enhancing the probability of achieving a solid, functional union.
Clinical Presentation and Diagnostic Evaluation
Patients presenting with an established scaphoid nonunion frequently report a remote history of wrist trauma, often dismissed at the time as a simple "sprain" that failed to resolve. The clinical presentation is typically characterized by a gradual, insidious onset of deep, aching wrist pain that is exacerbated by grip-intensive activities, weight-bearing across the extended wrist (such as pushing up from a chair), and extremes of motion. As the nonunion persists and carpal kinematics deteriorate, patients will progressively notice a significant reduction in their grip strength and a measurable loss of wrist range of motion, particularly in extension and radial deviation.
A meticulous physical examination is paramount for establishing the diagnosis and assessing the extent of carpal pathology. The hallmark of a scaphoid nonunion is localized tenderness to deep palpation within the anatomic snuffbox, a region bordered by the extensor pollicis longus dorsally and the extensor pollicis brevis and abductor pollicis longus volarly.
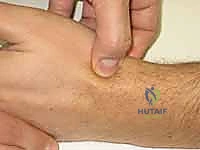
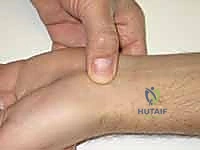
Furthermore, the examiner must systematically palpate the radial styloid-scaphoid articulation. Tenderness at this specific interface is often the earliest clinical indicator of developing SNAC arthropathy, signifying impingement and early cartilage wear. Palpation of the distal pole of the scaphoid, located on the volar aspect of the wrist just proximal to the base of the thumb metacarpal, will also frequently elicit sharp pain in the setting of a nonunion, as the uncoupled distal fragment moves abnormally against the surrounding soft tissues.
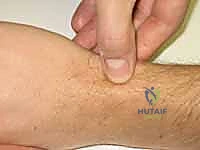
It is crucial to differentiate scaphoid nonunion from other causes of radial-sided wrist pain. The differential diagnosis includes scapholunate interosseous ligament tears, which present with tenderness specifically over the scapholunate interval; de Quervain's tenosynovitis, characterized by a positive Finkelstein's test and tenderness over the first dorsal compartment; radiocarpal or midcarpal synovitis; and intraosseous ganglia. A comprehensive clinical examination, combined with appropriate provocative maneuvers, is essential for isolating the scaphoid as the primary source of the patient's pathology before proceeding with advanced imaging.
Detailed Surgical Anatomy and Biomechanics
Osseous and Vascular Anatomy of the Scaphoid
The scaphoid is a complex, boat-shaped bone that bridges the proximal and distal carpal rows, functioning as a vital mechanical strut. Its surface is predominantly covered by articular cartilage—approximately 80% of its total surface area—which leaves very little extra-articular bone available for the ingress of nutrient vessels. This anatomical peculiarity is the primary reason for the scaphoid's tenuous blood supply. The bone is conventionally divided into three anatomic zones: the distal pole, the waist (the narrowest central portion), and the proximal pole. The waist is the most frequent site of fracture, acting as a stress riser during axial loading of the extended wrist.
The vascular anatomy of the scaphoid has been extensively mapped through microangiographic studies, most notably by Gelberman and Menen. The primary arterial supply is derived from the radial artery. The major blood supply enters the scaphoid through a narrow non-articular ridge located on the dorsal surface, at or just distal to the scaphoid waist. These intraosseous vessels then branch and arborize, flowing in a predominantly retrograde (distal-to-proximal) direction to perfuse the proximal pole. A secondary, smaller vascular contribution enters the volar aspect of the distal pole, supplying only the distal 20% to 30% of the bone.
Because of this distal-to-proximal intraosseous arborization, any fracture occurring at the scaphoid waist or proximal pole effectively severs the blood supply to the proximal fragment. Anatomical variations further complicate this issue; studies indicate that up to 30% of the population possesses either a single, tenuous intraosseous vessel or virtually no significant vascular channel reaching the proximal pole. In these individuals, even a non-displaced proximal pole fracture carries an exceptionally high risk of progressing to avascular necrosis and subsequent nonunion, making the understanding of this regional anatomy critical for the reconstructive surgeon.
The 1,2 Intercompartmental Supraretinacular Artery Pedicle
The successful execution of a dorsal vascularized bone graft relies entirely on a profound understanding of the vascular anatomy of the distal radius. The workhorse pedicle for addressing scaphoid nonunions via a dorsal approach is the 1,2-intercompartmental supraretinacular artery (1,2 IC SRA). This vessel is a consistent branch of the radial artery, originating approximately 5 centimeters proximal to the radiocarpal joint. It courses distally and dorsally, traversing the surface of the extensor retinaculum precisely within the anatomical valley between the first dorsal compartment (containing the APL and EPB) and the second dorsal compartment (containing the ECRL and ECRB).
The 1,2 IC SRA is an ideal pedicle for several reasons. First, its anatomical location is highly predictable, making surgical identification and harvest reliable. Second, the artery gives off numerous fine, nutrient branches that penetrate the underlying dorsal cortex of the distal radius. By harvesting a block of bone centered over these penetrating vessels, the surgeon captures a robust, corticocancellous graft with an intact microcirculation. Third, the pedicle possesses sufficient length and mobility to allow the harvested bone graft to be transposed distally into the scaphoid defect without placing undue tension on the delicate vascular anastomosis.
It is imperative to recognize that the 1,2 IC SRA is a superficial structure, lying directly upon the extensor retinaculum. It does not lie deep to the retinaculum or within the extensor compartments themselves. This superficial location makes the vessel highly vulnerable to iatrogenic injury during the initial surgical exposure. Furthermore, the venous drainage of the graft relies on the venae comitantes that run parallel and intimately associated with the artery. Therefore, a meticulous, atraumatic dissection technique is required to harvest the pedicle along with a generous cuff of surrounding perivascular retinacular tissue, ensuring the preservation of both arterial inflow and venous outflow.
Carpal Biomechanics in the Setting of Nonunion
To fully appreciate the goals of scaphoid reconstruction, one must understand the biomechanical consequences of its failure. In a normal wrist, the scaphoid acts as an oblique mechanical link, synchronizing the motion of the proximal and distal carpal rows. The proximal row (scaphoid, lunate, triquetrum) has no direct tendinous insertions; its movement is entirely passive, dictated by the geometry of the articular surfaces and the constraints of the intrinsic and extrinsic carpal ligaments. The scaphoid, due to its oblique orientation, naturally tends to flex under axial load, while the triquetrum tends to extend. The lunate, situated between them, is balanced by these opposing forces via the intact scapholunate and lunotriquetral ligaments.
When the scaphoid fractures and progresses to nonunion, this delicate biomechanical balance is shattered. The mechanical continuity of the scaphoid is lost, uncoupling the proximal and distal fragments. The distal scaphoid fragment, driven by the trapezium and trapezoid, flexes volarly. The proximal scaphoid fragment, still attached to the lunate via the intact scapholunate interosseous ligament, extends dorsally, taking the lunate with it. This divergent motion creates the classic dorsal intercalated segment instability (DISI) pattern. As the distal fragment flexes and the proximal fragment extends, a dorsal angular deformity develops at the nonunion site, clinically referred to as a "humpback" deformity.
The development of a humpback deformity has profound implications for radiocarpal contact mechanics. The flexed distal pole of the scaphoid impinges against the radial styloid during radial deviation, initiating the wear pattern characteristic of Stage I SNAC wrist. Furthermore, the overall shortening of the scaphoid alters the load transmission across the entire radiocarpal joint, leading to progressive cartilage degradation in the scaphoid fossa (Stage II SNAC) and eventually the midcarpal joint (Stage III SNAC). Therefore, the surgical intervention must not only achieve bony union but must also correct the humpback deformity, restoring the scaphoid to its anatomical length and alignment to normalize carpal kinematics and halt the progression of degenerative arthritis.
Exhaustive Indications and Contraindications
Patient Selection Criteria
The decision to proceed with a dorsal vascularized bone graft utilizing the 1,2 IC SRA must be predicated on a rigorous evaluation of the patient's clinical presentation, anatomical pathology, and functional demands. The primary indication for this procedure is an established scaphoid nonunion, particularly those involving the proximal pole or the proximal aspect of the scaphoid waist. Because these regions are notoriously prone to ischemia, the introduction of a vascularized graft significantly improves the biological milieu, offering a superior union rate compared to conventional non-vascularized techniques.
A critical indication is the presence of avascular necrosis (AVN) of the proximal pole. While the definitive diagnosis of AVN can be challenging, radiographic sclerosis, fragmentation, and the absence of punctate bleeding during intraoperative debridement are strong indicators. In the setting of true AVN, non-vascularized grafts act merely as inert spacers and frequently fail to incorporate. The vascularized graft, by contrast, provides immediate perfusion, facilitating the revitalization of the necrotic fragment. Furthermore, patients who have previously failed conventional bone grafting procedures (recalcitrant nonunions) are excellent candidates for a vascularized approach, as the local biological environment has already proven inadequate for standard healing.
However, the surgeon must carefully assess the geometry of the nonunion. While the dorsal 1,2 IC SRA graft is highly versatile, it possesses limitations regarding pedicle length and the ability to correct severe volar angular deformities. If the patient presents with a profound "humpback" deformity requiring a massive structural graft to restore scaphoid length and correct the DISI posture, a dorsal approach may be biomechanically challenging. In such instances, a volar approach utilizing a volar vascularized graft (e.g., from the volar distal radius) or a combination of a structural non-vascularized iliac crest graft supplemented by a vascularized pedicle may be more appropriate to achieve the necessary mechanical correction.
Table of Indications and Contraindications
| Category | Specific Clinical Scenarios | Rationale / Clinical Context |
|---|---|---|
| Primary Indications | Proximal pole scaphoid nonunion | High risk of ischemia necessitates vascularized tissue for healing. |
| Scaphoid waist nonunion with AVN | Necrotic proximal fragment requires immediate revascularization. | |
| Recalcitrant scaphoid nonunion | Failure of previous non-vascularized grafting indicates a compromised biological bed. | |
| Longstanding nonunion (>6 months) | Extensive sclerosis and fibrous interposition require robust biological stimulation. | |
| Relative Contraindications | Severe "Humpback" Deformity (Significant DISI) | Dorsal pedicle length may be insufficient to reach a volarly opened defect; volar approach preferred. |
| Active tobacco use / Nicotine dependence | Microvascular compromise significantly increases the risk of pedicle thrombosis and graft failure. | |
| Advanced patient age with low functional demand | The complexity and prolonged recovery of the procedure may outweigh the functional benefits. | |
| Absolute Contraindications | SNAC Wrist Stage II or III | Advanced midcarpal or extensive radiocarpal arthritis cannot be reversed by achieving scaphoid union. |
| Scapholunate Advanced Collapse (SLAC) | Primary ligamentous incompetence requires different reconstructive strategies (e.g., PRC, 4-corner fusion). | |
| Active local or systemic infection | High risk of graft infection, osteomyelitis, and catastrophic failure. |
Pre-Operative Planning, Templating, and Patient Positioning
Advanced Imaging and Pre-Operative Templating
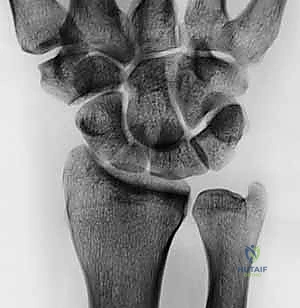
Thorough preoperative imaging is the cornerstone of successful scaphoid reconstruction. The evaluation begins with a standard radiographic series of the wrist, including high-quality posteroanterior (PA), true lateral, scaphoid (ulnar deviation), and semi-pronated oblique views. These initial radiographs provide a macroscopic assessment of the nonunion, allowing the surgeon to identify the fracture location, the degree of sclerosis, and the presence of cystic changes. Crucially, the radiographs must be scrutinized for early signs of degenerative joint disease.
Early nonunions may only show a distinct fracture line with minimal sclerosis. However, developing degenerative changes, such as subtle joint space narrowing or osteophyte formation at the radial styloid-scaphoid articulation, indicate early SNAC arthropathy and must be documented.

If advanced degenerative changes are present, involving the entire scaphoid fossa or extending into the midcarpal joint (capitolunate articulation), attempting a scaphoid reconstruction is contraindicated, as the joint mechanics are irretrievably damaged.
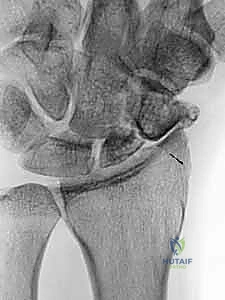
While plain radiographs are essential, Computed Tomography (CT) is the definitive modality for preoperative templating. High-resolution CT scans, reformatted in the sagittal and coronal planes along the longitudinal axis of the scaphoid, provide critical three-dimensional information. The CT scan allows the surgeon to precisely quantify the volume of the cystic voids, evaluate the preservation or loss of the normal trabecular architecture, and accurately measure the intrascaphoid angle to assess the severity of the humpback deformity. This data is used to mentally template the exact dimensions of the corticocancellous graft required to fill the defect and restore the scaphoid's native geometry. Magnetic Resonance Imaging (MRI), while useful for assessing vascularity, is often less critical than CT for structural planning, and findings of avascularity on MRI must always be correlated with intraoperative bleeding.
Operating Room Setup and Patient Positioning
Meticulous attention to the operating room setup and patient positioning is vital for the smooth execution of this complex microsurgical procedure. The patient is placed in the supine position on the operating table. The affected upper extremity is extended onto a radiolucent hand table or a specialized armboard. It is essential that the hand table is completely radiolucent and free of underlying metal supports that could obscure intraoperative fluoroscopic imaging. The arm is abducted to approximately 45 to 60 degrees and externally rotated to provide the surgeon with unencumbered, ergonomic access to the dorsoradial aspect of the wrist.
A pneumatic tourniquet is applied to the proximal brachium over a layer of soft padding. The management of the tourniquet in vascularized bone grafting differs significantly from standard orthopedic procedures. Crucially, the limb must not be exsanguinated with an Esmarch bandage prior to tourniquet inflation. Exsanguination empties the superficial venous and arterial network, rendering the delicate 1,2 IC SRA and its venae comitantes nearly invisible against the background of the extensor retinaculum. Instead, the arm is simply elevated for 60 seconds to allow for gravity drainage of capacitance vessels, and the tourniquet is then rapidly inflated to 250 mmHg. This technique traps a small amount of blood within the microvasculature, significantly enhancing the visual contrast of the pedicle and facilitating safe, accurate dissection.
The fluoroscopy unit (C-arm) must be positioned optimally before the sterile prep and drape. The C-arm should come in from the foot of the hand table or perpendicular to the patient, allowing the surgeon to obtain perfect PA, lateral, and scaphoid views by simply rotating the patient's forearm, without needing to reposition the heavy fluoroscopy machine. The monitor must be placed directly in the surgeon's line of sight. Finally, the surgical field is prepped and draped in a standard sterile fashion, ensuring that the iliac crest is also prepped and draped as a secondary site in the event that supplementary non-vascularized cancellous bone is required to support the vascularized construct.
Step-by-Step Surgical Approach and Fixation Technique
Incision, Exposure, and Pedicle Dissection
The surgical approach requires precision and a profound respect for the delicate dorsal soft tissues. With the tourniquet inflated (without prior Esmarch exsanguination), a gently curving, lazy-S or curvilinear incision is designed over the dorsoradial aspect of the wrist. The incision is centered precisely over the anatomical interval between the first and second dorsal extensor compartments, extending approximately 4 to 5 centimeters. The proximal extent reaches the distal radial metaphysis, while the distal extent curves gently toward the anatomic snuffbox and the scaphoid.
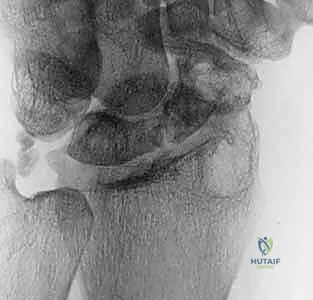
The skin and subcutaneous tissues are sharply incised. Extreme care must be taken during the subcutaneous dissection to identify, mobilize, and protect the sensory branches of the superficial radial nerve, which frequently cross the operative field. Retraction of these nerve branches should be gentle, utilizing vessel loops rather than rigid retractors to prevent neurapraxia. As the subcutaneous fat is cleared, the transverse fibers of the extensor retinaculum come into view. At this stage, the surgeon must utilize surgical loupes (minimum 2.5x to 3.5x magnification) to identify the 1,2 IC SRA. The vessel, accompanied by its paired venae comitantes, will be visible as a distinct longitudinal neurovascular bundle coursing distally on the superficial surface of the retinaculum, exactly between the first and second compartments.
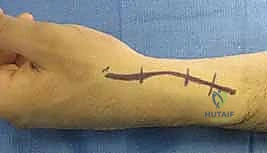
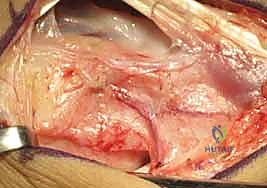
To mobilize the pedicle safely, the retinaculum must be unroofed. The critical technical pearl here is to avoid cutting directly over the artery. Instead, the retinaculum is incised longitudinally over the radial aspect of the first dorsal compartment (exposing the APL and EPB) and over the ulnar aspect of the second dorsal compartment (exposing the ECRL and ECRB). By elevating these retinacular flaps toward the midline interval, the surgeon creates a continuous, robust strip of retinaculum that contains the 1,2 IC SRA safely within its substance. This perivascular cuff of tissue protects the delicate microvasculature from sheer stress and preserves the vital venous drainage network.
Preparation of the Scaphoid Nonunion Site
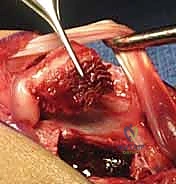
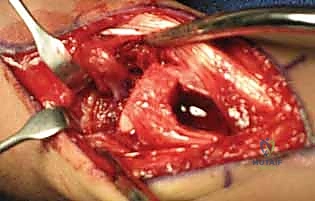
Once the pedicle is identified and safely isolated, attention is turned to the scaphoid nonunion. The capsule of the radiocarpal joint is incised longitudinally, usually in line with the interval between the third and fourth dorsal compartments, to expose the dorsal aspect of the scaphoid. The nonunion site is typically obscured by dense, hypertrophic fibrous tissue and sclerotic bone edges. Using a combination of fine rongeurs, sharp curettes, and a high-speed motorized burr (used with copious cool saline irrigation to prevent thermal necrosis), the fibrous nonunion is meticulously debrided.
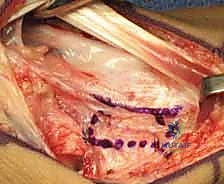
The debridement must be aggressive and uncompromising. The surgeon must excavate all necrotic, sclerotic, and fibrous tissue until healthy, punctate bleeding bone is encountered in both the proximal and distal fragments—the so-called "paprika sign." If the proximal pole is truly avascular, it may not bleed vigorously, but the sclerotic margins must still be resected to expose the underlying trabecular network, allowing the incoming vascularized graft to integrate. During this debridement, the surgeon must be careful to preserve the volar cortical hinge of the scaphoid, if intact, to maintain anatomical length and prevent destabilization of the construct. Once the defect is fully prepared, its dimensions are carefully measured using a sterile caliper to guide the subsequent graft harvest.
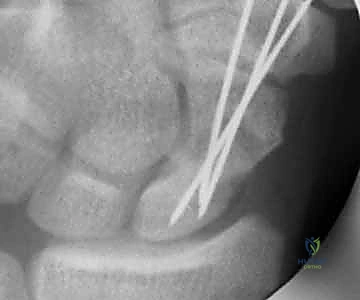
Graft Harvest and Inset
With the dimensions of the scaphoid defect known, the surgeon returns to the distal radius to harvest the graft. The previously isolated 1,2 IC SRA pedicle is traced proximally to the planned harvest site on the dorsal metaphysis of the distal radius. Using a fine sterile marking pen, a rectangular or trapezoidal cortical window is mapped out, ensuring that the distal insertion of the 1,2 IC SRA pedicle is perfectly centered within the planned bone block.
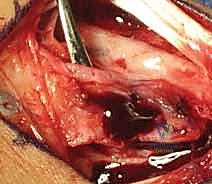
The osteotomy is performed using extremely sharp, fine osteotomes or a precise microsurgical saw. The cuts must be made with delicate, controlled taps to avoid fracturing the fragile cortical shell of the graft. The osteotomes are
Clinical & Radiographic Imaging Archive