Operative Management of Adhesive Capsulitis and Calcific Tendinitis
Key Takeaway
Adhesive capsulitis, or frozen shoulder, is characterized by synovial inflammation and reactive capsular fibrosis leading to profound glenohumeral stiffness. Management ranges from intra-articular corticosteroids and physical therapy to closed manipulation under anesthesia (MUA) and arthroscopic capsular release. This guide details the biomechanics, Lundberg classification, step-by-step surgical techniques including the FEAR sequence for MUA, and evidence-based protocols for treating concurrent calcific tendinitis of the rotator cuff.
ADHESIVE CAPSULITIS (FROZEN SHOULDER)
Much of our contemporary understanding of frozen shoulder, clinically termed adhesive capsulitis, is derived from the seminal work of Neviaser and Lundberg. Neviaser originally coined the term "adhesive capsulitis" to describe a pathognomonic contracted, thickened joint capsule that appears to be drawn tightly around the humeral head. This condition is characterized by a relative absence of synovial fluid and profound chronic inflammatory changes localized within the subsynovial layer of the capsule.
Current histological and biochemical evidence suggests that the underlying pathological cascade in adhesive capsulitis begins with robust synovial inflammation, which subsequently triggers reactive capsular fibrosis. At a molecular level, an upregulation of cytokines (such as TGF-β and PDGF) and matrix metalloproteinases (MMPs) has been heavily implicated in driving fibroblastic proliferation and the deposition of Type III collagen. Despite these advances in understanding the fibrotic cascade, the initial triggering event that precipitates this inflammatory response remains elusive.
Epidemiology and Risk Factors
The incidence of frozen shoulder in the general population is approximately 2%. However, several systemic and local conditions are associated with a markedly increased incidence. The typical demographic profile involves individuals between the ages of 40 and 70, with women comprising approximately 70% of the affected population.
Recognized risk factors include:
* Diabetes Mellitus: Patients with diabetes have a five-fold increased risk of developing adhesive capsulitis. Furthermore, diabetic frozen shoulders tend to be more recalcitrant to conservative management.
* Endocrine Disorders: Hyperthyroidism and hypothyroidism are well-documented systemic risk factors.
* Cardiovascular and Neurological Events: Stroke, myocardial infarction, and the presence of autoimmune diseases.
* Cervical Spine Pathology: Cervical disc disease frequently correlates with shoulder stiffness.
* Immobilization: Prolonged immobilization, regardless of the diverse underlying causes, is arguably the most significant mechanical factor related to the development of the condition.
Clinically, 20% to 30% of affected individuals will subsequently develop adhesive capsulitis in the contralateral shoulder. Interestingly, the condition rarely recurs in the same shoulder once fully resolved.
Classification Systems
Lundberg developed a widely accepted classification system for frozen shoulder based on the presence or absence of an inciting event:
- Primary Adhesive Capsulitis: Idiopathic in nature. These are frozen shoulders in patients who report no inciting event and present with no abnormality on clinical examination (other than profound loss of active and passive motion) or plain radiographs.
- Secondary Adhesive Capsulitis: Associated with a known precipitant. This includes patients with prior traumatic injuries or those developing stiffness following a surgical procedure. While postoperative stiffness is technically a secondary frozen shoulder, its clinical course and response to treatment can differ significantly from primary idiopathic cases.
Clinical Course and Nonoperative Management
The natural history of adhesive capsulitis is classically described in three overlapping phases: the painful "freezing" phase, the stiff "frozen" phase, and the recovery "thawing" phase. Although historically considered a self-limiting condition that resolves in 12 to 18 months, long-term follow-up studies reveal that many patients experience residual, albeit often asymptomatic, motion deficits. Furthermore, patients frequently underestimate their objective motion loss.
Early intervention is paramount. The duration of symptoms before medical care is sought directly correlates with the speed and completeness of recovery. Dominant shoulder involvement has been reported as predictive of a favorable outcome, whereas specific occupational demands do not show statistical significance in altering the natural history.
Initial treatment is strictly nonoperative, emphasizing the control of pain and inflammation:
* Pharmacotherapy: Nonsteroidal anti-inflammatory drugs (NSAIDs) and oral corticosteroids.
* Intra-articular Injections: Corticosteroid injections have proven highly beneficial, particularly when administered in the early freezing phase (Phase 1) or early frozen phase (Phase 2).
* Physical Therapy: Supervised physical therapy focusing on gentle, progressive passive and active range-of-motion exercises. Transcutaneous electrical nerve stimulation (TENS) and ultrasound may serve as helpful adjuncts.
* Hydrodilatation: Distention arthrography can mechanically stretch the capsule and disrupt early adhesions.
Clinical Pearl: During early physical therapy, aggressive abduction should be avoided. Forcing abduction in a stiff shoulder leads to obligate superior translation of the humeral head, causing severe subacromial impingement and exacerbating pain. Joint motion must become supple before terminal abduction is pursued.
Operative Management: Indications and Patient Selection
Many patients find the 12- to 18-month natural history unacceptable and request active intervention. With meticulous patient selection, significant improvement can be obtained in approximately 70% of patients undergoing procedural intervention.
Surgical or manipulative intervention is indicated for patients who have failed 3 to 6 months of dedicated conservative management and exhibit recalcitrant stiffness. The primary options include Closed Manipulation Under Anesthesia (MUA) and Arthroscopic Capsular Release.
Closed Manipulation Under Anesthesia (MUA)
Closed manipulation remains a highly efficacious first-line procedural intervention. Failures of MUA are typically related to the patient's inability to maintain postoperative motion due to pain, rather than intraoperative technical failures.
Surgical Warning: Complications during closed manipulation can be catastrophic. The proximal humerus may be fractured (often a spiral fracture of the surgical neck), or glenohumeral dislocation may occur. MUA must be strictly avoided in patients with osteopenia, osteoporosis, or recently healed fractures. In such patients, arthroscopic release is the mandatory alternative.
Surgical Technique: Closed Manipulation (The FEAR Sequence)
- Anesthesia and Positioning: Administer general anesthesia supplemented with an interscalene regional block. The block is critical not only for intraoperative muscle relaxation but for immediate postoperative pain control, allowing for aggressive early rehabilitation.
- Scapular Stabilization: The surgeon must firmly stabilize the scapula. Failure to fix the scapula will result in scapulothoracic motion masquerading as glenohumeral release, leading to an incomplete manipulation.
- Lever Arm Mechanics: Always use a short lever arm. Grasp the humerus as proximally as possible (near the axilla) rather than at the elbow or forearm. This minimizes the torque applied to the humeral diaphysis, drastically reducing the risk of iatrogenic fracture.
- The FEAR Sequence: Execute the manipulation in a safe, stepwise sequence to systematically rupture capsular adhesions:
- F - Flexion: Elevate the arm in the sagittal plane to stretch the posterior and inferior capsule.
- E - Extension: Extend the arm to release the anterior capsular structures.
- A - Abduction and Adduction: Carefully abduct the arm while stabilizing the scapula to release the inferior capsule (axillary pouch).
- R - Rotation (External and Internal): Perform external rotation in adduction (releasing the coracohumeral ligament and superior capsule), followed by external rotation in 90 degrees of abduction (releasing the middle and inferior glenohumeral ligaments). Finally, perform internal rotation.
- Prognostic Indicators: An audible and palpable "tearing" or release of adhesions is a highly favorable prognostic sign, indicating successful capsular rupture.
Arthroscopic Capsular Release
Arthroscopic release is the gold standard for patients in whom closed manipulation fails, those with prolonged recalcitrant adhesive capsulitis, or those with contraindications to MUA (e.g., osteopenia). Marked improvement is reported in 80% to 90% of patients undergoing this procedure.
Key Steps in Arthroscopic Release:
1. Diagnostic Arthroscopy: Establish a standard posterior portal. In severe adhesive capsulitis, the joint space is severely contracted, making initial trocar insertion challenging. A blunt trocar must be used to avoid iatrogenic chondral injury.
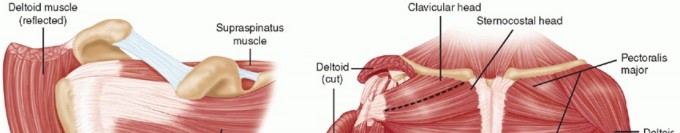
2. Rotator Interval Release: Establish an anterior working portal. The release begins at the rotator interval. The coracohumeral ligament (CHL) and the superior glenohumeral ligament (SGHL) are resected. The CHL is the primary restraint to external rotation in adduction; its complete release is paramount.
3. Anterior Capsulotomy: Using an electrothermal device or arthroscopic scissors, the capsule is released just lateral to the glenoid labrum, progressing inferiorly through the middle glenohumeral ligament (MGHL) and the anterior band of the inferior glenohumeral ligament (IGHL).
4. Inferior Capsulotomy: The release continues through the axillary pouch (6 o'clock position).
> Pitfall: The axillary nerve lies in close proximity (often within millimeters) to the inferior capsule. Thermal devices must be used with extreme caution in the 6 o'clock position. Many surgeons prefer blunt sweeping or cold-cutting instruments for the inferior release to prevent thermal neuropraxia.
5. Posterior Capsulotomy: If internal rotation remains restricted, the arthroscope is moved to the anterior portal, and the posterior capsule is released from the 6 o'clock position up to the 9 o'clock (or 3 o'clock) position.
If arthroscopic release fails to relieve symptoms, open release of contractures has historically been recommended, emphasizing the open release of the coracohumeral ligament and reestablishment of the interval between the supraspinatus and subscapularis. However, with modern arthroscopic techniques, open release is rarely required today.
Postoperative Rehabilitation
Rehabilitation following either MUA or arthroscopic release must be immediate and aggressive. The interscalene block provides a window of analgesia during which continuous passive motion (CPM) or therapist-directed passive stretching should be initiated. If an iatrogenic dislocation occurs after closed reduction, rehabilitation should still be aggressive, but the abducted, externally rotated position must be temporarily avoided to allow anterior capsular healing.
CALCIFIC TENDINITIS OF THE ROTATOR CUFF
Calcific tendinitis is a profoundly painful, largely self-limited disorder of the rotator cuff characterized by the infiltration and deposition of calcium hydroxyapatite crystals within the tendon substance.
Epidemiology and Pathogenesis
This condition affects approximately 10% of the general population, typically presenting in individuals older than 30 years. Women are affected more frequently than men, and up to 10% of affected patients will develop bilateral deposits. While many individuals with radiographic evidence of calcific deposits remain entirely asymptomatic, the condition can cause acute, excruciating pain during specific phases of the disease process.
The most common anatomical site of occurrence is within the supraspinatus tendon, specifically localizing 1.5 to 2 cm proximal to the tendon's insertion on the greater tuberosity.
Although the clinical course and histological changes of calcific tendinitis are well delineated, its precise etiology remains a subject of debate:
* Vascular Theory: Early hypotheses suggested a vascular etiology. Microangiographic studies identified an area of hypovascularity near Codman’s “critical zone,” just proximal to the supraspinatus insertion. It was postulated that this hypoperfusion initiates local tissue hypoxia, leading to fibrocartilaginous metaplasia, subsequent tendon fiber degeneration, and eventual dystrophic calcification.
* Cell-Mediated Theory (Uhthoff's Cycle): More recent histological studies contradict the hypovascular theory, demonstrating that the supraspinatus, including the critical zone, is well supplied by a robust vascular anastomosis. Uhthoff proposed a cell-mediated, multiphasic process:
1. Precalcific Phase: Fibrocartilaginous metaplasia of tenocytes.
2. Calcific Phase: Subdivided into formative (chalk-like calcium deposition), resting, and resorptive phases. The resorptive phase is characterized by intense vascular proliferation, macrophage infiltration, and edema. This resorptive phase is clinically the most painful period for the patient.
3. Postcalcific Phase: Fibroblasts remodel the defect, restoring normal tendon architecture.
Clinical Presentation and Diagnosis
Patients typically present with an insidious onset of shoulder pain that may acutely exacerbate, often mimicking an acute infection or gout attack due to the severity of the pain during the resorptive phase. Mechanical impingement symptoms may also occur due to the increased volume of the calcific deposit within the subacromial space.
Diagnosis is readily confirmed via standard orthogonal plain radiographs (Anteroposterior, Scapular Y, and Axillary views). The deposits appear as dense, well-circumscribed opacities during the formative phase, and become fluffy, ill-defined, and cloud-like during the painful resorptive phase. Ultrasound is highly sensitive for localizing the deposit and assessing its consistency.
Management Strategies
Because calcific tendinitis is ultimately a self-limiting condition, initial management is conservative.
Conservative Treatment:
* NSAIDs and analgesics.
* Subacromial corticosteroid injections to mitigate the intense inflammatory response during the resorptive phase.
* Physical therapy to maintain glenohumeral motion and prevent secondary adhesive capsulitis.
Minimally Invasive and Operative Treatment:
If symptoms are refractory to conservative measures for more than 3 to 6 months, or if the acute pain is intractable, intervention is warranted.
- Ultrasound-Guided Barbotage (Lavage): Under local anesthesia, a spinal needle is introduced into the calcific deposit under direct ultrasound guidance. The deposit is repeatedly punctured (needling) and lavaged with saline to aspirate the calcium, followed by a corticosteroid injection. This technique is highly effective for soft, toothpaste-like deposits in the resorptive phase.
- Arthroscopic Excision: For chronic, hard, chalk-like deposits that fail barbotage, arthroscopic removal is indicated.
- Technique: The subacromial space is viewed arthroscopically. The deposit often presents as a hyperemic, strawberry-like lesion on the bursal surface of the rotator cuff. A spinal needle is used to localize the exact center of the calcium deposit. A longitudinal incision is made in the tendon in line with its fibers. A curette or arthroscopic shaver is used to meticulously evacuate the calcium.
- Tendon Repair: If the evacuation results in a significant structural defect in the rotator cuff tendon (typically greater than 50% of the tendon thickness), a side-to-side or suture anchor repair of the tendon is performed to restore biomechanical integrity and prevent tear propagation.
Clinical Pearl: Complete excision of every microscopic calcium particle is not necessary and can lead to excessive iatrogenic tendon damage. The goal of arthroscopic surgery is decompression of the deposit to stimulate the local healing response and relieve the mechanical pressure within the tendon.
📚 Medical References
- adhesive capsulitis of the shoulder in a high-risk population, QJM 198:191, 1981.
- Budoff JE, Nirschl RP, Guidi EJ: Debridement of partial-thickness tears of the rotator cuff without acromioplasty: long-term follow-up and review of the literature, J Bone Joint Surg 80A:733, 1998.
- Budoff JE, Rodin D, Ochial D, et al: Arthroscopic rotator cuff debridement without decompression for the treatment of tendinosis, Arthroscopy 21:1081, 2005.
- Bulgen DY, Binder A, Hazleman BL, et al: Immunological studies in frozen shoulder, J Rheumatol 9:893, 1982.
- Bulgen DY, Binder AI, Hazleman BL, et al: Frozen shoulder: prospective clinical study with an evaluation of three treatment regimens, Ann Rheum Dis 43:353, 1984.
- Burkhart SS: Partial repair of
You Might Also Like