Growing Rod Instrumentation: A Masterclass in Early-Onset Scoliosis Management
Key Takeaway
This masterclass provides an exhaustive guide to growing rod instrumentation for early-onset scoliosis. We delve into comprehensive surgical anatomy, meticulous preoperative planning, and granular, real-time intraoperative execution, from incision to hardware placement. Fellows will gain critical insights into neurovascular risks, precise instrument use, and strategies for managing potential pitfalls, ensuring optimal outcomes for our youngest scoliosis patients.
Introduction and Epidemiology
Early-onset scoliosis is classically defined by the diagnosis of a progressive spinal deformity at or before the age of five years, though contemporary consensus groups often extend this definition to include all scoliosis presenting before ten years of age. The etiologies of early-onset scoliosis are heterogeneous and dictate both the natural history and the algorithmic approach to surgical intervention. Primary etiologies include congenital vertebral or spinal anomalies such as unilateral unsegmented bars and hemivertebrae, which create a localized growth imbalance. Neuromuscular diseases, including cerebral palsy, spinal dysraphism, and muscular dystrophy, represent another major category, often presenting with long, sweeping, collapsing curves. Syndromic associations, such as neurofibromatosis type 1, Marfan syndrome, and various connective tissue disorders, introduce unique dysplastic curve patterns. Finally, infantile idiopathic scoliosis represents a distinct entity, typically presenting between birth and three years of age, with an unknown etiology but a likely component of genetic susceptibility.

Progressive and severe curves in this demographic are not merely orthopedic concerns; they are life-limiting conditions. The natural history of untreated early-onset scoliosis is highly associated with progressive thoracic deformity, leading to thoracic insufficiency syndrome. This syndrome is characterized by the inability of the thorax to support normal respiration or lung growth, culminating in restrictive pulmonary disease, pulmonary hypertension, cor pulmonale, and significantly increased mortality. A history of early-onset scoliosis is associated with a higher risk of cardiopulmonary decompensation in middle-aged patients, which can lead to disabling and even fatal respiratory failure. The primary goal of growing rod instrumentation is to halt curve progression while permitting continued spinal and thoracic growth, thereby maximizing pulmonary alveolar development and preserving long-term cardiopulmonary function.
Surgical Anatomy and Biomechanics
Understanding the surgical anatomy and the biomechanics of the growing spine is critical for the application of distraction-based implants. Two distinct periods of increased growth velocity are associated with an increased incidence of curve progression. The T1 to S1 growth velocity is greatest from birth until the age of five years, averaging more than 2 cm per year. By the age of five years, approximately two-thirds of the final sitting height is achieved. The spinal growth rate slows between ages five and ten years, averaging roughly 1 cm per year, before increasing again during puberty and the adolescent growth spurt, returning to a rate of 1 to 2 cm per year.

The increased spinal growth during the first years of life is directly paralleled by an exponential increase in thoracic and lung dimensions. Thoracic volume at birth is approximately 5% of the expected adult volume. By five years of age, it expands to equal 30% of adult volume. A slower rate of thoracic growth occurs from five to ten years of age, by which time it has reached 50% of the adult volume. The final 50% of adult volume is achieved during the adolescent growth spurt from ten to fifteen years of age. Alveolar multiplication is most rapid during the first eight years of life; therefore, any intervention that restricts thoracic expansion during this critical window irreversibly limits maximum pulmonary capacity.
The pathogenesis of early-onset scoliosis relies heavily on the specific etiology. Vertebral anomalies cause scoliosis by an imbalance in bone growth, secondary to either an increase in growth on a side associated with a hemivertebra or growth retardation on the side associated with a unilateral unsegmented vertebral bar. In neuromuscular and central nervous disorders, an imbalance in muscular forces is pathogenic. This likely follows the Heuter-Volkmann principle, which states that the physeal growth rate is directly related to the mechanical forces applied to it; compression inhibits physeal growth, whereas tension promotes it. The etiology and pathogenesis of infantile idiopathic scoliosis differ significantly from adolescent idiopathic scoliosis. While external factors such as intrauterine molding and infant positioning may play a role, the underlying mechanics often involve a rotatory instability of the infantile spine.

Biomechanically, growing rod instrumentation relies on distraction forces applied across the concavity of the curve to correct the deformity and stimulate longitudinal growth. These systems are inherently load-sharing constructs. Because definitive fusion is avoided in the apical regions, the proximal and distal foundation anchors are subjected to repetitive cantilever bending and pullout forces over a prolonged period. This biomechanical reality necessitates robust foundation constructs, often utilizing multiple points of fixation, to prevent anchor failure prior to skeletal maturity.
Indications and Contraindications
The natural history of early-onset scoliosis dictates the indications for surgical intervention. The natural history of infantile idiopathic scoliosis is comparatively favorable when compared with late-onset scoliosis, as spontaneous resolution occurs in a large number of patients. However, progression of congenital curves depends on the specific type of anomaly and its growth potential. Early-onset scoliosis due to neuromuscular etiologies usually follows the natural history of the underlying disease, compounded by specific problems associated with progressive curves in a hypotonic or spastic patient. Regardless of the etiology, progression of scoliosis during the first five years of life adversely affects growth as well as pulmonary function.
Surgical intervention with growing rod instrumentation is generally indicated for progressive curves exceeding 50 degrees that have failed conservative management (such as serial Mehta casting or rigid bracing) in a child with significant remaining spinal growth (typically under 10 years of age).

Contraindications include near skeletal maturity (where definitive fusion is more appropriate), severe osteoporosis that precludes adequate anchor purchase, and active systemic or surgical site infection. Poor soft tissue envelopes, often seen in severe syndromic or neuromuscular patients, represent a relative contraindication and require meticulous surgical planning to prevent wound breakdown and hardware prominence.
| Parameter | Operative Indications for Growing Rods | Non Operative Management Indications |
|---|---|---|
| Curve Magnitude | Progressive curve > 50 degrees | Curve < 40 degrees, or non-progressive |
| Age and Growth | Significant growth remaining (typically < 10 years) | Near skeletal maturity (Risser $\ge$ 3) |
| Conservative Therapy | Documented failure of serial casting or bracing | Curve responsive to casting/bracing |
| Pulmonary Status | Impending or established thoracic insufficiency | Normal pulmonary function tests |
| Bone Quality | Adequate bone stock for anchor fixation | Severe osteopenia precluding fixation |
| Soft Tissue | Adequate paraspinal muscle and skin coverage | Severe cachexia or compromised skin envelope |
Pre Operative Planning and Patient Positioning
Evaluation of the patient with early-onset scoliosis must be exhaustive. It includes a complete history, detailing the family history, prenatal history, birth history, and developmental milestones. Infantile idiopathic scoliosis has been associated with breech presentation and, particularly in boys, with premature birth.
Physical examination requires observation of gait (if the patient is ambulatory), respiratory mechanics, and truncal and pelvic balance in both the coronal and sagittal planes. The skin must be inspected for cutaneous lesions suggestive of spinal dysraphism (e.g., hairy patches, dimples, hemangiomas) or neurofibromatosis (café-au-lait macules). Any prominence on the Adams forward bending test should be quantified with a scoliometer. Crucially, any deficits in motor, sensory, or reflex function, including asymmetric abdominal reflexes, sustained clonus, or altered lower extremity deep tendon reflexes, mandate comprehensive neuraxis imaging to rule out tethered cord, syringomyelia, or Chiari malformations.

Preoperative imaging requires high-quality standing (or upright seated) posteroanterior and lateral full-spine radiographs. Supine bending or traction films are essential to assess curve flexibility and to differentiate structural from compensatory curves. An MRI of the entire neural axis from the posterior fossa to the sacrum is mandatory for all patients with early-onset scoliosis prior to surgical intervention to rule out intraspinal anomalies that could lead to catastrophic neurological injury during distraction. A preoperative 3D computed tomography scan may be indicated in cases of complex congenital scoliosis to delineate aberrant pedicle anatomy and plan anchor trajectories.
Multidisciplinary optimization is critical. Pulmonology and cardiology consultations are required to assess baseline respiratory function, pulmonary hypertension, and cardiac reserve. Nutritional status must be optimized, particularly in neuromuscular patients, to mitigate the risk of postoperative wound complications and infection.

In the operating room, patient positioning requires meticulous attention to detail. The patient is placed prone on a pediatric Jackson frame or OSI table, ensuring the abdomen is completely free to decrease intra-abdominal pressure, thereby reducing epidural venous engorgement and minimizing intraoperative blood loss. All bony prominences are padded. Intraoperative neuromonitoring, including somatosensory evoked potentials and motor evoked potentials, is standard of care. Baseline signals must be obtained prior to positioning and closely monitored throughout the procedure, particularly during foundation placement and rod distraction.
Detailed Surgical Approach and Technique
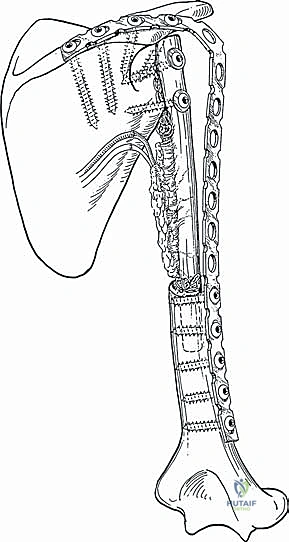
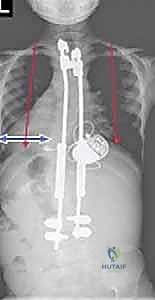
The surgical technique for growing rod instrumentation has evolved significantly, transitioning from single-rod constructs to dual-rod constructs, and more recently, to magnetically controlled growing rods. Dual-rod constructs provide superior biomechanical stability, better control of the sagittal profile, and a lower incidence of implant failure compared to single-rod systems.
Foundation Selection and Exposure
The procedure begins with the selection of proximal and distal foundation sites. The proximal foundation is typically located in the upper thoracic spine (T2-T4), while the distal foundation is anchored in the lower lumbar spine or pelvis, depending on the curve type and pelvic obliquity. Exposure is strictly limited to the foundation sites to prevent inadvertent spontaneous fusion of the intervening segments. A standard midline longitudinal incision is made over the planned proximal and distal foundations. Subperiosteal dissection is performed only at the levels chosen for instrumentation.

Proximal Anchor Placement
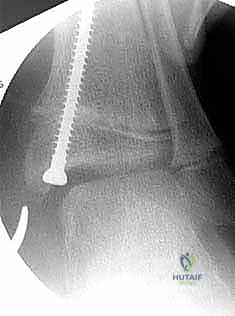
The proximal foundation typically utilizes pedicle screws, pedicle hooks, or transverse process hooks. Due to the small size of pediatric upper thoracic pedicles, a claw construct (e.g., a supralaminar hook combined with an infralaminar or pedicle hook) is often employed to maximize pullout strength and distribute cantilever forces. If pedicle screws are used, precise anatomical landmarks and fluoroscopic or navigation guidance are utilized to ensure safe trajectory without breaching the medial pedicle wall. The foundation spans two to three levels to ensure adequate fixation.
Distal Anchor Placement
The distal foundation relies heavily on pedicle screws placed in the lower lumbar spine (L3-L5). In non-ambulatory neuromuscular patients with significant pelvic obliquity, the distal foundation must be extended to the pelvis utilizing iliac or S2-alar-iliac screws. Screws are placed bilaterally over two to three segments. Meticulous decortication and bone grafting (autograft or allograft) are performed strictly within the confined areas of the proximal and distal foundations to achieve localized arthrodesis, providing a stable anchor for the distraction rods.
Rod Contouring and Passage
Once the foundations are established, the intervening soft tissue envelope must be prepared for rod passage. A submuscular or subfascial plane is developed between the proximal and distal incisions using a Cobb elevator or specialized rod passers. It is imperative to stay superficial to the periosteum of the apical vertebrae to avoid stimulating osteogenesis and causing spontaneous autofusion of the curve.

Two rods are contoured to match the desired sagittal profile—maintaining thoracic kyphosis and lumbar lordosis. In traditional growing rod systems, the rods are connected via a tandem connector (domino) located in the thoracolumbar region. In magnetically controlled growing rod systems, the actuator mechanism is positioned centrally. The rods are passed blindly through the prepared submuscular plane and engaged into the proximal and distal foundation anchors.
Distraction and Final Assembly
After rod engagement, distraction is applied. The goal of the index procedure is not complete deformity correction, but rather curve stabilization and restoration of spinal balance. Over-distraction must be strictly avoided to prevent neurological injury and immediate anchor pullout. Neuromonitoring signals are continuously evaluated during this phase. Once appropriate distraction is achieved, the set screws are definitively torqued. If traditional growing rods are used, the tandem connectors are secured, leaving adequate rod length to accommodate future lengthenings.

Closure Techniques
Closure of the incisions in early-onset scoliosis patients requires meticulous technique due to the high risk of wound dehiscence and hardware prominence. The deep fascial layers are closed with heavy absorbable sutures to ensure a robust soft tissue envelope over the anchors. Subcutaneous tissues are closed in multiple layers to eliminate dead space, and the skin is approximated with a subcuticular closure. Drains may be utilized at the foundation sites depending on the extent of dead space and intraoperative bleeding.
Complications and Management
Growing rod instrumentation is associated with one of the highest complication rates in pediatric orthopedic surgery. The repetitive nature of the lengthening procedures, the prolonged duration of implant retention, and the biomechanical stress on the anchors contribute to a cumulative complication rate that often exceeds 50% over the course of treatment. A well-documented phenomenon in distraction-based constructs is the "Law of Diminishing Returns," wherein the amount of spinal length gained decreases with each subsequent lengthening procedure, largely due to progressive stiffening and spontaneous autofusion of the uninstrumented segments.

Implant-related complications are the most frequent. Rod fracture occurs due to metal fatigue from cyclical loading across the unfused apex. Management requires surgical revision with rod exchange. Anchor pullout or failure, particularly at the proximal thoracic foundation, is secondary to the constant cantilever forces. Salvage strategies involve extending the foundation proximally or utilizing alternative fixation modalities such as sublaminar bands.
Deep surgical site infections are a devastating complication, with rates significantly higher than in primary adolescent idiopathic scoliosis surgery. The presence of a large dead space, repeated surgical exposures in traditional growing rods, and compromised host factors contribute to this risk. Staphylococcus aureus and Cutibacterium acnes are the most common pathogens. C. acnes infections often present insidiously with delayed wound drainage or implant prominence without systemic signs of infection.

Neurological injury is rare but catastrophic, typically occurring during the index distraction or subsequent aggressive lengthenings. Immediate loss of neuromonitoring signals mandates immediate release of distraction, elevation of mean arterial pressure, and potential removal of the implants if signals do not recover.
| Complication | Estimated Incidence | Etiology and Risk Factors | Management and Salvage Strategies |
|---|---|---|---|
| Rod Fracture | 15% - 30% | Metal fatigue, cyclical loading, pseudoarthrosis at foundation | Surgical revision with rod exchange; consider larger diameter rods |
| Anchor Pullout | 10% - 25% | Cantilever forces, poor bone quality, over-distraction | Extend foundation to adjacent levels; utilize sublaminar bands or claws |
| Deep Infection | 10% - 20% | Repeated exposures, poor soft tissue envelope, biofilm formation | Aggressive I&D, targeted IV antibiotics; implant retention if possible, removal if refractory |
| Spontaneous Autofusion | 30% - 50% | Subperiosteal dissection, hematoma ossification, diminished returns | Accept fusion if near maturity; anterior release or osteotomies if severe restriction occurs |
| Junctional Kyphosis | 10% - 15% | Failure to contour sagittal plane, disruption of posterior tension band | Proximal extension of instrumentation; meticulous preservation of interspinous ligaments |
| Neurological Deficit | < 1% | Over-distraction, ischemic injury to spinal cord, unrecognized tethering | Immediate release of distraction, optimize hemodynamics, high-dose steroids (controversial) |
Post Operative Rehabilitation Protocols
Immediate postoperative care takes place in the pediatric intensive care unit for close hemodynamic and respiratory monitoring. Pain management is typically achieved with patient-controlled analgesia or continuous epidural infusions, transitioning to oral medications as tolerated. Early mobilization is encouraged to prevent pulmonary complications. Depending on the stability of the intraoperative construct and the patient's bone quality, a custom-molded thoracolumbosacral orthosis may be prescribed for 3 to 6 months postoperatively to protect the foundation anchors from excessive rotational and bending forces.

The lengthening protocol depends entirely on the type of implant utilized. For traditional growing rods, patients must return to the operating room every 6 months for an open lengthening procedure. This involves a small incision over the tandem connectors, loosening of the set screws, and manual distraction using a spreader. The repeated anesthetic exposures and surgical incisions are significant drawbacks of this technique.
Magnetically controlled growing rods have revolutionized the postoperative protocol. Lengthenings are performed in the outpatient clinic setting using an external remote controller. The standard protocol involves lengthenings every 2 to 3 months. The external remote controller generates a rotating magnetic field that drives the internal actuator of the rod, resulting in precise, non-invasive distraction. This paradigm shift has drastically reduced the psychological trauma, anesthetic risks, and infection rates associated with repetitive open surgeries.
The culmination of the growing rod treatment pathway is the transition to a definitive spinal fusion. This is typically considered when the patient approaches skeletal maturity (Risser stage 3 or 4, or closure of the triradiate cartilage), or when the "Law of Diminishing Returns" renders further lengthenings futile. The final procedure involves subperiosteal exposure of the entire curve, removal of the growing rod implants, widespread facetectomies, complete pedicle screw instrumentation, and robust bone grafting to achieve a solid, definitive arthrodesis.
Summary of Key Literature and Guidelines
The evolution of growing rod instrumentation is heavily grounded in pivotal academic literature. Early work by Akbarnia et al. established the superiority of dual-rod constructs over single-rod systems, demonstrating significantly better initial curve correction, maintenance of correction, and a lower rate of implant failure. The Growing Spine Study Group has been instrumental in publishing large, multicenter retrospective and prospective analyses that define the natural history and complication profiles of these interventions.
Bess et al. provided a landmark analysis on the complications of traditional growing rods, quantifying the direct correlation between the number of surgical lengthenings and the exponential increase in complication rates, particularly surgical site infections and spontaneous autofusion. This data served as the primary catalyst for the development of non-invasive lengthening technologies.
The introduction of magnetically controlled growing rods was validated by Cheung et al. and further refined by Skaggs et al., who published early results demonstrating comparable curve control and spinal growth to traditional systems, but with a profound reduction in surgical interventions. Current consensus guidelines from the Scoliosis Research Society and the Pediatric Orthopaedic Society of North America support the use of magnetically controlled growing rods as the standard of care for distraction-based treatment in early-onset scoliosis, provided the patient does not have contraindications such as a strict requirement for frequent, incompatible MRI evaluations. Ongoing research continues to focus on optimizing lengthening frequencies, mitigating actuator failures in magnetic rods, and understanding the long-term pulmonary outcomes of distraction-based therapies.
Clinical & Radiographic Imaging












You Might Also Like