Comprehensive Introduction and Patho-Epidemiology
Neurofibromatosis is a complex, hereditary, hamartomatous disorder derived from the neural crest, representing one of the most challenging systemic conditions encountered in orthopedic surgery. The aberrant proliferation of these hamartomatous tissues can manifest across any organ system, but the musculoskeletal manifestations demand particular surgical vigilance. Clinically, the disorder is bifurcated into two primary classifications: peripheral neurofibromatosis (NF-1) and central neurofibromatosis (NF-2).
The classic peripheral form, NF-1, historically described by Friedrich Daniel von Recklinghausen in 1882, is an autosomal dominant condition affecting approximately 1 in 3,000 to 1 in 4,000 individuals worldwide. The genetic locus for NF-1 is mapped to chromosome 17q11.2. The gene encodes for neurofibromin, a cytoplasmic GTPase-activating protein that acts as a negative regulator of the Ras signaling pathway. Pathophysiologically, loss of heterozygosity or mutation in this gene leads to unregulated Ras activity, which not only predisposes patients to developing Schwann cell tumors and distinct pigmentation abnormalities but also profoundly alters skeletal development. Recent molecular studies demonstrate that neurofibromin deficiency in mesenchymal cells leads to impaired osteoblast differentiation and hyperactive osteoclastogenesis, directly explaining the generalized osteopenia, localized bone dysplasias, and failure of fusion masses commonly seen in this patient population.
Conversely, NF-2 is a much rarer autosomal dominant disorder (1 in 40,000) mapped to chromosome 22q12.2, encoding the protein merlin. It is characterized predominantly by bilateral vestibular schwannomas (acoustic neuromas) and multiple meningiomas. NF-2 notably lacks the severe bone involvement and orthopedic manifestations seen in NF-1, making it largely a neurosurgical entity rather than an orthopedic one.
Clinical Diagnosis and Systemic Evaluation
The diagnosis of NF-1 remains primarily clinical, relying on a strict set of criteria established by the National Institutes of Health (NIH). For a definitive diagnosis, a patient must exhibit at least two of the following pathognomonic features:
* A minimum of six café au lait macules (larger than 1.5 cm in diameter in postpubertal patients, and larger than 5 mm in prepubertal patients).
* Two or more neurofibromas of any histological type, or one plexiform neurofibroma.
* Axillary or inguinal freckling (Crowe sign).
* Optic pathway glioma.
* Two or more iris Lisch nodules (identified via slit-lamp examination).
* A distinctive osseous lesion (e.g., sphenoid wing dysplasia, pseudoarthrosis of the tibia, or thinning of long bone cortex).
* A first-degree relative with a definitive diagnosis of NF-1 based on the aforementioned criteria.
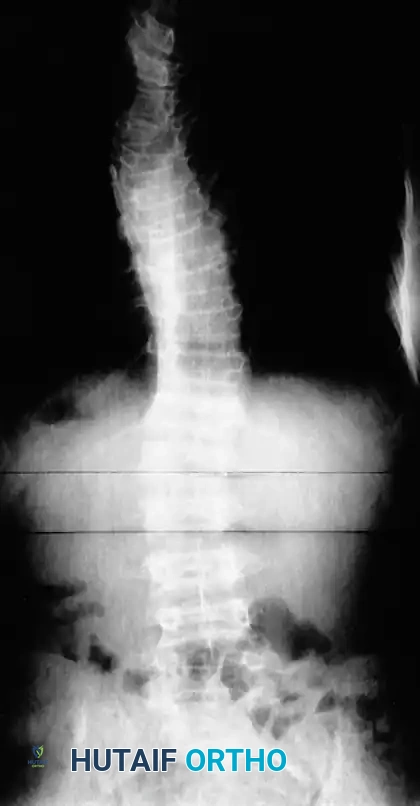
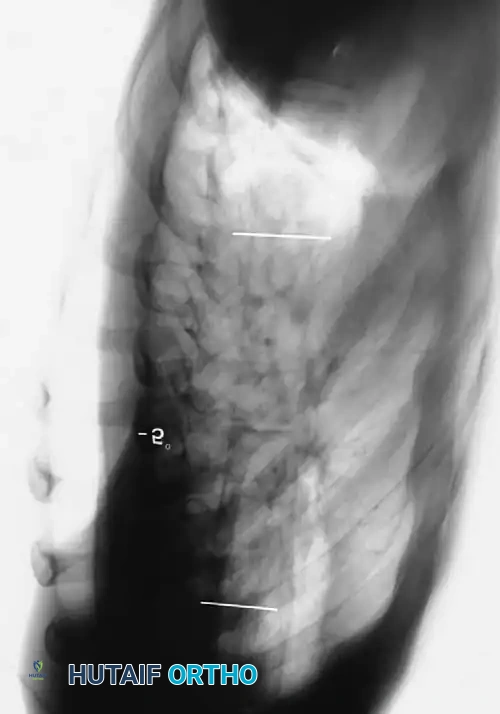
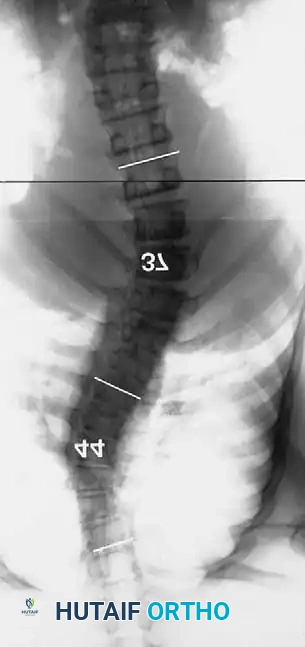
Orthopedic manifestations are profoundly common in this cohort, with spinal deformity—most notably scoliosis and kyphoscoliosis—reigning as the most frequent osseous defect. Epidemiological data from Crawford and Bagamery indicate that in a generalized pool of scoliosis patients, approximately 3% have underlying neurofibromatosis. Conversely, within a dedicated population of neurofibromatosis patients, up to 60% exhibit some form of spinal disorder. Akbarnia et al. refined this epidemiological understanding, noting structural spinal deformities in exactly 10% of an unselected cross-section of 220 NF patients, which is now widely accepted as the true baseline prevalence rate for structural curves requiring orthopedic monitoring.
Differentiating Structural from Functional Deformity
Functional scoliosis in NF-1 patients may not always be structural. Before diagnosing a primary spinal deformity, the orthopedic surgeon must rigorously evaluate the patient for leg-length discrepancies. NF-1 is notorious for causing lower extremity hypertrophy (hemihypertrophy secondary to plexiform neurofibromas or vascular anomalies) or congenital dysplasia of the long bones (such as anterolateral bowing of the tibia leading to pseudarthrosis). A thorough clinical examination utilizing block testing to level the pelvis, combined with scanogram standing radiographs, is mandatory to differentiate a compensatory functional curve from a true structural, rotational spinal deformity.
Detailed Surgical Anatomy and Biomechanics
Spinal deformities in NF-1 are biomechanically, radiographically, and prognostically divided into two distinct categories: Nondystrophic and Dystrophic. Understanding the anatomical nuances of these two classifications is the cornerstone of surgical decision-making.
Nondystrophic Scoliosis
Nondystrophic deformities behave biomechanically like typical adolescent idiopathic scoliosis (AIS). The curve patterns (predominantly right thoracic), progression risks, and responses to conservative treatment closely mirror those of idiopathic, congenital, or Scheuermann kyphosis etiologies. The vertebrae lack the intrinsic dysplastic features seen in the dystrophic variant. In these cases, the underlying NF-1 diagnosis appears to have minimal influence on the curve's natural history, with one critical, highly unpredictable exception: the phenomenon of "modulation."
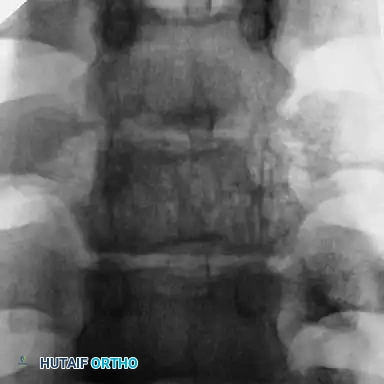
The Phenomenon of Modulation
Crawford highlighted a critical biomechanical and radiographic phenomenon termed "modulation"—the transition of a seemingly benign, nondystrophic curve into a highly aggressive dystrophic one over time. The exact trigger for this transition remains an area of active research, but it is believed to be linked to the progressive manifestation of neurofibromin deficiency in the local osseous environment during periods of rapid skeletal growth. A spinal deformity developing before 7 years of age demands hyper-vigilant observation. If a previously nondystrophic curve acquires three "penciled" ribs or a combination of three dystrophic radiographic features, rapid clinical progression and biomechanical collapse are almost a certainty.
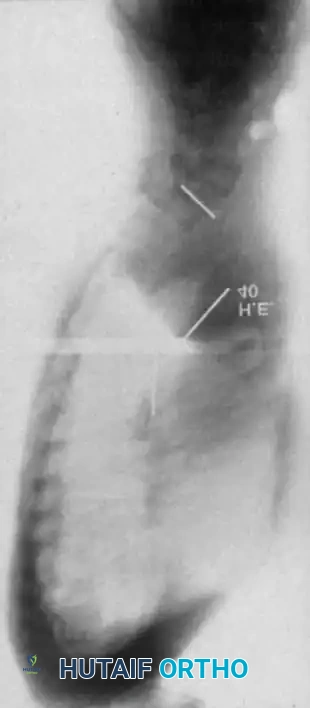
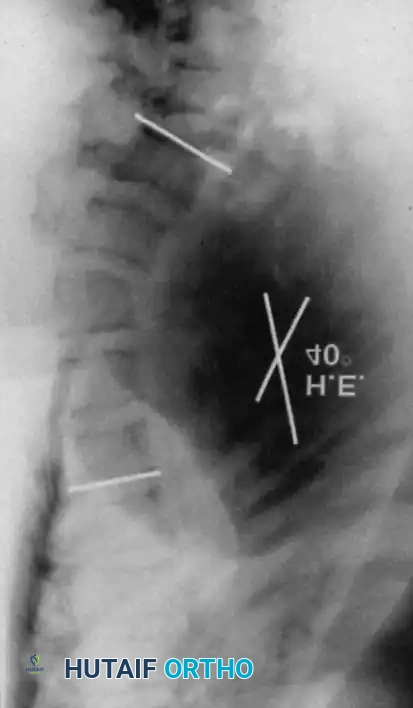
Dystrophic Scoliosis and Kyphoscoliosis
Dystrophic scoliosis is the most aggressive, unpredictable, and surgically challenging variant. It is characterized by a short-segment, sharply angulated curve, often involving fewer than six vertebrae. Radiographically and anatomically, dystrophic curves present with a constellation of severe structural anomalies that drastically alter the biomechanical load-bearing capacity of the spine:
* Severe wedging of the apical vertebral bodies: The anterior and lateral columns fail to develop symmetrically, leading to acute angular deformity.
* Extreme axial rotation: The vertebrae rotate severely into the convexity, pulling the dysplastic ribs with them.
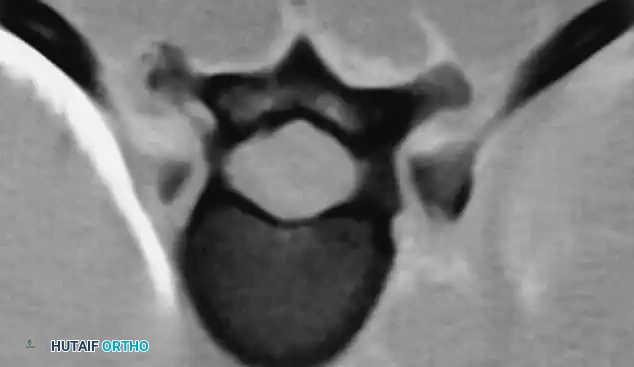
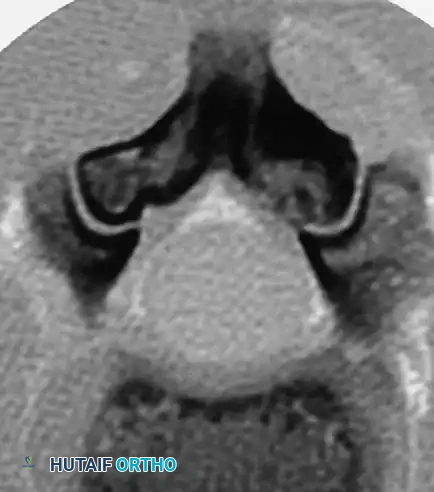
* Posterior scalloping of the vertebral bodies: This is a hallmark sign, often secondary to dural ectasia. The enlarged subarachnoid space physically erodes the posterior cortex of the vertebral body and the pedicles, leaving a paper-thin osseous shell that is highly prone to iatrogenic fracture during pedicle screw insertion.
* Spindling of the transverse processes: The transverse processes become thin, elongated, and biomechanically incompetent, making hook fixation nearly impossible.
* Enlargement of the neural foramina: Often caused by the presence of dumbbell neurofibromas extending from the spinal canal into the paraspinal musculature.
* "Penciled ribs": The ribs rotate 90 degrees in the anteroposterior direction due to the severe vertebral rotation, making them appear abnormally thin and dysplastic on an AP radiograph. They lose their structural ability to support the thoracic cage, contributing to restrictive pulmonary disease.
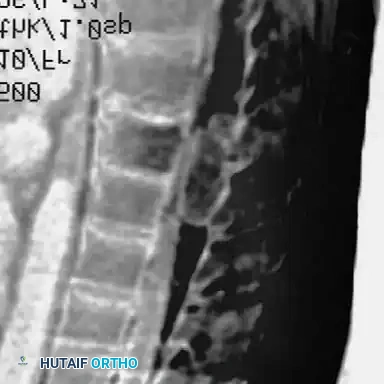
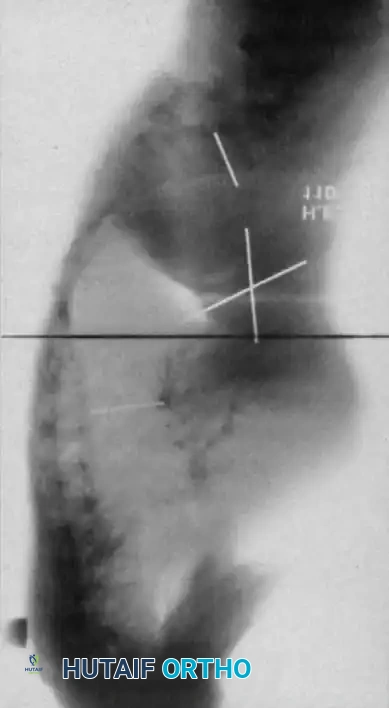
Curves with significant sagittal plane deformity are highly prevalent in dystrophic scoliosis. Neurofibromatosis kyphoscoliosis is particularly notorious, characterized by acute, rigid angulation in the sagittal plane and striking destruction or deformity of the vertebral bodies near the apex. The apical vertebrae are often so dysplastic that the anterior column is functionally absent, placing immense tension on the posterior soft tissues and rendering the spinal cord highly vulnerable to stretching across the apex of the kyphosis. Severe thoracic lordoscoliosis has also been documented by Winter, presenting unique respiratory and biomechanical challenges due to the intrusion of the spine into the thoracic cavity, drastically reducing vital capacity.
Exhaustive Indications and Contraindications
The decision matrix for surgical intervention in NF-1 spinal deformities is dictated primarily by the classification of the curve (dystrophic vs. nondystrophic), the presence of sagittal plane deformity (kyphosis), and the patient's neurological status. Observation of a progressing dystrophic spinal deformity is strictly unjustified. Because only a fraction of dystrophic curves remain small, early operative intervention is the absolute gold standard to prevent catastrophic neurological compromise and severe cardiopulmonary restriction.
Indications and Contraindications Matrix
| Clinical Scenario | Primary Indications | Contraindications |
|---|---|---|
| Nondystrophic Curve < 25° | Observation with serial standing radiographs every 6 months. | Surgical intervention. |
| Nondystrophic Curve 25° - 40° | TLSO Bracing (if growing child); closely monitor for modulation. | Bracing if patient is skeletally mature. |
| Nondystrophic Curve > 45° | Posterior Spinal Fusion (PSF) with segmental instrumentation. | Anterior-only approach (unless specific thoracolumbar curve criteria met). |
| Dystrophic Curve (Any size) | BRACING IS CONTRAINDICATED. Winter et al. reported 100% failure rate with bracing in dystrophic curves. | TLSO or Milwaukee brace. |
| Dystrophic Scoliosis < 20° | Close observation (every 3-4 months). Immediate surgery if any progression is noted. | Waiting for standard AIS surgical thresholds (e.g., 45°). |
| Dystrophic Scoliosis 20° - 40° | Early Posterior Spinal Fusion (PSF) to prevent need for anterior surgery. | Observation; Bracing. |
| Dystrophic Scoliosis > 40° | Combined Anterior and Posterior Spinal Fusion (APSF). | Posterior-only fusion (high risk of crankshaft phenomenon and pseudarthrosis). |
| Dystrophic Kyphoscoliosis > 50° | 360-degree fusion: Anterior release, corpectomy, structural strut graft, followed by robust PSF. | Posterior-only fusion (guaranteed hardware failure and kyphotic progression). |
| Presence of Dumbbell Tumor | Neurosurgical resection prior to or concurrent with orthopedic correction. | Blind correction maneuvers without addressing the intraspinal lesion (risk of paraplegia). |
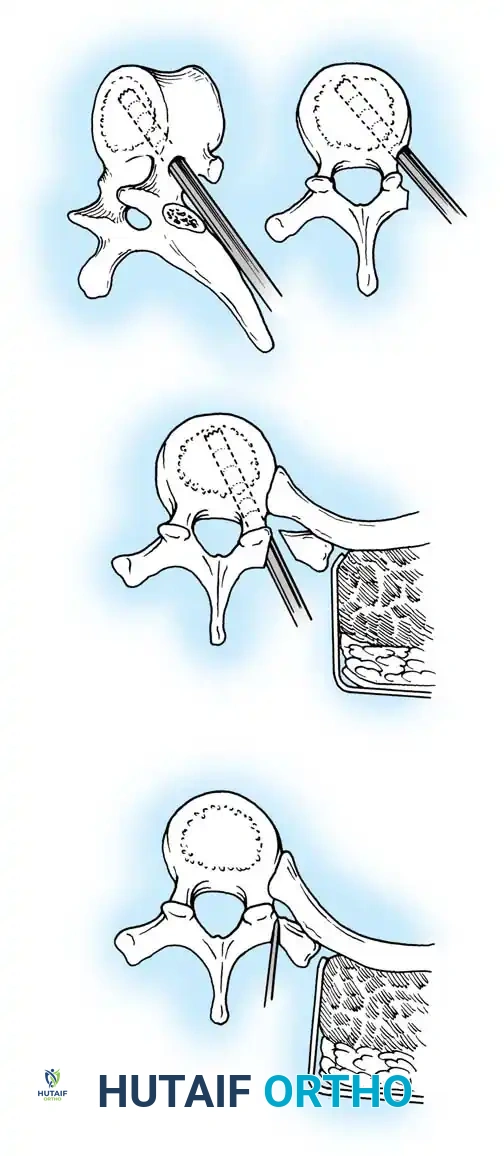
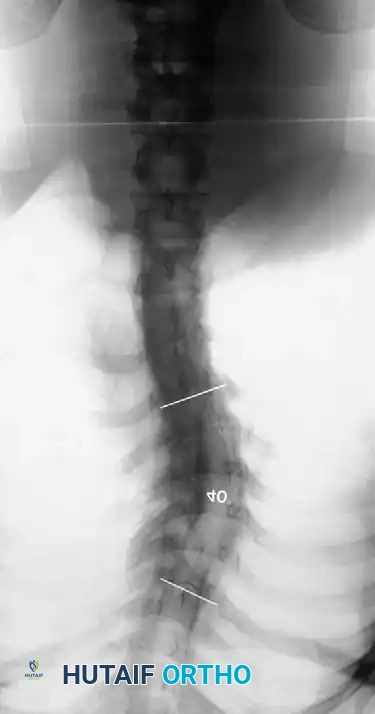
Pre-Operative Planning, Templating, and Patient Positioning
The preoperative workup for an NF-1 patient with spinal deformity must be exhaustive and uncompromising. The margin for error is exceptionally narrow due to the dysplastic bone, altered vascular anatomy, and the high prevalence of occult intraspinal lesions.
Advanced Imaging Modalities
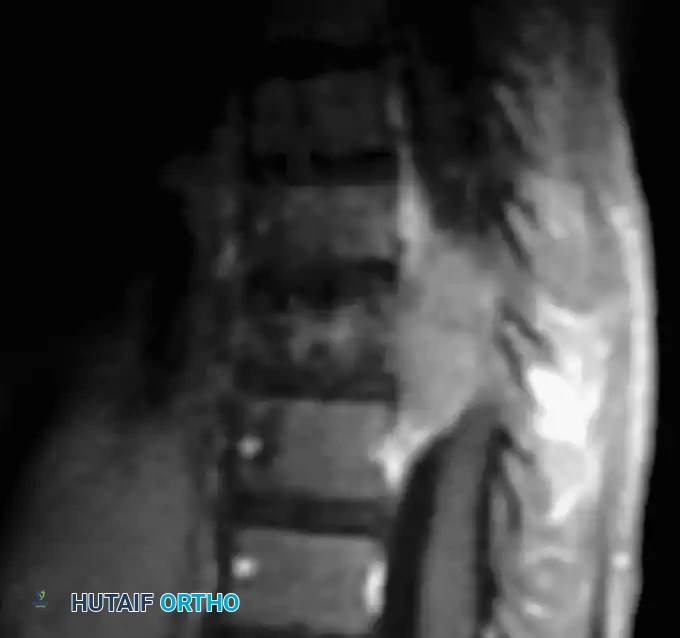
The presence of intraspinal lesions—such as pseudomeningoceles, dural ectasia, or intraspinal neurofibromas (dumbbell tumors)—must be definitively ruled out before any surgical instrumentation or correction maneuver is attempted. Impingement of these occult lesions against the spinal cord during the correction maneuvers of instrumentation has been documented to cause acute, irreversible paraplegia.
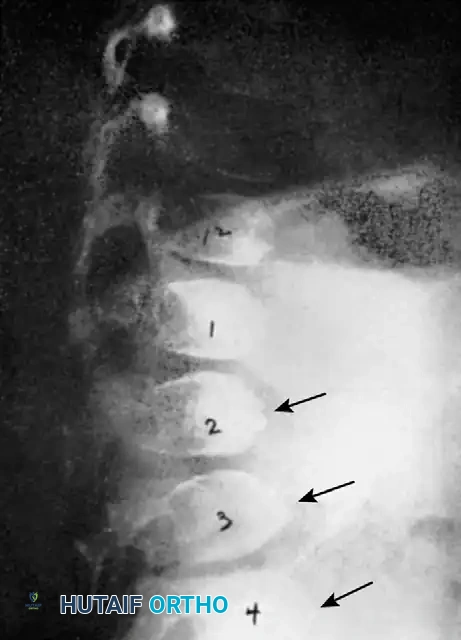
Routine total spine Magnetic Resonance Imaging (MRI) is mandatory for every NF-1 patient scheduled for spine surgery. Ramachandran et al. demonstrated that 37% of NF-1 patients (in both dystrophic and nondystrophic groups) harbored intraspinal and paraspinal neurofibromas, often adjacent to the convexity of the curve. MRI expertly delineates the extent of dural ectasia, the presence of syrinx, and the exact location of the conus medullaris, which may be tethered.
While MRI is superior for evaluating cord displacement and the subarachnoid extent of neurofibromas, severe kyphoscoliotic deformities may render MRI inadequate due to signal artifact and complex multi-planar anatomy that obscures the true canal dimensions. In such challenging cases, complete high-volume CT myelography with 3D reconstruction in the prone, lateral, and supine positions is required to accurately map the spinal canal and evaluate the osseous stock available for pedicle screw purchase.
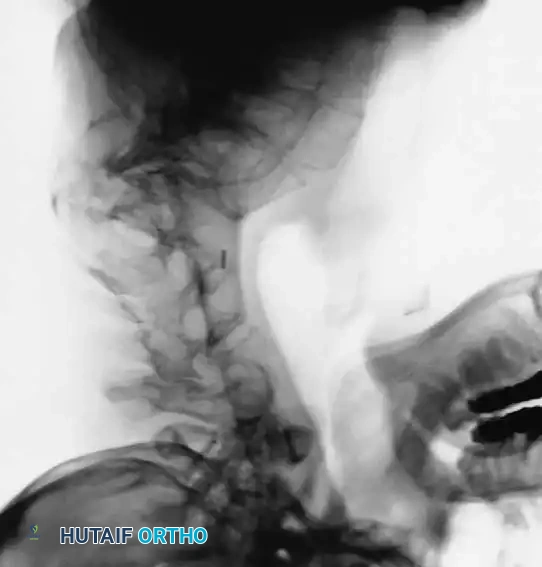
Halo-Gravity Traction (HGT)
For severe, rigid, neglected curves (typically >100 degrees of scoliosis or >90 degrees of kyphosis), preoperative Halo-Gravity Traction is an invaluable adjunct. HGT allows for gradual, awake correction of the deformity over 3 to 6 weeks. This slow stretch maximizes soft tissue compliance, improves pulmonary function prior to major thoracic surgery, and crucially allows for continuous clinical neurological monitoring. If the patient develops any cranial nerve palsies or lower extremity weakness during traction, weight is immediately reduced. HGT significantly decreases the need for aggressive, high-risk intraoperative correction maneuvers.
Patient Positioning and Neuromonitoring
Intraoperative neuromonitoring is non-negotiable. Multi-modality monitoring including Somatosensory Evoked Potentials (SSEPs), Motor Evoked Potentials (MEPs), and free-running electromyography (EMG) must be utilized. Baseline signals must be obtained prior to positioning.
Positioning for the posterior approach requires a standard Jackson spinal table to allow the abdomen to hang free, reducing epidural venous engorgement. Care must be taken to pad all bony prominences, as NF-1 patients often have fragile skin and subcutaneous neurofibromas that can be compressed, leading to necrosis. For the anterior approach, the patient is placed in the lateral decubitus position. The surgeon must be prepared for massive, atypical venous plexuses often associated with paraspinal neurofibromas, requiring meticulous hemostasis and cell-saver availability.
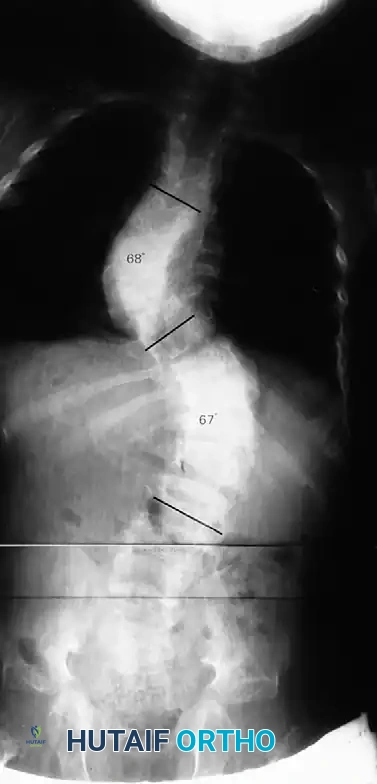
Step-by-Step Surgical Approach and Fixation Technique
The surgical strategy is dictated by the curve's classification and severity. The fundamental goal is to achieve a solid, balanced arthrodesis while protecting the neural elements.
Management of Nondystrophic Curves
Nondystrophic curves share the prognosis and evolutionary trajectory of idiopathic curves, albeit with a statistically higher risk of pseudarthrosis following operative fusion due to the underlying neurofibromin-deficient osteopenia.
If the deformity exceeds 40 to 45 degrees, posterior spinal fusion with segmental instrumentation is recommended. Modern segmental instrumentation (e.g., Cotrel-Dubousset principles) utilizing high-density pedicle screw constructs provides excellent correction. The surgical technique involves standard subperiosteal exposure, facetectomies to enhance fusion, and bilateral pedicle screw placement. Because modulation can occur postoperatively, it is highly recommended to extend the fusion construct slightly longer than one would for a standard AIS curve, ensuring the end vertebrae are neutral and stable. Copious autogenous bone grafting, often supplemented with allograft, is essential to mitigate the heightened pseudarthrosis risk.
Management of Dystrophic Scoliosis Without Kyphosis
Patients presenting with dystrophic scoliosis lacking a kyphotic component should be observed at 6-month intervals if the curve is under 20 degrees. The moment progression is documented, posterior spinal fusion must be executed.
If posterior fusion is performed early (curves 20-40 degrees), an anterior approach may be avoided. However, for neglected curves exceeding 80 degrees, Betz et al. strongly recommend a combined anterior and posterior arthrodesis. Primary anterior and posterior fusion is the standard for dystrophic scoliosis >40 degrees unless specific contraindications exist (e.g., massive anterior neurofibromas, excessive venous plexuses, severe osteopenia, or thrombocytopenia secondary to splenic fibromas).
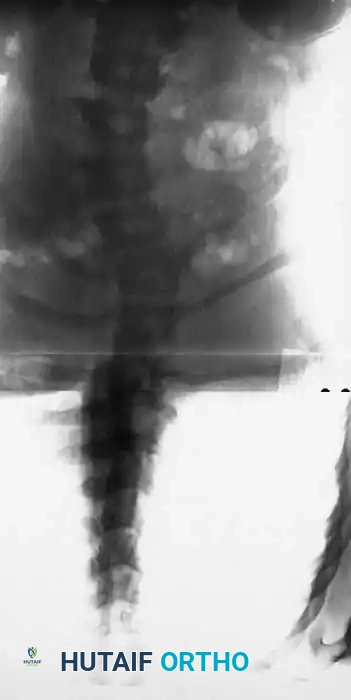
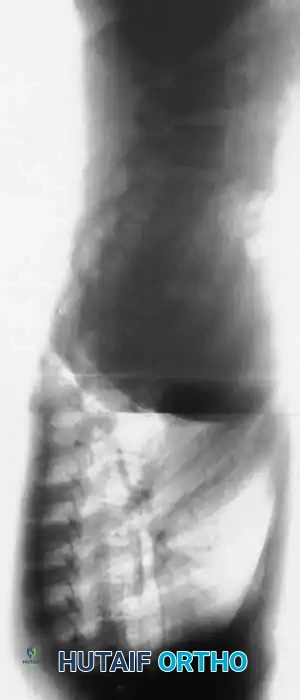
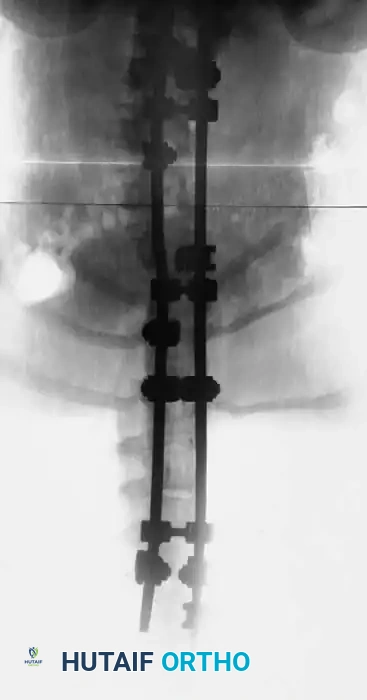
Segmental pedicle screw systems provide superior biomechanical purchase in dysplastic bone, permitting early ambulation. However, pedicle preparation in dystrophic vertebrae requires extreme caution. The pedicles are often sclerotic on the concavity and paper-thin on the convexity due to dural ectasia. Preoperative CT navigation or intraoperative robotic assistance/O-arm imaging is highly beneficial to ensure safe trajectory and avoid breaching the medial wall into the enlarged spinal canal. If the instrumentation purchase is tenuous, sublaminar wires or hooks may be utilized as salvage fixation points. Postoperative bracing is mandatory if fixation is suboptimal.
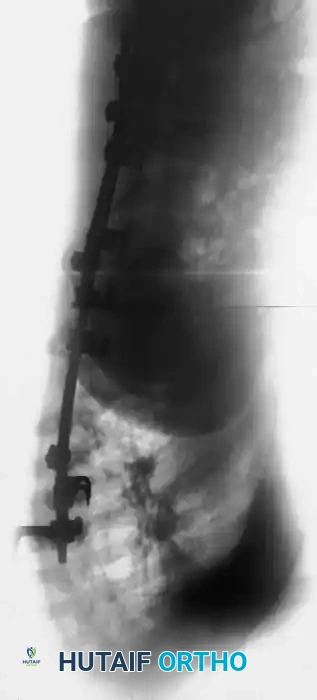
Management of Dystrophic Kyphoscoliosis
Dystrophic scoliosis coupled with angular kyphosis represents one of the most formidable challenges in spine surgery. Patients with this pathology respond exceptionally poorly to posterior fusion alone. Consistent, long-term success is achieved almost exclusively through combined anterior and posterior (360-degree) fusions.
Winter and Hsu emphasized that the primary reasons for failure in these curves are inadequate bone graft volume, insufficient fusion area, and failure to support the anterior column biomechanically. The surgical strategy must include the following meticulous steps:
- Anterior Release and Corpectomy: The patient is positioned laterally. A thoracotomy is performed on the convexity of the curve. The segmental vessels are carefully ligated. The entire structural area of the deformity must be addressed anteriorly. This requires complete, aggressive disc excision back to the posterior longitudinal ligament. In severe angular kyphosis, an anterior corpectomy of the apical dysplastic vertebrae is required to decompress the anterior aspect of the spinal cord and create a bed for the strut graft.
- Structural Strut Grafting: Strong strut grafts are biomechanically imperative to support the anterior column and prevent kyphotic collapse. Fibular allografts or robust autografts, keyed into the healthy vertebral bodies above and below the apex, are utilized. The graft must be placed under compression to ensure stability.
- Vascularized Rib Grafts: For severe, rigid kyphoscoliotic deformities with a high risk of non-union, vascularized rib graft augmentation (as described by Bradford) provides superior biological healing potential in the hostile, dysplastic environment of the NF-1 spine. The rib is harvested with its intercostal neurovascular bundle intact and swung into the anterior defect, providing immediate living bone to bridge the gap.
- Posterior Instrumentation: Following the anterior stage (either same day or staged 5-7 days later), robust posterior segmental instrumentation and arthrodesis are performed. The posterior construct acts as a tension band, compressing the anterior strut graft.
For smaller dystrophic kyphoscoliosis (kyphosis <40 degrees), posterior instrumentation and arthrodesis may be attempted early, but the fusion mass must be explored at 6 to 12 months. If kyphosis exceeds 50 degrees, the combined anterior/posterior approach is non-negotiable. The anterior fusion must extend one or two levels beyond the end vertebrae of the kyphotic segment to prevent junctional failure.
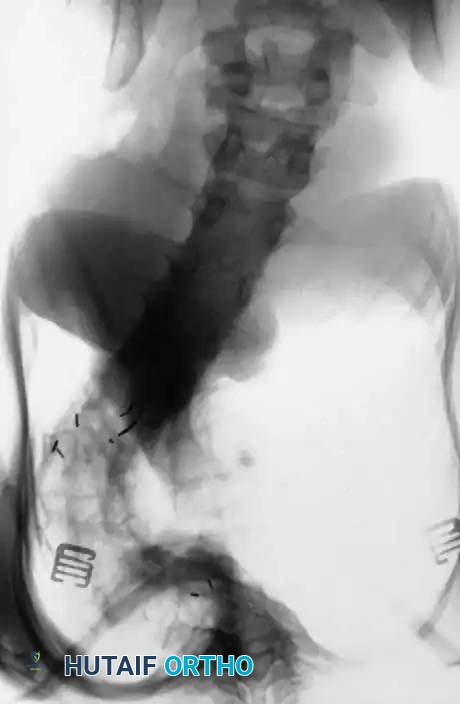
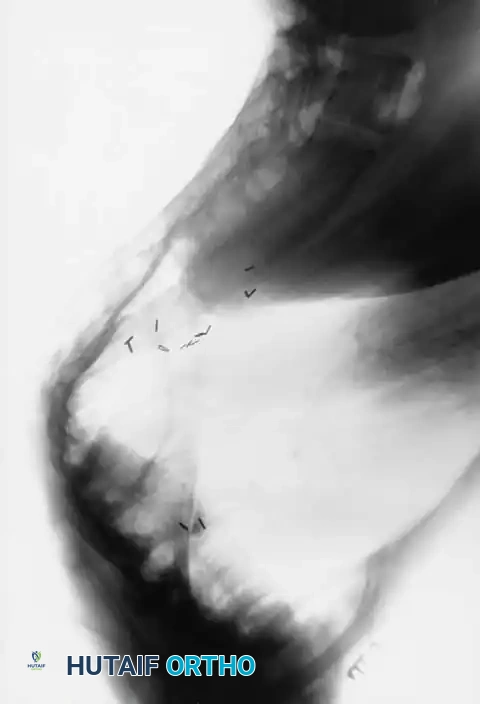
Complications, Incidence Rates, and Salvage Management
The surgical management of spinal deformities in NF-1 is fraught with complications. The intrinsic biological defect in bone healing, combined with the extreme biomechanical forces of dystrophic curves, creates a highly hostile surgical environment.
Management of Spinal Cord Compression
Neurological deficits in NF-1 patients can arise from two distinct etiologies: direct compression from an intraspinal tumor (e.g., neurofibroma) or mechanical compression from severe angular kyphosis stretching the cord over the apical vertebrae.
If spinal cord compression is caused by a kyphoscoliotic deformity, posterior laminectomy is absolutely contraindicated. Laminectomy destroys the only remaining tension band, leading to rapid, catastrophic progression of the kyphosis and worsening of the neurological deficit. The correct approach is an anterior decompression (corpectomy) to remove the offending apical bone, followed by robust anterior strut grafting and posterior stabilization.
Dural Ectasia and Dural Tears
Dural ectasia results in a thinned, friable dura that is highly susceptible to iatrogenic tearing during exposure, facetectomy, or screw placement. Primary repair of these tears is often impossible due to the tissue quality. Salvage management requires the use of synthetic dural patches, muscle-fascial flaps, and fibrin glue sealants. Postoperative subarachnoid lumbar drains may be required for 5-7 days to divert CSF and allow the repair to heal, preventing the formation of massive pseudomeningoceles.
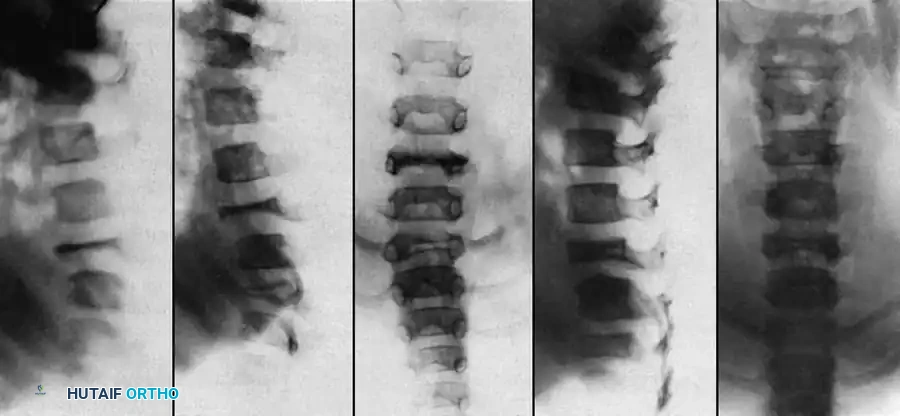
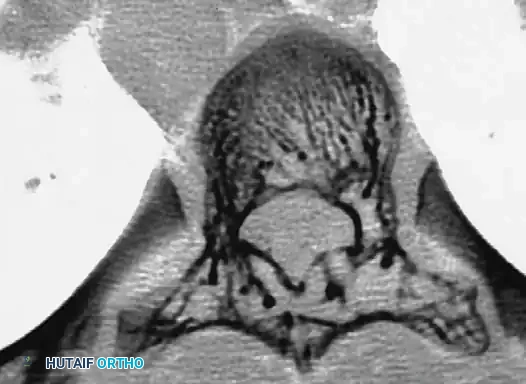
Complication Matrix
| Complication | Estimated Incidence | Pathophysiology & Risk Factors | Salvage Management |
|---|---|---|---|
| Pseudarthrosis | 15% - 35% | Impaired osteoblast function; poor vascularity; massive biomechanical stress. | Routine 1-year exploration if suspected. Augmentation with iliac crest autograft and BMP (Bone Morphogenetic Protein). |
| Implant Failure / Pullout | 10% - 20% | Severe osteopenia; dysplastic pedicles lacking cortical bone. | Extension of fusion construct; use of cement-augmented pedicle screws; combined anterior-posterior approach. |
| Neurological Deficit | 2% - 5% | Cord stretch during correction; unrecognized dumbbell tumor; epidural hematoma. | Immediate cessation of correction; release of traction; wake-up test; emergent MRI; anterior decompression if caused by kyphotic apex. |
| Dural Tear / CSF Leak | 5% - 10% | Dural ectasia causing paper-thin dura; erosion of posterior vertebral elements. | Fascial patch, fibrin glue, watertight fascial closure, lumbar subarachnoid drain for 5-7 days. |
| Junctional Kyphosis | 10% - 15% | Stopping fusion short of the neutral vertebrae; progressive dystrophic changes at adjacent levels. | Revision surgery to extend the fusion proximally or distally; ensuring anterior column support. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation of the NF-1 spine patient requires a delicate balance between early mobilization to prevent pulmonary complications and strict protection of the tenuous fusion mass.
Phase I: Immediate Post-Operative (Days 0-7)
Patients are typically monitored in the Pediatric Intensive Care Unit (PICU) or Surgical ICU for the first 24-48 hours. Strict neurological checks are performed every 2 hours. Hemodynamic stability is paramount, as blood loss from anterior approaches and dysplastic bone can be substantial. Chest tubes from the thoracotomy are managed and typically removed when output is <50cc per shift. Log-rolling is strictly enforced.
Phase II: Mobilization and Bracing (Weeks 1-6)
Unlike standard AIS patients who
Clinical & Radiographic Imaging Archive