Chondromyxoid Fibroma: Comprehensive Orthopedic Case Study & Diagnostic Imaging Analysis
Key Takeaway
Chondromyxoid fibroma diagnosis involves clinical assessment and advanced imaging. Radiographs reveal eccentric lytic lesions with sclerotic rims. CT refines cortical details. MRI shows characteristic T2-hyperintense, lobulated lesions with internal septations. Definitive diagnosis relies on histopathological confirmation via biopsy, crucial for differentiation from other bone tumors, such as low-grade chondrosarcoma.
Patient Presentation and History
A 28-year-old male, otherwise fit and healthy with no significant medical comorbidities, presented to the orthopedic oncology clinic with a 6-month history of insidious onset, progressively worsening right knee pain. The pain was characterized as a dull, deep ache localized specifically to the anteromedial aspect of the proximal tibia. Symptoms were mechanically exacerbated by weight-bearing activities, particularly ascending and descending stairs, and only partially relieved by rest and over-the-counter non-steroidal anti-inflammatory drugs.
The patient reported occasional nocturnal pain that disrupted sleep, a classic red flag symptom necessitating thorough oncologic workup. There was no history of acute, high-energy trauma, although he recalled a minor fall onto the flexed knee approximately 8 months prior, which caused transient discomfort but resolved within a few days. He denied any fevers, chills, night sweats, unexplained weight loss, or other significant constitutional symptoms. His occupation involves light manual labor, which he has found increasingly difficult to perform due to the localized tibial pain. He is a non-smoker and consumes alcohol socially. A comprehensive review of systems was otherwise unremarkable.
In the context of orthopedic oncology, this demographic and clinical presentation—a young adult male with progressive, mechanically exacerbated pain and nocturnal symptoms in a metaphyseal location—raises a high index of suspicion for a primary bone neoplasm. While the age and location heavily favor benign entities, the presence of rest pain necessitates a rigorous diagnostic algorithm to rule out aggressive or malignant pathology.
Clinical Examination Findings
Inspection and Palpation of the Right Lower Extremity
Careful inspection of the right lower extremity revealed mild but distinct fullness over the anteromedial aspect of the proximal right tibia, located approximately 5 cm distal to the medial tibial plateau and immediately medial to the tibial tubercle. There was no overt skin discoloration, erythema, venous engorgement, or visible angular deformity. The quadriceps musculature appeared symmetrical bilaterally, with no significant vastus medialis obliquus atrophy, suggesting the patient had maintained relatively normal biomechanical function until recently.
Palpation elicited highly localized, moderate tenderness directly over the anteromedial proximal tibial metaphysis. A firm, fixed, non-pulsatile mass measuring approximately 4.0 x 3.0 cm was palpable within the underlying osseous structures, deep to the pes anserinus insertion and the medial extensor retinaculum. The overlying skin was normothermic, lacking the calor often associated with aggressive inflammatory processes or highly vascularized high-grade sarcomas. Regional lymph node basins, specifically the inguinal and popliteal regions, were palpated with no evidence of pathological lymphadenopathy.
Range of Motion and Joint Biomechanics
Evaluation of the right knee joint demonstrated full, symmetric active and passive range of motion from 0 to 140 degrees of flexion. However, the patient reported mild discomfort at terminal flexion, likely secondary to increased capsular tension compressing the adjacent metaphyseal expansion. There was no palpable crepitus, and meniscal provocative tests (McMurray, Apley) were negative. Ligamentous testing (Lachman, anterior/posterior drawer, varus/valgus stress) demonstrated a stable joint, ruling out concurrent intra-articular derangement. Patellar tracking was centralized and normal. The ipsilateral hip and ankle joints exhibited full, pain-free range of motion.
Neurological and Vascular Assessment
A comprehensive neurovascular examination of the right lower extremity was normal. Sensation was intact to light touch and pinprick across all dermatomes from L2 through S1. Motor strength was graded 5/5 in all major muscle groups, including hip flexors, knee extensors, knee flexors, ankle dorsiflexors, ankle plantarflexors, and extensor hallucis longus. Deep tendon reflexes (patellar and Achilles) were graded 2+ and were symmetrical to the contralateral limb. No pathological reflexes or signs of upper motor neuron lesions were present.
Vascular assessment revealed robust, symmetrical 2+ dorsalis pedis and posterior tibial pulses. Capillary refill in the digits was brisk, occurring in under two seconds. There were no clinical signs of venous insufficiency or deep vein thrombosis.
Gait Analysis
Observational gait analysis revealed a mild antalgic gait pattern. The patient favored the right lower extremity, demonstrating a shortened stance phase on the affected side to minimize axial loading through the proximal tibia. Despite the antalgia, he remained capable of independent ambulation without the use of assistive devices.
Imaging and Diagnostics
Radiographic Evaluation
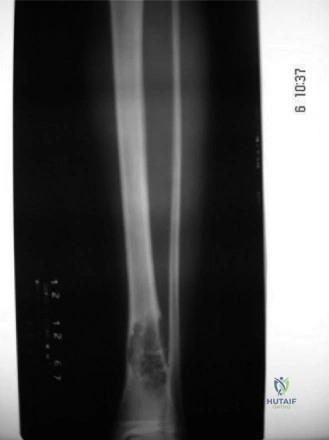
Initial orthogonal anteroposterior and lateral radiographs of the right knee and proximal tibia were obtained. These images revealed an eccentric, geographic lytic lesion located in the metaphysis of the proximal right tibia, extending towards but not crossing the physis (which was closed). The lesion measured approximately 4.5 x 3.0 cm and exhibited a well-defined, lobulated, sclerotic rim, indicative of a slow-growing, benign process that allows the host bone time to mount a reactive osteoblastic response.
Internal trabeculations were prominently featured, imparting a classic "soap bubble" appearance. There was no evidence of an aggressive periosteal reaction (such as a Codman triangle or sunburst pattern), and no frank cortical breach or soft tissue extension was visible on plain films. The joint space appeared well-maintained, and no pathological fracture was evident despite the significant cortical thinning.

Computed Tomography Analysis
A non-contrast computed tomography scan of the right tibia with fine axial cuts and multiplanar reconstructions was performed to further characterize the bony architecture, evaluate cortical integrity, and assist in preoperative templating. This modality confirmed the presence of the eccentric, expansile lytic lesion within the proximal tibial metaphysis, situated immediately adjacent to the anteromedial cortex.
The CT provided exquisite detail of the lesion's internal septations and confirmed the presence of a thick, continuous sclerotic rim at the deep medullary margin. It demonstrated a exceedingly thin but intact cortical shell over the medial aspect of the lesion. While there was no overt cortical breakthrough, the CT highlighted critical thinning of the anteromedial cortex to less than 1 mm in certain areas, increasing the risk of an impending pathological fracture. No aggressive periosteal reaction or extraosseous soft tissue mass was identified. The subchondral bone plate of the medial tibial plateau appeared uninvolved, providing a crucial anatomical buffer for potential surgical reconstruction. CT was critical for evaluating the structural integrity of the subchondral bone plate and mapping the precise three-dimensional geometry of the lesion for preoperative templating.
Magnetic Resonance Imaging
To definitively evaluate the marrow space, assess the exact extent of the lesion, and characterize the internal matrix, a magnetic resonance imaging study with and without intravenous gadolinium contrast was obtained.
The MRI demonstrated a well-circumscribed, lobulated mass in the proximal tibial metaphysis. On T1-weighted sequences, the lesion exhibited intermediate to low signal intensity, consistent with solid cellular and chondroid tissue. On T2-weighted and Short Tau Inversion Recovery sequences, the lesion demonstrated heterogeneously high signal intensity, characteristic of a high water content matrix such as myxoid or chondroid tissue. The peripheral rim and internal septations demonstrated low signal intensity on all sequences, corresponding to the sclerosis seen on radiographs.
Following the administration of gadolinium, the lesion exhibited a distinct peripheral and septal enhancement pattern, with central areas of non-enhancement. This enhancement pattern is highly characteristic of chondromyxoid fibroma, reflecting the vascularized fibrous septa intervening between the relatively avascular chondroid and myxoid lobules. The MRI confirmed the absence of any soft tissue extension, joint effusion, or skip metastases within the tibial diaphysis. Furthermore, the distance from the superior pole of the tumor to the articular cartilage was accurately measured at 12 mm, confirming that joint-sparing intralesional surgery was anatomically feasible.
Histopathological Examination
Given the clinical and radiographic findings, a percutaneous image-guided core needle biopsy was performed prior to definitive surgical intervention to establish a definitive tissue diagnosis and rule out malignant mimics, such as clear cell chondrosarcoma or conventional low-grade chondrosarcoma.
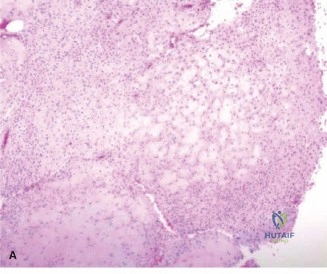
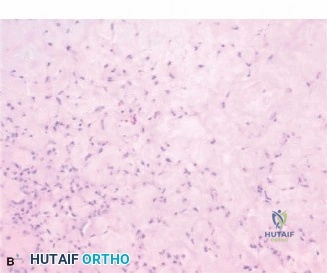
Histological analysis of the core biopsy specimens revealed a distinctly lobulated architecture. The lobules were composed of a biphasic population of cells set within a variably myxoid and chondroid background matrix. At the periphery of these lobules, there was increased cellularity consisting of spindle-shaped and stellate cells. The intervening stroma between the lobules was highly vascularized and fibrous, occasionally containing multinucleated giant cells.
Importantly, while mild cellular atypia and pleomorphism were noted—a common finding in chondromyxoid fibroma that can lead to misdiagnosis—there were no atypical mitotic figures, and no frank anaplasia or permeative growth patterns characteristic of malignancy were observed. The synthesis of the clinical, radiographic, and histopathological data definitively confirmed the diagnosis of a Chondromyxoid Fibroma.
Differential Diagnosis
The presentation of an eccentric, geographic, lytic metaphyseal lesion in a young adult requires a systematic evaluation of several competing pathologies. The differential diagnosis for this specific radiographic and clinical picture includes several benign and potentially locally aggressive bone tumors.
Comparative Pathology Analysis
| Pathology | Typical Age Group | Typical Location | Radiographic Features | Histological Hallmarks |
|---|---|---|---|---|
| Chondromyxoid Fibroma | 10 - 30 years | Metaphysis of long bones (Tibia most common) | Eccentric, geographic, lytic, lobulated, sclerotic rim, "soap bubble" appearance. | Lobular architecture, biphasic cellularity, myxoid/chondroid matrix, hypercellular periphery with stellate cells. |
| Chondroblastoma | 10 - 20 years | Epiphysis or Apophysis | Geographic, lytic, well-defined, often with fine internal calcifications. | Chondroblasts ("cobblestone" appearance), pericellular "chicken-wire" calcification, multinucleated giant cells. |
| Giant Cell Tumor of Bone | 20 - 40 years | Epiphysis/Metaphysis (extends to subchondral bone) | Eccentric, lytic, expansile, lacking a sclerotic rim, often abutting articular surface. | Mononuclear stromal cells mixed with numerous, uniformly distributed osteoclast-like multinucleated giant cells. |
| Aneurysmal Bone Cyst | 10 - 20 years | Metaphysis of long bones, posterior elements of spine | Highly expansile, eccentric, lytic, "blown-out" appearance, fluid-fluid levels on MRI. | Blood-filled cystic spaces lacking endothelial lining, separated by fibrous septa containing giant cells and woven bone. |
| Non-Ossifying Fibroma | 5 - 20 years | Metadiaphysis of long bones | Eccentric, multiloculated, lytic lesion with a thick sclerotic rim, parallel to long axis of bone. | Spindle cells in a storiform (cartwheel) pattern, foamy histiocytes, hemosiderin-laden macrophages, scattered giant cells. |
| Enchondroma | 20 - 50 years | Metaphysis/Diaphysis (Tubular bones of hands/feet common) | Central, geographic, lytic with stippled "rings and arcs" chondroid calcification. | Benign hyaline cartilage lobules, hypocellular, small uniform chondrocytes in lacunae. |
The patient's age and the lesion's location in the proximal tibial metaphysis align closely with Chondromyxoid Fibroma, Giant Cell Tumor, and Aneurysmal Bone Cyst. However, the presence of a thick sclerotic rim makes Giant Cell Tumor less likely, as GCTs typically lack a reactive margin. Aneurysmal Bone Cysts are highly expansile and typically demonstrate fluid-fluid levels on MRI, which were absent in this case. Non-Ossifying Fibromas are typically asymptomatic and discovered incidentally, often resolving by adulthood. Chondroblastoma is primarily epiphyseal, whereas this lesion is distinctly metaphyseal. Enchondromas are usually centrally located and demonstrate classic chondroid calcifications, unlike the eccentric, myxoid-dominant appearance seen here. Therefore, the integrated clinical and radiographic picture strongly pointed toward Chondromyxoid Fibroma, which was subsequently confirmed by biopsy.
Surgical Decision Making and Classification
Enneking Staging System
In the context of the Enneking staging system for benign musculoskeletal tumors, this lesion is classified as a Stage 2 (Active) benign tumor. Stage 1 (Latent) tumors are typically asymptomatic, static, and heavily ossified or sclerotic (e.g., a healed NOF). Stage 3 (Aggressive) tumors demonstrate rapid growth, cortical destruction, soft tissue extension, and severe symptoms (e.g., an aggressive GCT).
This patient's Chondromyxoid Fibroma demonstrates active growth characteristics, evidenced by progressive clinical symptoms, cortical thinning, and a lobulated expansile nature, despite the presence of a reactive sclerotic rim. The active nature of the lesion, combined with the structural compromise of the proximal tibia, dictates the necessity for surgical intervention.
Indications for Surgical Intervention
Non-operative management or observation is not appropriate for an Enneking Stage 2 Chondromyxoid Fibroma. The primary indications for surgical intervention in this specific case included:
1. Symptom Relief: The patient was experiencing progressive, mechanically exacerbated pain and nocturnal rest pain that interfered with his quality of life and occupational duties.
2. Prevention of Pathological Fracture: The anteromedial cortex was critically thinned (<1 mm). Given the proximal tibia's role in major axial load transmission, the patient was at an unacceptably high risk for a pathological fracture, which would significantly complicate reconstruction and compromise joint biomechanics.
3. Local Tumor Control: Chondromyxoid Fibromas have a known propensity for local recurrence if not adequately treated. Surgical eradication is required to halt the progression of the disease.
Adjuvant Selection Rationale
The surgical management of Chondromyxoid Fibroma has evolved. Historically, simple intralesional curettage was associated with unacceptably high local recurrence rates, often reported between 20% and 25%. This high recurrence rate is attributed to the lobulated growth pattern of the tumor, which leaves microscopic nests of neoplastic cells hidden within the scalloped ridges of the surrounding host bone.
To minimize recurrence, the current gold standard is intralesional extended curettage combined with the use of local adjuvants.
* Extended Curettage: Involves the use of a high-speed burr to mechanically remove an additional 1-2 mm of the reactive bony margin, effectively breaking down the bony ridges and eliminating microscopic tumor extensions.
* Chemical/Thermal Adjuvants: Following mechanical burring, adjuvants are utilized to induce necrosis of any remaining microscopic disease. Options include phenol (causes protein coagulation and cellular necrosis), absolute alcohol (often used to neutralize phenol and act as a secondary desiccating agent), hydrogen peroxide (provides mechanical bubbling action to clear debris and mild oxidative necrosis), liquid nitrogen (cryotherapy), or argon beam coagulation.
For this patient, the surgical plan consisted of a wide cortical window, meticulous intralesional extended curettage utilizing a high-speed burr, chemical adjuvant treatment with hydrogen peroxide and pulsatile lavage, followed by reconstruction of the cavitary defect.
Regarding reconstruction, the structural void must be addressed. Options include autograft, allograft, or polymethylmethacrylate bone cement. Given the size of the defect and the proximity to the subchondral plate, PMMA offers distinct advantages. The exothermic reaction of PMMA polymerization provides an additional thermal adjuvant effect, further reducing recurrence risk. Furthermore, PMMA provides immediate structural stability, allowing for early weight-bearing rehabilitation. To prevent thermal injury to the overlying articular cartilage, a small layer of cancellous allograft can be placed at the subchondral interface prior to cementation.
Surgical Technique and Intervention
Preoperative Preparation and Positioning
The patient was brought to the operating theater and placed in the supine position on a radiolucent Jackson table. General endotracheal anesthesia was administered. A thorough preoperative time-out was conducted, verifying the patient identity, surgical site (right proximal tibia), and the procedure. Intravenous prophylactic antibiotics (Cefazolin 2g) were administered 30 minutes prior to incision.
A non-sterile pneumatic tourniquet was applied to the proximal right thigh. The right lower extremity was meticulously prepped and draped in standard sterile orthopedic fashion. The limb was exsanguinated using an Esmarch bandage, and the tourniquet was inflated to 250 mmHg to ensure a bloodless surgical field, which is critical for visualizing the tumor margins and ensuring complete curettage.
Surgical Approach
An anteromedial longitudinal incision was made over the proximal tibia, centered directly over the palpable mass and guided by preoperative imaging. The incision measured approximately 8 cm in length. Dissection was carried sharply through the skin and subcutaneous tissues down to the crural fascia.
The fascia was incised longitudinally. The pes anserinus insertion was identified and carefully retracted posteriorly, while the medial border of the patellar tendon and extensor retinaculum were retracted laterally. This exposure provided direct visualization of the anteromedial aspect of the proximal tibial metaphysis. The overlying periosteum appeared elevated and reactive but was intact. The periosteum was incised and elevated using a Cobb elevator, revealing the thinned, expanded anteromedial cortex.
Extended Curettage and Adjuvant Application
Using a 5 mm drill bit, multiple drill holes were created in a rectangular configuration outlining the planned cortical window over the most expansile portion of the lesion. These holes were connected using a sharp osteotome to remove the cortical lid. The cortical lid was sent for permanent pathological analysis along with the main tumor specimen.
Upon entering the medullary cavity, the macroscopic appearance of the tumor was identifiable as a firm, lobulated, grayish-white mass with a glistening, myxoid quality. Meticulous intralesional curettage was performed using a series of progressively sized straight and angled curettes. The tumor was systematically removed from the periphery towards the center. Special attention was paid to the deep margins and the superior pole adjacent to the subchondral plate. All visible macroscopic tumor was excised and sent for definitive histopathology.
Following complete macroscopic removal, the critical step of extended curettage was initiated. A large, round, high-speed burr was utilized to systematically abrade the entire inner surface of the cavity. The burring was carried out until healthy, bleeding cancellous bone was encountered circumferentially, effectively erasing the sclerotic rim and any microscopic scalloping where tumor lobules might reside. Intraoperative fluoroscopy was utilized intermittently to ensure the burr did not compromise the articular surface or the posterior tibial cortex.
Once mechanical extension was complete, the cavity was subjected to adjuvant treatment. The cavity was repeatedly filled with 3% hydrogen peroxide, allowed to dwell for several minutes to utilize its oxidative and mechanical foaming properties, and then thoroughly suctioned. This was followed by copious irrigation with normal saline using a pulsatile lavage system to remove all cellular debris and residual chemical agents.
Defect Reconstruction
The resulting cavitary defect measured approximately 5.0 x 3.5 x 3.5 cm. To reconstruct the defect and provide immediate biomechanical stability, a hybrid reconstruction technique was employed.
To protect the articular cartilage from the exothermic heat of the bone cement, a 5 mm layer of cancellous bone allograft chips was densely packed into the superior aspect of the cavity, directly against the intact subchondral bone plate.
Subsequently, standard viscosity polymethylmethacrylate bone cement was mixed. During the doughy phase, the PMMA was digitally packed into the remaining cavitary defect, ensuring complete pressurization and interdigitation into the surrounding cancellous trabeculae. The cement was contoured to sit flush with the surrounding anteromedial tibial cortex.
The exothermic polymerization process was allowed to complete, providing both structural integrity and a final thermal adjuvant effect to the cavity walls. Intraoperative fluoroscopy confirmed excellent fill of the defect with the radiopaque cement, with no extravasation into the joint space or soft tissues, and restoration of the mechanical axis.
The wound was copiously irrigated. The periosteum and crural fascia were meticulously closed over the cement mantle using heavy absorbable sutures to prevent soft tissue tethering. The subcutaneous tissues were closed in layers, and the skin was approximated using surgical staples. A sterile compressive dressing and a hinged knee brace locked in extension were applied. The tourniquet was deflated, and distal pulses were confirmed to be bounding.
Post Operative Protocol and Rehabilitation
Early Mobilization Phase
The utilization of PMMA bone cement for defect reconstruction provides immediate structural stability, allowing for an accelerated rehabilitation protocol compared to reconstruction with bone graft alone, which requires prolonged protection for graft incorporation.
Postoperatively, the patient was admitted for 24-hour observation and pain management. Deep vein thrombosis prophylaxis was initiated. The patient was evaluated by physical therapy on postoperative day one. Because the subchondral plate was preserved and the defect was structurally supported by fully polymerized PMMA, the patient was permitted immediate weight-bearing as tolerated on the right lower extremity with the use of crutches for balance.
The hinged knee brace was unlocked to allow active and active-assisted range of motion exercises from 0 to 90 degrees immediately, with progression to full flexion as wound healing permitted. The primary goals of the early phase (Weeks 0-2) were to control edema, achieve full active knee extension, restore quadriceps activation, and ensure primary wound healing. The surgical staples were removed at two weeks postoperatively, at which point the incision was well-healed with no signs of infection.
