Case Study: Diagnosing Distal Fibula Osteoid Osteoma in a Young Athlete
Key Takeaway
Osteoid osteoma diagnosis in young athletes often begins with characteristic symptoms like chronic, nocturnal, NSAID-responsive localized pain. Physical examination reveals point tenderness. Definitive diagnosis relies on imaging, with X-rays showing cortical thickening and CT scans precisely locating the radiolucent nidus, often surrounded by reactive sclerosis, as seen in the distal fibula case.
Patient Presentation and History
We present a typical, yet diagnostically challenging, case of Osteoid Osteoma in an active young patient, highlighting the characteristic clinical features and the diagnostic journey.
A 16-year-old male, otherwise fit and healthy, presented to the orthopedic oncology and trauma clinic with a nine-month history of progressive right ankle pain. He is an avid soccer player, highly active in competitive high school sports, and participates in year-round club tournaments. The chronic nature of his symptoms, combined with his athletic profile, initially led primary care providers and physical therapists down a diagnostic pathway focused on sports-related overuse injuries, delaying the identification of the true underlying pathology.
The patient initially reported a vague, intermittent ache in his right ankle, predominantly localized over the lateral malleolus. Over several months, the pain progressively worsened in both intensity and frequency, evolving from an activity-related annoyance to a debilitating condition. Key characteristics of the pain profile included nocturnal exacerbation, which is a hallmark of this pathology. The pain was consistently worse at night, frequently waking him from sleep and severely disrupting his circadian rhythm.
He reported near-complete, albeit temporary, relief with over-the-counter non-steroidal anti-inflammatory drugs, specifically ibuprofen. By the time of his presentation to our clinic, he was consuming maximum daily doses of NSAIDs to maintain basic daily function. This profound response to prostaglandin-inhibiting medication serves as a crucial diagnostic clue, directly pointing toward the cyclooxygenase-2 rich environment of an osteoid osteoma nidus. While the pain was present at rest, it was significantly aggravated by physical activity, particularly the cutting, pivoting, and high-impact running associated with soccer.
The pain was consistently localized to the lateral aspect of the right ankle, specifically around the distal fibula. A thorough review of his athletic history revealed no specific inciting event, acute trauma, or inversion injury that would suggest a lateral ankle sprain or syndesmotic disruption.
No constitutional symptoms such as fever, chills, unexplained weight loss, or night sweats were reported, lowering the clinical suspicion for aggressive malignant processes or systemic infections. There was no neurological compromise, paresthesia, or weakness reported in the distal extremity. He denied any skin changes, massive swelling, or systemic warmth initially, though subtle localized soft tissue swelling became apparent in the later months of his symptomatic period. The chronic pain was severely impacting his sleep quality, academic performance due to fatigue, and his psychological well-being due to his inability to participate in competitive sports.
His past medical history was entirely unremarkable. He had undergone no prior surgeries, possessed no significant medical illnesses, and had no known drug allergies. His social history noted him as a non-smoker with occasional, socially acceptable alcohol use for his age demographic. He lives at home with his parents and attends high school full-time. Family history was negative for primary bone malignancies, metabolic bone diseases, or systemic rheumatologic conditions.
Clinical Examination Findings
The physical examination was conducted with a focus on differentiating intra-articular ankle pathology, ligamentous instability, and osseous lesions.
Upon general inspection, the patient appeared to be in no acute distress while seated at rest. However, dynamic gait analysis revealed a subtle antalgic limp on the right side. There was a demonstrably decreased stance phase on the affected right lower extremity, with a slight external rotation of the foot to offload the lateral column during toe-off. There was no obvious gross deformity, malalignment of the mechanical axis, or significant atrophy of the gastrocnemius-soleus complex or anterior compartment musculature.
Local inspection of the right ankle revealed mild, localized soft tissue swelling over the distal lateral malleolus, extending slightly proximal to the joint line. There were no significant erythema, warmth, trophic skin changes, or venous distention observed. The skin overlying the lateral ankle was intact with no sinus tracts or prior surgical scars.
Palpation was the most localizing aspect of the examination. Exquisite, point tenderness was elicited directly over the lateral cortex of the distal fibula, approximately three centimeters proximal to the distal tip of the lateral malleolus. This tenderness was highly reproducible and distinct from the anterior talofibular ligament and the calcaneofibular ligament. Palpation of the syndesmosis, including the anterior inferior tibiofibular ligament, yielded no pain. There was a very mild increase in local skin temperature directly over the point of maximum tenderness compared to the contralateral ankle, likely secondary to localized hyperemia. No crepitus, fluctuance, or palpable soft tissue mass was noted.
Range of motion testing of the right ankle demonstrated restrictions primarily secondary to pain and muscular guarding. Dorsiflexion was limited to 10 degrees, compared to a normal 20 degrees on the contralateral side, and was painful at the end range. Plantarflexion was limited to 35 degrees, compared to 45 degrees contralaterally, also eliciting pain at the extreme of motion. Inversion and eversion were restricted to 10 degrees and 5 degrees respectively, both provoking pain that radiated proximally up the fibular shaft. Subtalar and midfoot articulations demonstrated mildly restricted motion, which normalized when the ankle was stabilized, indicating the restriction was primarily protective guarding rather than true capsular contracture. The contralateral left ankle exhibited a full, supple, and painless range of motion in all planes.
Special orthopedic tests were performed to rule out confounding athletic injuries. The anterior drawer test and talar tilt test were negative, indicating an intact lateral ligamentous complex. The syndesmotic squeeze test, performed at the mid-calf, elicited pain distally at the site of the fibular lesion, which can occasionally be a false positive for syndesmotic injury when an osseous lesion is present in the fibula due to the transmission of compressive forces. The external rotation stress test was negative for widening or medial clear space pain.
Neurological assessment revealed normal motor function. Motor strength was graded 5/5 in all major muscle groups of the right lower extremity, including foot dorsiflexion (tibialis anterior), plantarflexion (gastrocnemius-soleus), inversion (tibialis posterior), eversion (peroneus longus and brevis), and great toe extension (extensor hallucis longus). Sensory examination demonstrated intact light touch and pinprick sensation in all dermatomes of the right foot and ankle, specifically mapping the L4, L5, S1, and S2 distributions. There was no paresthesia or dysesthesia in the distribution of the superficial peroneal nerve or the sural nerve, which traverse near the anatomical location of the pain. Deep tendon reflexes, specifically the patellar and Achilles reflexes, were 2+ and symmetric bilaterally. The Babinski reflex was downgoing, and there was no sustained ankle clonus, ruling out upper motor neuron pathology. Vascular examination showed strong, symmetric, and bounding dorsalis pedis and posterior tibial pulses, with brisk capillary refill in all digits.
Imaging and Diagnostics Modalities
The diagnostic workup for this patient required a tiered approach, moving from basic radiographic screening to advanced cross-sectional imaging to accurately delineate the lesion and plan therapeutic intervention.
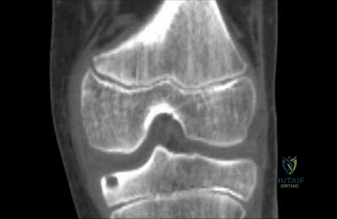
Initial plain radiography included weight-bearing anteroposterior, mortise, and lateral views of the right ankle. The radiographs demonstrated a subtle area of cortical thickening and dense sclerosis along the lateral cortex of the distal fibula, approximately three centimeters proximal to the tibial plafond. Within this area of cortical hypertrophy, a faint, tiny radiolucent nidus was suspected, measuring less than one centimeter in diameter. However, due to the overlapping osseous shadows of the tibia and fibula on the AP and mortise views, and the dense sclerosis surrounding the lesion, the nidus was not definitively visualized on plain films alone. There was no evidence of periosteal reaction, soft tissue calcification, or joint space narrowing. The syndesmotic clear space and tibiofibular overlap were within normal anatomical limits.
Given the high clinical suspicion for an osteoid osteoma based on the classic history of nocturnal pain relieved by NSAIDs and the equivocal plain radiographs, advanced cross-sectional imaging was mandated.
Magnetic Resonance Imaging of the right ankle without and with intravenous gadolinium contrast was obtained. The MRI is notoriously sensitive but often lacks specificity for osteoid osteoma, frequently leading to misdiagnosis in the uninitiated observer. The T1-weighted images demonstrated a focal area of intermediate signal intensity within the lateral fibular cortex. The fluid-sensitive sequences, including T2-weighted fat-suppressed and Short Tau Inversion Recovery sequences, revealed extensive, ill-defined bone marrow edema extending throughout the distal fibular diaphysis and metaphysis. Furthermore, there was significant adjacent soft tissue edema in the peroneal compartment. This extensive "flare phenomenon" of edema is a classic MRI finding for osteoid osteoma but can easily be misinterpreted as aggressive osteomyelitis, a stress fracture, or even a malignant bone sarcoma like Ewing sarcoma by inexperienced radiologists. The actual nidus was visible as a small, rounded structure showing intense enhancement following gadolinium administration, reflecting its highly vascular nature.
To definitively characterize the osseous architecture of the lesion and confirm the diagnosis, a high-resolution, thin-slice Computed Tomography scan without contrast was performed. The CT scan is universally considered the gold standard imaging modality for the diagnosis and precise anatomical localization of an osteoid osteoma.

The CT scan utilizing sub-millimeter slice thickness (0.625 mm) with multiplanar reformats (axial, coronal, and sagittal) provided definitive diagnostic clarity. The imaging revealed a well-circumscribed, round, radiolucent nidus measuring exactly 8 millimeters in maximum diameter, situated within the lateral cortex of the distal fibula. The nidus contained a central fleck of mineralization, representing the classic "target sign." Surrounding the radiolucent nidus was a profound, thick rim of reactive, dense cortical sclerosis. The precise localization of the nidus via CT is not only diagnostic but absolutely critical for preoperative templating, particularly when considering minimally invasive ablation techniques where millimeter precision dictates the success of the procedure and the safety of adjacent neurovascular structures.
While a Technetium-99m bone scintigraphy was not strictly necessary given the definitive CT findings, it is worth noting that such a study would typically demonstrate the classic "double-density sign," characterized by an intense focal area of radiotracer uptake at the nidus, superimposed on a broader, less intense area of uptake corresponding to the surrounding reactive sclerosis and bone marrow edema. In this case, the combination of clinical history, MRI edema patterns, and the definitive CT demonstration of the cortically based nidus with surrounding sclerosis provided an unequivocal diagnosis of a distal fibula osteoid osteoma.
Differential Diagnosis Analysis
The presentation of chronic, localized ankle pain in a young athlete requires a broad differential diagnosis. The clinical picture can easily be obscured by the patient's high activity level, leading clinicians toward sports-related overuse injuries. A structured comparison of the primary differential diagnoses is essential for accurate clinical reasoning.
| Pathology | Primary Demographic | Clinical Hallmark | Radiographic Findings | Advanced Imaging Characteristics |
|---|---|---|---|---|
| Osteoid Osteoma | Males, 5 to 25 years old | Severe nocturnal pain, dramatic relief with NSAIDs, point tenderness. | Cortical sclerosis, subtle radiolucent nidus (<1.5 cm). | CT: Definitive radiolucent nidus with central calcification. MRI: Extensive bone marrow/soft tissue edema. |
| Stress Fracture | Athletes, military recruits, sudden increase in activity | Activity-related pain, relieved by rest. Insidious onset. | Often normal initially. Later shows periosteal reaction or cortical radiolucent line. | MRI: Bone marrow edema, low signal fracture line on T1/T2. CT: Cortical break or focal periosteal reaction. |
| Osteoblastoma | Males, 10 to 30 years old | Dull, aching pain. Less nocturnal exacerbation. Poor NSAID response. | Expansile, radiolucent lesion >1.5 cm. Less reactive sclerosis. | CT: Larger lesion (>1.5 cm), expansile remodeling, variable mineralization. MRI: Edema present, but less intense than osteoid osteoma. |
| Brodie Abscess | Children and young adults | Insidious pain, intermittent swelling. May have mild systemic symptoms. | Lytic lesion with a sclerotic rim, often metaphyseal. | MRI: "Penumbra sign" (hyperintense inner rim on T1 post-contrast). CT: Intraosseous abscess cavity, potential cortical cloaca. |
| Chronic Tendinopathy | Athletes, repetitive loading sports | Pain localized to tendon trajectory, worse with specific resistance testing. | Usually normal osseous structures. May show calcific tendinitis. | MRI: Tendon thickening, intrasubstance hyperintensity on fluid sequences, peritendinous fluid. Normal bone marrow. |
Differentiating Osteoid Osteoma from Stress Fractures
In a 16-year-old soccer player, a distal fibula stress fracture is the most common misdiagnosis for this presentation. Both conditions present with localized pain and point tenderness over the fibula. However, the pain pattern is the key differentiator. Stress fracture pain is fundamentally mechanical; it worsens with weight-bearing and impact activities and significantly improves with rest. Osteoid osteoma pain is primarily biochemical, driven by local prostaglandin E2 production. Therefore, the pain often peaks at night when the patient is completely at rest and is dramatically relieved by NSAIDs. Furthermore, while both can show bone marrow edema on MRI, the CT scan definitively separates the two by revealing either a cortical fracture line or a discrete nidus.
Differentiating Osteoid Osteoma from Osteoblastoma
Osteoblastoma is histologically identical to osteoid osteoma but differs significantly in its clinical behavior and size. By definition, an osteoid osteoma nidus is less than 1.5 centimeters in diameter, whereas an osteoblastoma is larger than 1.5 centimeters. Osteoblastomas tend to be more locally aggressive, causing expansile bone remodeling. Clinically, the pain associated with osteoblastoma is less likely to be strictly nocturnal and is generally less responsive to salicylates and NSAIDs compared to osteoid osteoma.
Differentiating Osteoid Osteoma from Brodie Abscess
A Brodie abscess, a form of subacute osteomyelitis, can mimic an osteoid osteoma both clinically and radiographically. Both present with localized pain and a lytic lesion surrounded by sclerosis. However, patients with a Brodie abscess may have a history of prior infection, subtle constitutional symptoms, or elevated inflammatory markers (ESR, CRP), though these are not always present. MRI is highly useful here; the "penumbra sign" (a rim of vascularized granulation tissue lining the abscess cavity that enhances on T1 post-contrast images) is highly specific for a Brodie abscess and absent in osteoid osteoma.
Surgical Decision Making and Classification
Osteoid osteomas are classified as benign, bone-forming tumors. Under the Enneking surgical staging system for benign musculoskeletal tumors, an osteoid osteoma is typically classified as a Stage 1 (latent) or Stage 2 (active) lesion. They do not metastasize, but their active production of prostaglandins causes severe, debilitating pain and local inflammatory reactions that can lead to secondary issues such as muscle atrophy, growth disturbances in skeletally immature patients, and joint contractures if located intra-articularly or juxta-articularly.
The decision-making process for the management of an osteoid osteoma involves weighing the natural history of the disease against the risks of intervention.
Non Operative Management
The natural history of an osteoid osteoma is eventual spontaneous "burnout" and resolution, which can take anywhere from two to six years. Therefore, prolonged conservative management with continuous NSAID therapy is a theoretically viable option. However, in a young, highly active athlete, the prospect of taking daily high-dose NSAIDs for several years is fraught with complications, including gastrointestinal toxicity, renal impairment, and the psychological burden of chronic pain. Furthermore, the functional limitations imposed by the pain were actively preventing this patient from participating in soccer, negatively impacting his quality of life. Given the duration of his symptoms (nine months) and his desire to return to competitive sports, non-operative management was deemed inappropriate and definitive intervention was indicated.
Operative and Ablative Interventions
The historical gold standard for treatment was open surgical en bloc resection or unroofing and curettage of the nidus. While highly effective at eliminating the tumor, open surgery in the distal fibula requires a significant incision, periosteal stripping, and cortical bone removal, which creates a stress riser. In an athlete, this necessitates a prolonged period of protected weight-bearing and carries a risk of iatrogenic fracture, potentially requiring prophylactic internal fixation with plates and screws.
In modern orthopedic oncology, minimally invasive percutaneous techniques have largely supplanted open surgery as the primary treatment modality for extra-articular osteoid osteomas. The current gold standard is Computed Tomography Guided Radiofrequency Ablation. RFA utilizes high-frequency alternating current to generate frictional heat within the tissue, inducing coagulative necrosis of the nidus. RFA offers success rates exceeding 90%, comparable to open resection, but with significantly reduced morbidity, minimal bone destruction, and a dramatically accelerated rehabilitation timeline.
Other percutaneous options include cryoablation and Microwave Ablation. Cryoablation utilizes argon gas to create an ice ball, causing cellular death through osmotic dehydration and membrane rupture. It has the advantage of preserving the bone matrix and allowing the ice ball to be visualized on CT, providing excellent intraoperative monitoring of the ablation zone. However, RFA remains the most widely accessible and studied modality.
Specific Considerations for the Distal Fibula
The anatomical location of this specific lesion presented a unique challenge. The distal fibula is highly subcutaneous, with minimal soft tissue coverage. The nidus was located in the lateral cortex. The primary risk of RFA in this location is thermal necrosis of the overlying skin and thermal injury to the superficial peroneal nerve or the sural nerve, depending on the exact trajectory. If the distance between the nidus and the skin or a major nerve is less than 1.0 centimeter, standard RFA carries an unacceptably high risk of collateral thermal damage.
In this patient, careful preoperative CT templating demonstrated that the nidus was approximately 8 millimeters from the epidermal surface. To safely proceed with RFA, advanced thermal protection techniques, such as the injection of a subcutaneous fluid blanket (hydrodissection) using sterile saline or dextrose solution, would be required to physically separate the skin and nerves from the ablation zone. After extensive discussion with the patient and his family regarding the risks of thermal injury versus the morbidity of open surgery, the decision was made to proceed with CT-guided Radiofrequency Ablation with continuous temperature monitoring and hydrodissection, keeping open surgical curettage as a backup option if percutaneous access proved unsafe.
Surgical Technique and Intervention
The procedure was performed in the interventional radiology suite under general anesthesia. General anesthesia is strictly required for RFA of osteoid osteomas, as the heating process is excruciatingly painful and absolute patient immobility is mandatory during the precise targeting and ablation phases.
Patient Positioning and Preparation
The patient was positioned supine on the CT gantry table. A radiolucent bump was placed under the ipsilateral hip to internally rotate the lower extremity, bringing the lateral aspect of the distal fibula into a purely vertical orientation to optimize the trajectory for the drill and ablation probe. A grounding pad was placed on the contralateral thigh to complete the radiofrequency circuit. The right ankle and lower leg were widely prepped with chlorhexidine and draped in a standard sterile orthopedic fashion.
Localization and Hydrodissection
An initial non-contrast limited CT scan of the distal fibula was obtained to localize the nidus. A radiopaque grid was placed over the skin of the lateral ankle to correlate the internal anatomy with the skin surface. The exact entry point, trajectory angle, and depth to the center of the nidus were calculated using the CT workstation software.
Given the proximity of the nidus to the skin surface (8 mm), hydrodissection was performed prior to osseous penetration. A 22-gauge spinal needle was advanced into the subcutaneous tissue directly overlying the lesion. Under intermittent CT fluoroscopy, 15 milliliters of sterile 5% Dextrose in Water (D5W) was injected to create a fluid cushion. D5W is preferred over normal saline as it is non-ionic and does not conduct the radiofrequency current, thereby providing a superior thermal insulation barrier. The fluid injection successfully expanded the distance between the lateral fibular cortex and the skin surface to 18 millimeters, providing a safe margin for thermal ablation.
Osseous Access and Drilling
Following successful hydrodissection, a small 3-millimeter stab incision was made at the marked entry site. A 14-gauge coaxial bone biopsy needle system, consisting of an outer cannula and an inner trocar, was advanced through the soft tissues down to the lateral cortex of the fibula. CT fluoroscopy confirmed the tip of the needle was perfectly centered over the dense reactive sclerosis surrounding the nidus.
Using a sterile battery-powered drill, a 2.0-millimeter Kirschner wire was passed through the cannula and drilled through the sclerotic cortical bone directly into the center of the radiolucent nidus. The dense sclerosis of an osteoid osteoma can be formidable, requiring firm, controlled pressure to penetrate without skiving off the convex fibular surface. Once the K-wire was confirmed to be precisely in the epicenter of the nidus via multiplanar CT imaging, a cannulated drill bit was passed over the wire to enlarge the tract, followed by the advancement of the outer working cannula into the nidus. The inner drill and wire were then removed.
Radiofrequency Ablation Protocol
A single-tine, 17-gauge radiofrequency ablation electrode with a 10-millimeter active uninsulated tip was introduced through the working cannula. The active tip was positioned directly within the 8-millimeter nidus. Final CT images were obtained to verify that the active tip was entirely contained within the bone and not protruding into the surrounding soft tissues or the syndesmotic space.
The electrode was connected to the radiofrequency generator. The ablation protocol was initiated. The generator was set to incrementally increase the temperature at the electrode tip to 90 degrees Celsius. Once the target temperature of 90 degrees was achieved, it was maintained continuously for exactly 6 minutes. This specific temperature-time combination is the established protocol to generate a sphere of coagulative necrosis approximately 1.0 to 1.5 centimeters in diameter, ensuring complete destruction of the nidus while sparing the surrounding normal bone.
During the ablation cycle, cold sterile saline was continuously dripped over the skin surface of the lateral ankle to provide additional external cooling. Following the completion of the 6-minute cycle, the electrode and cannula were removed. The stab incision was closed with a single simple interrupted non-absorbable suture, and a sterile compressive dressing was applied. The patient was awakened from anesthesia and transferred to the post-anesthesia care unit in stable condition.
Post Operative Protocol and Rehabilitation
The primary advantage of Radiofrequency Ablation over open surgical resection is the rapid postoperative recovery profile. Because the structural integrity of the distal fibula is maintained—with only a 2.0-millimeter drill hole created rather than a large cortical window—the risk of postoperative fracture is negligible.
Immediate Postoperative Phase (Weeks 0 to 2)
The patient was discharged home on the same day of the procedure. He was instructed to bear weight as tolerated on the right lower extremity using crutches for support only as needed for comfort. A controlled ankle motion walking boot was provided to reduce stress on the lateral column during the initial inflammatory phase, though its use was strictly for comfort and not for structural protection.
Pain management is a critical indicator of success in the immediate postoperative period. Patients typically experience surgical site pain from the drill tract and soft tissue manipulation for 48 to 72 hours, which is managed with short-acting oral narcotics and acetaminophen. However, the deep, excruciating, nocturnal pain characteristic of the osteoid osteoma should be completely
