Perioperative & Orthopaedic Medicine: Preventing Dangerous Blood Clots
Key Takeaway
Here are the crucial details you must know about Perioperative & Orthopaedic Medicine: Preventing Dangerous Blood Clots. Thromboembolic disease, a common perioperative and orthopaedic complication, involves improper blood clotting (thrombosis) that can migrate (embolism), potentially leading to deep venous thrombosis (DVT) or pulmonary embolism (PE). Key factors contributing include endothelial damage from surgery, blood stasis due to immobility, and hypercoagulability. Age and prior DVT are significant risk factors.
Comprehensive Introduction and Patho-Epidemiology
Thromboembolic disease represents one of the most insidious and potentially devastating complications encountered in the field of orthopaedic surgery. Encompassing both deep venous thrombosis (DVT) and pulmonary embolism (PE), venous thromboembolism (VTE) often manifests with a clinically silent progression that can culminate in sudden, catastrophic cardiopulmonary collapse. The high-risk nature of orthopaedic procedures—particularly major joint arthroplasties and pelvic or lower extremity trauma surgeries—creates a perfect storm for thrombogenesis. Understanding the fundamental pathophysiology, epidemiology, and long-term sequelae of VTE is absolutely paramount for the operating surgeon, as the balance between mitigating clot formation and preventing catastrophic postoperative hemorrhage dictates the perioperative medical management of every surgical patient.
The pathophysiological foundation of thromboembolic disease is elegantly described by the Virchow triad, which delineates three primary prerequisites for in vivo thrombosis: endothelial damage, venous stasis, and hypercoagulability. Endothelial denudation is an unavoidable consequence of surgical trauma. The mechanical disruption of the vascular intima exposes subendothelial collagen and tissue factor, rapidly initiating the coagulation cascade. Platelets immediately undergo a tripartite physiological response: adhesion to the exposed subendothelium via von Willebrand factor, activation and secretion of prothrombotic mediators (such as thromboxane A2 and adenosine diphosphate), and subsequent aggregation to form the primary hemostatic plug. Concurrently, the exposure of tissue factor triggers the extrinsic coagulation cascade, ultimately leading to the generation of thrombin, which converts soluble fibrinogen into an insoluble cross-linked fibrin mesh that stabilizes the nascent thrombus.

Venous stasis and hypercoagulability further compound the risk profile in the orthopaedic patient. Stasis allows for the localized accumulation of activated clotting factors and prevents their clearance by the reticuloendothelial system. In the perioperative period, stasis is exacerbated by profound immobility secondary to pain, regional or general anesthesia, and the mechanical application of pneumatic tourniquets. Hypercoagulability is induced by the systemic inflammatory response to surgical trauma, which upregulates acute-phase reactants and procoagulant factors while simultaneously suppressing endogenous fibrinolytic activity. This transient, acquired hypercoagulable state can persist for weeks following major orthopaedic interventions, necessitating prolonged vigilance and extended-duration prophylaxis in high-risk populations.

The epidemiological landscape of thromboembolic disease in orthopaedics is highly variable, heavily dependent on the anatomical location of the surgery and the specific definitions utilized in clinical studies (e.g., symptomatic versus asymptomatic, distal versus proximal DVT). Distal thromboses, defined as those occurring below the popliteal trifurcation, are relatively common but carry a substantially lower risk of clinically significant pulmonary embolization. Conversely, proximal DVTs (involving the popliteal, femoral, or iliac veins) pose a severe threat for massive embolization. Complications of thromboembolic disease extend beyond acute mortality and include recurrent DVT—which carries a risk four to eight times higher after an initial event—and postthrombotic syndrome. Postthrombotic syndrome is a chronic, debilitating condition characterized by ambulatory venous hypertension, chronic edema, hyperpigmentation, induration, and eventual venous ulceration, fundamentally altering the patient's long-term quality of life.
Detailed Surgical Anatomy and Biomechanics
A profound understanding of the venous anatomy and the biomechanics of venous return in the lower extremity is essential for comprehending the pathogenesis of orthopaedic-induced VTE. The venous system of the lower limb is anatomically divided into the superficial, deep, and perforating venous networks. The deep venous system, which accompanies the major arterial trunks, is the primary conduit for venous return and the predominant site for clinically significant DVT formation. The deep veins of the calf—the anterior tibial, posterior tibial, and peroneal veins—coalesce to form the popliteal vein, which traverses the adductor hiatus to become the femoral vein. The soleal and gastrocnemius venous sinuses serve as large capacitance reservoirs within the muscular compartments of the calf. These thin-walled, valveless sinusoids are highly susceptible to stasis during periods of immobility, making them the most frequent nidus for initial thrombus formation.

The biomechanics of venous return against gravity rely heavily on the integrity of the calf muscle pump and the competence of the venous bicuspid valves. During ambulation, the contraction of the gastrocnemius and soleus muscles compresses the deep venous sinuses, forcefully propelling blood proximally toward the heart. The bicuspid valves, constructed of delicate folds of tunica intima, prevent the retrograde flow of blood during muscular relaxation. In the perioperative period, the cessation of the calf muscle pump due to anesthesia and bed rest leads to profound pooling of blood within the lower extremity. The resultant distension of the venous capacitance vessels can cause transient valvular incompetence, further exacerbating venous stasis and creating a localized hypoxic environment that activates the vascular endothelium to express pro-inflammatory and pro-coagulant adhesion molecules.
Orthopaedic surgical interventions introduce unique biomechanical and anatomical disruptions that drastically increase thrombogenic potential. The application of a pneumatic tourniquet, while essential for maintaining a bloodless surgical field, completely obliterates arterial inflow and venous outflow, creating absolute stasis distal to the cuff. Upon deflation, the sudden reperfusion releases a massive bolus of accumulated metabolic byproducts, tissue factor, and microthrombi into the systemic circulation. Furthermore, intramedullary reaming and the pressurization of polymethylmethacrylate (PMMA) bone cement during total joint arthroplasty force marrow fat and highly thrombogenic cellular debris into the venous circulation, frequently resulting in intraoperative micro-embolic showers.

Surgical positioning also plays a critical role in altering venous hemodynamics. Prolonged extreme flexion of the hip or knee, such as that required during total knee arthroplasty or certain arthroscopic procedures, can mechanically kink the popliteal or femoral veins, severely impeding venous outflow. Similarly, the use of a fracture table with aggressive traction can cause direct compression of the perineal and inguinal vasculature. The operating surgeon must remain acutely aware of these anatomical and biomechanical variables, employing meticulous surgical technique, minimizing tourniquet time, and ensuring optimal patient positioning to mitigate the iatrogenic induction of venous stasis and endothelial injury.
Exhaustive Indications and Contraindications
The selection of an appropriate thromboprophylactic regimen requires a nuanced, individualized risk-benefit analysis, weighing the imperative to prevent fatal pulmonary embolism against the potentially catastrophic consequences of postoperative hemorrhage, such as epidural hematoma, periprosthetic joint infection, or compartment syndrome. The armamentarium of VTE prophylaxis includes both mechanical modalities and a diverse array of pharmacological agents, each with specific mechanisms of action, indications, and contraindications. Pharmacologic prophylaxis is universally mandated by Surgical Care Improvement Project (SCIP) quality measures for high-risk procedures, notably total hip arthroplasty (THA), total knee arthroplasty (TKA), and hip fracture surgery.
Aspirin, an irreversible inhibitor of the cyclooxygenase (COX) enzyme, reduces the synthesis of thromboxane A2, thereby impairing platelet activation and aggregation. While historically considered one of the weakest prophylactic agents, recent literature and contemporary guidelines have validated its use as a primary prophylactic agent in standard-risk arthroplasty patients, particularly when combined with mechanical prophylaxis. Low-molecular-weight heparin (LMWH), such as enoxaparin, reversibly inhibits Factor Xa and Factor IIa via the activation of antithrombin III (ATIII). LMWH provides robust VTE protection without the need for routine laboratory monitoring, though it carries a higher bleeding risk than aspirin and is contraindicated in patients with a history of heparin-induced thrombocytopenia (HIT).

Direct oral anticoagulants (DOACs), including direct Factor Xa inhibitors (e.g., rivaroxaban, apixaban) and direct thrombin inhibitors (e.g., dabigatran), have gained significant traction due to their oral administration and lack of monitoring requirements. Rivaroxaban, a direct Xa inhibitor, provides excellent efficacy but is associated with a higher risk of major bleeding events compared to LMWH in some orthopaedic cohorts. Warfarin, a vitamin K antagonist that inhibits the hepatic synthesis of Factors II, VII, IX, and X, as well as proteins C and S, requires rigorous monitoring of the International Normalized Ratio (INR) with a target of 2.0 to 3.0. Its use is increasingly relegated to patients with pre-existing indications for systemic anticoagulation (e.g., mechanical heart valves, atrial fibrillation) due to its narrow therapeutic index and numerous drug-diet interactions.
Table: Pharmacologic Thromboprophylaxis in Orthopaedic Surgery
| Agent Class | Mechanism of Action | Primary Indications | Absolute/Relative Contraindications | Reversal Agent |
|---|---|---|---|---|
| Aspirin | Irreversible COX-1/2 inhibition; decreases TXA2 | Standard-risk THA/TKA, multimodal protocols | Active GI bleed, severe allergy, concurrent NSAID use | Platelet transfusion (partial efficacy) |
| LMWH (Enoxaparin) | ATIII-mediated inhibition of Factor Xa > IIa | High-risk THA/TKA, trauma, pelvic/acetabular surgery | History of HIT, active major bleed, severe renal impairment | Protamine Sulfate (partial reversal) |
| Warfarin | Vitamin K antagonist (Factors II, VII, IX, X, C, S) | Extended prophylaxis, concurrent A-fib/mechanical valve | Pregnancy (teratogenic), inability to monitor INR, non-compliance | Vitamin K, Fresh Frozen Plasma (FFP), PCC |
| Fondaparinux | Synthetic pentasaccharide; indirect Xa inhibitor via ATIII | Hx of HIT, high-risk arthroplasty | Severe renal impairment (CrCl < 30 mL/min), low body weight | None (Andexanet alfa off-label) |
| Direct Xa Inhibitors | Direct, reversible inhibition of Factor Xa (e.g., Rivaroxaban) | THA/TKA prophylaxis, outpatient transition | Active bleeding, severe hepatic impairment, concurrent CYP3A4 inhibitors | Andexanet alfa |
Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning for VTE mitigation begins with rigorous patient-specific risk stratification. The baseline risk of thromboembolic disease increases exponentially with age, particularly in patients over 40 years old. A comprehensive history must interrogate for prior VTE events—which remain the strongest independent predictor of future thrombosis—as well as active malignancy, obesity, and the use of exogenous estrogens or selective estrogen receptor modulators. Furthermore, the surgeon must be highly suspicious of underlying genetic thrombophilias in patients with a strong family history of unprovoked clots. Factor V Leiden, the most common inherited thrombophilia, involves a point mutation that renders Factor V resistant to degradation by activated Protein C, thereby prolonging the active clotting process. Other critical genetic factors include the Prothrombin G20210A mutation, hyperhomocysteinemia, and deficiencies in endogenous anticoagulants such as Antithrombin III, Protein C, and Protein S.

Procedure-specific factors must also be meticulously templated into the prophylactic algorithm. The risk of VTE is heavily dictated by the anatomical location and magnitude of the surgical insult. Procedures distal to the knee carry a remarkably low risk of clinically significant PE, and routine chemoprophylaxis is generally not recommended for isolated distal fractures, arthroscopic procedures, or upper extremity surgeries in the absence of patient-specific risk factors. Conversely, major pelvic trauma and hip fracture surgeries carry the highest risk of VTE in the orthopaedic spectrum, surpassing even elective THA and TKA. Interestingly, while TKA is associated with a higher overall incidence of distal DVT, THA carries a higher relative risk of proximal DVT and subsequent pulmonary embolism.
Patient positioning and intraoperative setup require deliberate planning to minimize iatrogenic venous stasis. When utilizing a fracture table for femoral or hip procedures, the perineal post must be adequately padded, and traction should be applied only for the minimum duration necessary to achieve reduction, thereby preventing prolonged occlusion of the pelvic venous outflow tract. In the lithotomy position or during the use of a leg holder for knee arthroscopy, care must be taken to avoid acute angulation of the popliteal fossa. Additionally, the pre-operative templating of tourniquet use should include strategies for intermittent deflation if the procedure is anticipated to exceed 120 minutes, allowing for the restoration of venous outflow and the flushing of accumulated prothrombotic metabolites from the distal extremity.

Step-by-Step Surgical Approach and Fixation Technique
While the primary prevention of VTE is largely pharmacological and mechanical, specific intraoperative surgical techniques and interventional procedures play a direct role in mitigating embolic risk. The choice of anesthesia is the first critical "surgical" step; neuraxial anesthesia (spinal or epidural) has been definitively shown to reduce the incidence of DVT and blood loss compared to general anesthesia. This protective effect is attributed to sympathetic blockade, which induces profound venodilation and increases lower extremity venous blood flow, counteracting the stasis component of Virchow's triad. Furthermore, meticulous surgical technique—emphasizing atraumatic tissue handling, precise hemostasis, and the avoidance of excessive venous retraction—directly minimizes endothelial injury and the subsequent release of systemic tissue factor.

In highly selected scenarios where pharmacological prophylaxis is absolutely contraindicated (e.g., severe traumatic brain injury, active massive hemorrhage, or recent spinal cord surgery), or in patients who have suffered a massive PE despite therapeutic anticoagulation, the surgical placement of an Inferior Vena Cava (IVC) filter becomes a critical, life-saving intervention. The modern surgical approach for IVC filter deployment is performed percutaneously under fluoroscopic guidance. The patient is positioned supine, and the right internal jugular or right common femoral vein is accessed using the Seldinger technique. A guidewire is advanced into the IVC, and an inferior venacavogram is performed to delineate the renal veins, assess for preexisting caval thrombus, and measure the caval diameter.
The deployment of the IVC filter requires exacting precision. The delivery sheath is advanced over the guidewire, and the filter is deployed strictly infrarenally—typically with the apex positioned just below the confluence of the lowest renal vein. This positioning is critical; suprarenal deployment risks renal vein thrombosis and subsequent renal failure, while placement too distally may fail to capture emboli originating from the iliac veins. Once the filter is unsheathed, its metallic struts expand to engage the caval wall, creating a mechanical sieve that traps large, potentially fatal emboli while maintaining continuous venous blood flow. A post-deployment venogram confirms adequate expansion, proper alignment, and the absence of caval injury. Modern retrievable filters should be aggressively tracked and removed as soon as the patient's bleeding risk subsides and definitive chemoprophylaxis can be safely initiated.
Complications, Incidence Rates, and Salvage Management
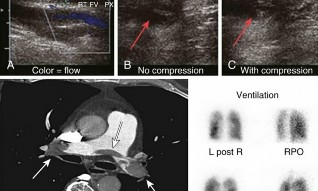
Despite the most rigorous prophylactic protocols, thromboembolic complications remain a persistent threat in orthopaedic surgery, necessitating rapid diagnostic acumen and aggressive salvage management. The clinical diagnosis of DVT is notoriously unreliable; classic signs such as calf pain, palpable cords, swelling, and a positive Homan's sign are absent in the majority of cases. Studies demonstrate that 50% of patients with classic clinical signs have no DVT on advanced imaging, and conversely, 50% of patients with venogram-proven extensive thrombosis exhibit entirely normal physical findings. Therefore, diagnosis relies heavily on a high index of clinical suspicion and objective imaging.

Duplex compression ultrasonography is the most practical, non-invasive, and easily repeatable bedside diagnostic modality. The hallmark finding of a DVT on ultrasound is a "noncompressible vein," a sign that is approximately 95% sensitive and specific for proximal lower extremity thrombosis. While D-dimer assays are highly sensitive, their specificity in the postoperative orthopaedic patient is exceptionally poor, as surgical trauma universally elevates D-dimer levels; thus, a positive D-dimer is practically useless, though a negative result can confidently rule out a significant clot. For suspected Pulmonary Embolism, Spiral CT Pulmonary Angiography is the gold standard, capable of visualizing filling defects within the pulmonary arterial tree. In patients with severe contrast allergies or renal failure, a Ventilation-Perfusion (V/Q) scan is the preferred alternative.

The complications of VTE extend beyond the acute event. Pulmonary embolism can manifest as sudden death (particularly with massive saddle emboli), pleuritic chest pain, dyspnea, tachypnea, and right-sided heart failure (cor pulmonale). Electrocardiogram (ECG) findings are often nonspecific, with sinus tachycardia being the most common abnormality, though the classic S1Q3T3 pattern indicates severe right ventricular strain. Chest radiographs may initially be normal but can later reveal focal oligemia (Westermark sign) or a peripheral wedge-shaped opacity (Hampton hump) indicative of pulmonary infarction. Long-term complications include Postthrombotic Syndrome (PTS), characterized by chronic venous insufficiency, intractable edema, induration, hyperpigmentation, and stasis ulceration.

Table: Complications of VTE and Prophylaxis
| Complication | Incidence / Risk Factors | Diagnostic Modality | Salvage Management / Treatment |
|---|---|---|---|
| Pulmonary Embolism (PE) | Highest after hip fracture surgery and THA | Spiral CT Angiography, V/Q Scan | Systemic anticoagulation; catheter-directed thrombolysis or surgical embolectomy for massive saddle PE with hemodynamic instability. |
| Postthrombotic Syndrome | 20-50% of patients within 1-2 years of DVT | Clinical diagnosis (Villalta scale), Ultrasound for reflux | Graduated compression stockings, elevation, wound care for ulcerations. |
| Major Postoperative Bleeding | Increased with DOACs, Warfarin, LMWH | Clinical exam, serial Hemoglobin/Hematocrit | Discontinuation of agent, reversal agents (Protamine, Vit K, Andexanet alfa), surgical evacuation of hematoma. |
| Heparin-Induced Thrombocytopenia | 0.1-5% with Unfractionated Heparin; lower with LMWH | Serotonin Release Assay (SRA), ELISA for PF4 antibodies | Immediate cessation of all heparin products; transition to direct thrombin inhibitor (Argatroban, Bivalirudin) or Fondaparinux. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation phase is a critical period where mechanical prophylaxis and early mobilization synergize with pharmacological agents to prevent VTE. The absolute cornerstone of mechanical prophylaxis is early, aggressive mobilization. Having the patient out of bed and bearing weight on postoperative day zero dramatically increases venous return via the activation of the calf muscle pump, directly counteracting the stasis component of Virchow's triad. Graduated elastic compression hose are frequently utilized to reduce venous capacitance and increase deep venous flow velocity; however, literature clearly indicates that they are insufficient as a standalone prophylactic measure and must be used as an adjunct.

Intermittent Pneumatic Compression Devices (IPCDs) play a vital role in the immediate postoperative phase, particularly while the patient remains bedbound. IPCDs not only mechanically expel blood from the deep venous sinuses of the calf, but they also induce a systemic biochemical effect. The cyclical compression of the extremity stimulates the vascular endothelium to release endogenous tissue plasminogen activator (tPA), thereby enhancing the body's intrinsic fibrinolytic system. The 2012 American College of Chest Physicians (ACCP) guidelines strongly endorse the use of IPCDs (Grade 1C
Clinical & Radiographic Imaging Archive


