Welcome, fellows, to the operating theater. Today, we are undertaking a complex yet profoundly rewarding procedure: segmental hook and pedicle screw instrumentation for scoliotic spinal deformity. Our overriding surgical objective is to achieve a balanced, three-dimensional correction of the spine, thereby improving the patient's cosmetic appearance, arresting relentless curve progression, and ultimately providing an unassailable biomechanical construct for solid spinal fusion. This endeavor is a journey of precision, exhaustive anatomical mastery, and meticulous, unforgiving surgical technique. Let us scrub in and dissect the nuances of this masterclass.
Comprehensive Introduction and Patho-Epidemiology
Scoliosis is fundamentally a complex, three-dimensional torsional deformity of the spine and trunk, rather than a simple lateral curvature in the coronal plane. The evolution of surgical management for this pathology represents one of the most remarkable arcs in orthopedic history. We have transitioned from the era of Harrington rods, which relied primarily on unidirectional distraction and often resulted in the dreaded "flatback" syndrome, to the advent of segmental sublaminar wiring by Luque, and subsequently to the revolutionary Cotrel-Dubousset instrumentation utilizing hooks for true derotation. Today, all-pedicle screw constructs and hybrid hook-screw systems represent the gold standard, offering unprecedented three-column control, superior curve correction, and reduced reliance on postoperative bracing.
The pathophysiology of Adolescent Idiopathic Scoliosis (AIS), the most common variant we encounter, remains frustratingly elusive despite decades of rigorous research. Current consensus points toward a multifactorial etiology involving pleiotropic genetic influences, subtle central nervous system abnormalities, and biomechanical growth modulations. The Hueter-Volkmann principle plays a critical role in the progression of the deformity; asymmetric loading across the vertebral endplates inhibits growth on the concave side while accelerating it on the convex side, thereby exacerbating the wedging of the vertebral bodies and intervertebral discs. This self-perpetuating biomechanical cycle dictates that without intervention, severe curves will inevitably worsen, leading to severe truncal asymmetry and potential cardiopulmonary compromise.
Epidemiologically, while minor spinal asymmetries are relatively common in the adolescent population, curves exceeding the surgical threshold are significantly less frequent and exhibit a striking sex predilection. The ratio of females to males with curves greater than 30 degrees approaches 10:1. The risk of progression is intimately tied to the patient's remaining growth potential, which we meticulously assess using parameters such as the Risser sign, the Sanders skeletal maturity staging system, menarchal status, and the magnitude of the curve at initial presentation. A premenarchal female with a Risser 0 and a 40-degree curve faces a near-certainty of progression to a surgical magnitude.
Our surgical goals must be clearly delineated and communicated to the patient and their family. The primary objective is the definitive arrest of curve progression through a solid osseous arthrodesis. Secondary to this, but of paramount importance to the patient, is the optimization of three-dimensional spinal balance—restoring coronal plumbline, normalizing sagittal contours (specifically thoracic kyphosis and lumbar lordosis), and minimizing axial rotation to reduce the rib hump. We must achieve these goals while maximizing neurological safety, minimizing the number of fused motion segments, and preventing catastrophic perioperative complications.
Detailed Surgical Anatomy and Biomechanics
Fellows, a superficial anatomical understanding is insufficient; a profound, three-dimensional comprehension of scoliotic spinal anatomy is the bedrock of safe instrumentation. The scoliotic spine is a severely distorted environment where normal anatomical relationships are fundamentally altered. The vertebral bodies undergo significant rotation in the transverse plane, with the spinous processes rotating toward the concavity of the curve. Consequently, relying on standard midline landmarks for pedicle screw starting points will inevitably lead to disastrous lateral breaches on the convexity and medial breaches on the concavity.
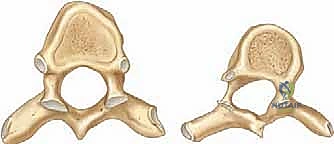
Pedicle morphology is highly asymmetrical in the scoliotic spine. The pedicles on the concave side, particularly at the periapical levels, are consistently narrower, more sclerotic, and dysmorphic compared to their convex counterparts. Furthermore, the spinal cord does not sit centrally within the canal; it migrates and hugs the concavity of the curve. The width of the epidural space is often less than 1 mm at the thoracic apical vertebral levels on the concave side, compared to 3 to 5 mm on the convex side. This drastically reduces our margin for error during instrumentation on the concave side, transforming a millimeter of deviation into a potential neurological catastrophe.
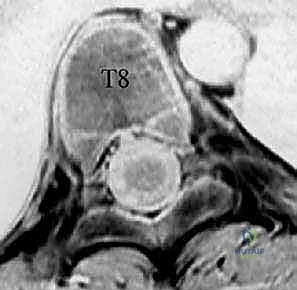
The thoracic spine presents unique biomechanical and anatomical challenges. The facets are coronally oriented, and the pedicles are inherently smaller. The average thoracic pedicle length is 16 to 22 mm, but the critical dimension is the endosteal width, which can be as narrow as 2.5 mm on the concavity. The medial pedicle wall is two to three times thicker than the lateral wall; thus, errant forces during pedicle preparation typically result in lateral breaches. Furthermore, thoracic transverse processes do not align consistently with the pedicle in the axial plane. They are rostral to the pedicle in the upper thoracic spine and caudal in the lower thoracic spine, crossing over around T6-T7. Starting points must be adjusted accordingly: moving proximally from T12 towards the apex, the starting point trends medial and cephalad, whereas from the apex proximally, it shifts lateral and caudal.
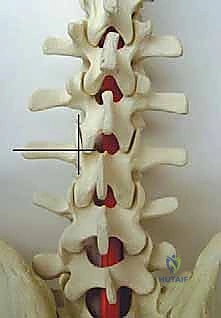
The lumbar spine, while possessing larger pedicles (average endosteal width of 4.8 to 9.5 mm) and offering a greater margin of safety, demands equal vigilance. Lumbar facets are sagittally oriented, and the pedicles angle outward progressively from L1 to L5. The standard starting point lies at the intersection of a vertical line tangential to the lateral border of the superior articular process and a horizontal line bisecting the transverse process. However, neurovascular dangers remain omnipresent. Inferior pedicular breaches in the lumbar spine directly endanger the exiting nerve root within the neural foramen. Lateral breaches in the thoracic spine endanger the parietal pleura, lungs, segmental vessels, and the sympathetic chain. Critically, from T5-T10, the aorta frequently shifts posterolaterally, resting directly against the left lateral vertebral body and pedicle, making a left-sided lateral breach potentially fatal.
Exhaustive Indications and Contraindications
The decision to proceed with surgical intervention in spinal deformity is a complex calculus weighing the natural history of the specific curve against the inherent risks of major spinal reconstruction. In Adolescent Idiopathic Scoliosis, the universally accepted indication for surgery is a curve magnitude exceeding 45 to 50 degrees in a growing child, or a curve exceeding 50 degrees in a skeletally mature patient. Curves of this magnitude have been shown in long-term natural history studies to progress at a rate of 1 to 2 degrees per year even after skeletal maturity, leading to progressive truncal decompensation, chronic back pain, and in severe thoracic curves (>90 degrees), restrictive pulmonary disease and cor pulmonale.
In Adult Degenerative Scoliosis, the indications are primarily driven by symptomatology rather than pure curve magnitude. Surgery is indicated for patients with severe, recalcitrant mechanical back pain, neurogenic claudication, or radiculopathy secondary to asymmetric disc collapse and foraminal stenosis that has failed exhaustive conservative management (including physical therapy, NSAIDs, and epidural steroid injections). Furthermore, documented progressive coronal or sagittal decompensation, resulting in an inability to maintain a horizontal gaze or stand upright without external support, is a strong indication for surgical realignment and stabilization.
Contraindications must be rigorously respected to prevent disastrous outcomes. Absolute contraindications include active systemic or local spinal infections, which must be eradicated prior to any instrumentation. Severe medical comorbidities, such as unstable ischemic heart disease, severe restrictive lung disease precluding safe extubation, or uncorrectable coagulopathies, render the perioperative risk unacceptably high. Relative contraindications include profound osteoporosis, which severely compromises pedicle screw purchase and increases the risk of proximal junctional kyphosis (PJK) or hardware pullout. In such cases, surgical strategies must be modified to include cement augmentation of screws, increased points of fixation, or multiple-rod constructs.
The table below summarizes the critical indications and contraindications for segmental instrumentation in scoliotic deformity.
| Category | Specific Criteria / Conditions | Clinical Implications |
|---|---|---|
| Absolute Indications | AIS Cobb angle > 50° (mature) or > 45° (immature) | High risk of lifelong progression, cosmetic deformity, and eventual cardiopulmonary compromise. |
| Absolute Indications | Documented curve progression despite compliant bracing | Indicates failure of conservative management; surgery required to arrest deformity. |
| Relative Indications | Adult scoliosis with severe radiculopathy/claudication | Surgery aimed at decompression and stabilizing the spine to prevent recurrent stenosis. |
| Relative Indications | Sagittal imbalance (SVA > 5cm) with severe pain | Correction required to restore horizontal gaze and reduce muscular fatigue. |
| Absolute Contraindications | Active osteomyelitis or epidural abscess at surgical site | High risk of hardware colonization and catastrophic systemic sepsis. |
| Absolute Contraindications | Severe cardiopulmonary disease (e.g., severe cor pulmonale) | Patient unlikely to survive the hemodynamic shifts and extubation demands of major spinal surgery. |
| Relative Contraindications | Severe Osteoporosis (T-score < -3.0) | High risk of screw pullout, subsidence, and proximal junctional failure; requires specialized techniques. |
| Relative Contraindications | Poor patient compliance or severe psychiatric illness | Inability to adhere to postoperative restrictions, increasing the risk of construct failure or nonunion. |
Pre-Operative Planning, Templating, and Patient Positioning
Before a scalpel ever touches the skin, the entire operation must be executed mentally and digitally. Preoperative planning is the blueprint for our success. Our diagnostic workup begins with high-quality, standing full-length PA and lateral radiographs of the spine on a single cassette. We assess global coronal and sagittal balance (measuring the Central Sacral Vertical Line and Sagittal Vertical Axis), pelvic parameters (Pelvic Incidence, Pelvic Tilt, Sacral Slope), and the magnitude of the curves using the Cobb method. Supine left and right side-bending radiographs are absolutely critical; they delineate the structural versus compensatory nature of the curves, dictating our fusion levels.
Advanced imaging is non-negotiable for complex deformities. A fine-cut Computed Tomography (CT) scan with 3D reconstructions allows for meticulous evaluation of pedicle morphology, particularly at the dysmorphic apical segments. We template our screw trajectories, diameters, and lengths on the axial, sagittal, and coronal planes. Magnetic Resonance Imaging (MRI) is mandatory in patients with atypical curve patterns (e.g., left thoracic curves), rapid progression, neurological deficits, or severe pain to rule out intraspinal anomalies such as a syrinx, Chiari malformation, or tethered cord.
Fusion level selection is heavily guided by the Lenke Classification system for AIS, which categorizes curves into six main types based on the structural nature of the proximal thoracic, main thoracic, and thoracolumbar/lumbar curves. We apply lumbar modifiers (A, B, C) based on the relationship of the CSVL to the lumbar apex, and sagittal modifiers (-, N, +) based on T5-T12 kyphosis. The overarching philosophy is to fuse all structural curves while sparing motion segments in non-structural, compensatory curves. Identifying the Upper Instrumented Vertebra (UIV) and Lower Instrumented Vertebra (LIV) requires analyzing the stable vertebra, neutral vertebra, and the rotation of the end vertebrae on bending films to prevent postoperative decompensation or adding-on phenomena.
Patient positioning is the foundation of a safe operation. Following induction of total intravenous anesthesia (TIVA)—which is mandatory to avoid suppressing neuromonitoring signals—the patient is carefully log-rolled onto a Jackson radiolucent frame. We utilize somatosensory evoked potentials (SSEPs), motor evoked potentials (MEPs), and continuous electromyography (EMG). The abdomen must hang completely free; any compression increases intra-abdominal pressure, which is transmitted to the epidural venous plexus, resulting in torrential intraoperative bleeding. All bony prominences are meticulously padded to prevent pressure sores and neuropathies, and the arms are positioned with less than 90 degrees of abduction and forward flexion to protect the brachial plexus.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach begins with a meticulously straight, midline longitudinal incision extending from one level above the planned UIV to one level below the LIV. We perform a subperiosteal dissection, strictly maintaining the plane between the periosteum and the paraspinal musculature to minimize blood loss. Self-retaining retractors are placed, and the exposure is carried out laterally to the tips of the transverse processes in the thoracic spine, and to the lateral aspect of the pars interarticularis in the lumbar spine. It is absolutely vital to preserve the facet joint capsules at the levels immediately adjacent to the planned fusion to prevent adjacent segment disease.
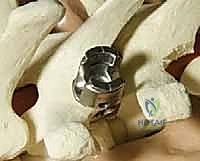
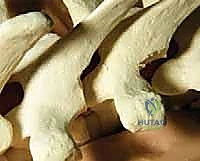
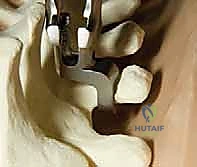
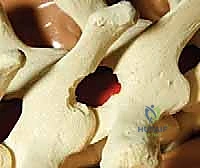
Once exposed, we perform aggressive, bilateral facetectomies at all levels within the fusion construct. This serves a dual purpose: it destabilizes the spine to facilitate deformity correction (often supplemented by multilevel Ponte osteotomies for rigid curves) and provides a highly vascularized bed for the bone graft. We then proceed with pedicle screw placement using the freehand technique. The starting point is identified using anatomical landmarks and decorticated with a high-speed burr. A curved pedicle probe (gearshift) is introduced, initially pointing lateral to avoid the medial wall, then rotated 180 degrees to point medial once the probe has passed the pedicle isthmus and entered the vertebral body.
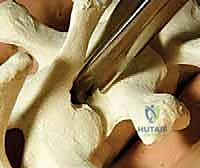
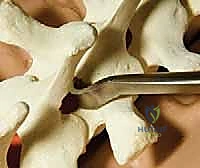
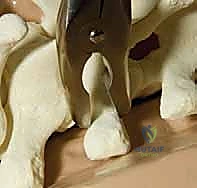
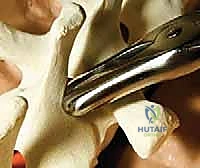
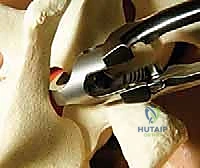
Following probing, the tract is meticulously palpated with a ball-tip feeler to ensure five intact bony walls (medial, lateral, superior, inferior, and anterior). The tract is then tapped to the appropriate diameter, re-palpated, and the pedicle screw is inserted. In hybrid constructs, hooks may be utilized, particularly at the UIV to soften the transition zone and reduce PJK. Pedicle hooks are placed in the thoracic spine by entering the facet joint and engaging the pedicle under the superior articular process. Transverse process hooks and sublaminar hooks provide additional points of fixation, particularly in the upper thoracic spine where pedicles may be prohibitively small.
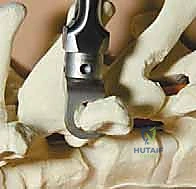
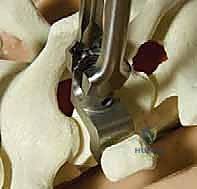
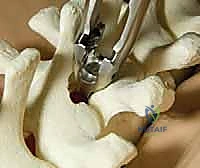
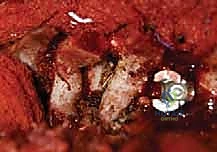
With all anchors placed, we transition to rod contouring and deformity correction. The rods are custom-bent to the desired physiological sagittal profile (restoring thoracic kyphosis and lumbar lordosis). The concave rod is typically seated first. Correction maneuvers involve a combination of rod derotation (rotating the rod 90 degrees to convert the scoliotic coronal curve into a sagittal kyphosis), translation (pulling the spine to the rod), and direct vertebral rotation (DVR) using specialized derotation tubes attached to the apical pedicle screws. Compression and distraction are applied segmentally to fine-tune the coronal alignment and level the end vertebrae. Throughout these maneuvers, we maintain continuous communication with the neurophysiologist to monitor for any signal degradation.
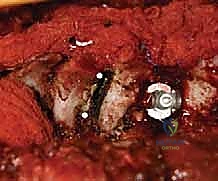
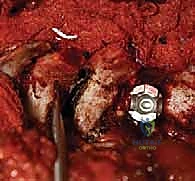
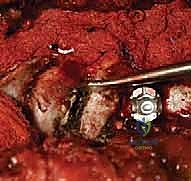
Complications, Incidence Rates, and Salvage Management
Despite meticulous planning and execution, spinal deformity surgery carries inherent and significant risks. The most devastating complication is a major neurological deficit, including paraplegia. The incidence of spinal cord injury in AIS surgery is approximately 0.5% to 1.0%. If a significant, sustained drop in MEPs or SSEPs occurs during correction, we immediately halt the procedure, increase mean arterial pressure (>90 mmHg), optimize oxygenation, and reverse the corrective maneuvers. If signals do not return, a Stagnara wake-up test is performed. If a deficit is confirmed, all hardware must be completely removed, and high-dose corticosteroids may be considered, though their efficacy remains debated.

Vascular and visceral injuries, though rare, can be rapidly fatal. Aortic injury from an errant left-sided thoracic pedicle screw is a catastrophic event requiring immediate cardiothoracic surgical intervention, massive transfusion protocols, and potentially endovascular stenting. Pleural tears and pneumothorax are more common, particularly during exposure or lateral breaches. Small pleural tears can often be managed with primary repair or a small red rubber catheter placed on suction during closure; larger tears or significant pneumothoraces necessitate formal chest tube placement.
Hardware failure and pseudarthrosis are long-term complications that typically present with increasing pain, loss of correction, or visible implant breakage on radiographs. The incidence of pseudarthrosis in modern all-pedicle screw constructs is low (1-3%) but increases significantly in adult deformity or in constructs extending to the sacrum/pelvis. Salvage management requires revision surgery, aggressive debridement of the nonunion site, exchange of broken hardware, and robust biological supplementation using autograft, allograft, and potentially osteoinductive agents like rhBMP-2.
Surgical Site Infections (SSI) occur in 1% to 4% of cases. Acute infections (within 30 days) typically present with wound drainage, erythema, and elevated inflammatory markers (CRP, ESR). The gold standard for acute SSI is urgent return to the operating room for radical irrigation and debridement, with retention of the hardware, followed by targeted intravenous antibiotic therapy based on intraoperative cultures. Delayed infections (months to years later) often involve low-virulence organisms (e.g., Cutibacterium acnes) forming biofilms on the implants, frequently necessitating complete hardware removal once a solid fusion mass is confirmed.
| Complication | Estimated Incidence | Salvage Strategy / Management |
|---|---|---|
| Neurological Deficit | 0.5% - 1.0% | Raise MAP >90, reverse correction, wake-up test, consider hardware removal, high-dose steroids. |
| Dural Tear | 2.0% - 5.0% | Primary watertight repair (6-0 Prolene), fibrin glue, Valsalva maneuver to confirm seal, bed rest. |
| Pleural Tear / Pneumothorax | 1.0% - 3.0% | Primary repair if small; formal thoracostomy tube if large or symptomatic; postoperative CXR. |
| Surgical Site Infection (Acute) | 1.0% - 4.0% | Urgent operative irrigation and debridement (I&D), retain hardware, targeted IV antibiotics. |
| Pseudarthrosis / Hardware Failure | 1.0% - 3.0% (Higher in adults) | Revision surgery, hardware exchange, aggressive decortication, biological augmentation (autograft/BMP). |
| Proximal Junctional Kyphosis (PJK) | 10% - 30% | Observation if asymptomatic; revision extension of fusion if severe pain, neurological deficit, or hardware pullout. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following major spinal deformity correction has evolved dramatically with the implementation of Enhanced Recovery After Surgery (ERAS) protocols. In the immediate postoperative phase (0 to 6 weeks), the focus is on rapid mobilization, aggressive pain management, and pulmonary hygiene. Patients are typically mobilized out of bed with physical therapy on postoperative day one. Intravenous narcotics are rapidly transitioned to a multimodal oral regimen (acetaminophen, gabapentinoids, muscle relaxants, and short-acting opioids). Due to the biomechanical rigidity of modern pedicle screw constructs, postoperative bracing (e.g., TLSO) is rarely indicated for AIS, though it may still be utilized in adult patients with poor bone quality.
During the intermediate phase (6 weeks to 3 months), patients are encouraged to gradually increase their daily activities and walking distance. Bending, lifting (greater than 10 pounds), and twisting (the "BLT" restrictions) remain strictly prohibited to protect the developing fusion mass. Radiographic follow-up at the 6-week and 3-month marks is critical to assess hardware integrity, maintenance of correction, and the early signs of incorporating bone graft. Physical therapy may be formally initiated during this phase, focusing on gentle isometric core stabilization and lower extremity flexibility.
The late phase (3 to
