Pediatric Neurofibromatosis Type 1 & Congenital Pseudarthrosis of the Tibia: A Case Study
Key Takeaway
Congenital pseudarthrosis of the tibia (CPT) is a rare dysplastic bone condition often associated with Neurofibromatosis Type 1 (NF1). Diagnosis involves characteristic clinical signs like café-au-lait macules and Lisch nodules, coupled with specific radiographic evidence of anterolateral tibial bowing and an 'hourglass' deformity, particularly in pediatric patients with a limp.
Patient Presentation and History
A 3-year-old male presented to our pediatric orthopedic clinic with a chief complaint of progressive right lower extremity bowing and a noticeable limp. The deformity had been insidious in onset, first observed by his parents around 18 months of age, and had gradually worsened. He ambulated independently but with a noticeable Trendelenburg gait and occasional tripping. There was no history of trauma, specific injury, or acute pain episodes. The parents also reported the presence of multiple birthmarks on his torso and extremities since infancy.
His birth history was uncomplicated, full-term, via spontaneous vaginal delivery. Developmental milestones were met within normal limits. Past medical history was significant for a previously diagnosed mild scoliosis, currently managed with observation. There was no known family history of similar orthopedic conditions or genetic syndromes. On further questioning, the parents noted that the patient had undergone an ophthalmology evaluation at 2 years of age due to suspicious lesions on his iris, which were identified as Lisch nodules. No specific genetic testing had been performed prior to this presentation.
To fully contextualize this presentation, it is imperative to understand the underlying molecular pathology associated with Neurofibromatosis Type 1. The condition is an autosomal dominant genetic disorder caused by a mutation in the NF1 gene located on chromosome 17q11.2. This gene is responsible for encoding neurofibromin, a cytoplasmic protein that acts as a negative regulator of the Ras signaling pathway. In the absence of functional neurofibromin, Ras activity is constitutively upregulated, leading to abnormal cellular proliferation and differentiation. In the skeletal system, this dysregulation profoundly affects osteoblast and osteoclast function. Current literature suggests that the localized skeletal dysplasias seen in NF1, such as anterolateral bowing and subsequent congenital pseudarthrosis of the tibia, are the result of a "second hit" somatic mutation within the periosteal cells of the affected limb. This creates a localized environment characterized by highly cellular, fibromatous tissue that inhibits normal endochondral ossification and promotes aggressive localized osteolysis.
The insidious progression of the anterolateral bowing in this patient is pathognomonic for the pre-pseudarthrotic phase of this disease. The mechanical axis deviation caused by the bowing, coupled with the structurally inferior woven bone produced by the dysplastic periosteum, places the tibia at an extraordinarily high risk for spontaneous fracture. Once a fracture occurs in this biological environment, spontaneous union is virtually impossible without aggressive, combined mechanical and biological surgical intervention.
Clinical Examination Findings
General Assessment
The patient was an alert, cooperative 3-year-old male. His vital signs were stable, and he appeared well-nourished. Height and weight were at the 50th percentile for age.
Musculoskeletal Inspection
A significant anterolateral bowing deformity of the right tibia was evident, particularly in the distal and middle thirds. The apex of the deformity was prominent, with overlying skin appearing thin and taut. A subtle leg length discrepancy was noted, with the right lower extremity appearing approximately 1-1.5 cm shorter when measured via the block method to level the pelvis.
Multiple café-au-lait macules were observed over the trunk, bilateral upper extremities, and left lower extremity. Six macules greater than 0.5 cm in their greatest dimension were clearly identified, fulfilling a primary diagnostic criterion for Neurofibromatosis Type 1 established by the National Institutes of Health. There was no axillary or inguinal freckling appreciated at this time. No cutaneous or plexiform neurofibromas were palpated or visible.
The patient ambulated with a right-sided limp, consistent with a Trendelenburg pattern and a subtle circumduction of the right lower limb. This gait adaptation is a direct biomechanical compensation for the leg length discrepancy and the significant mechanical axis deviation induced by the anterolateral diaphyseal bowing. The center of rotation of angulation was clinically localized to the junction of the middle and distal thirds of the tibial shaft.
Palpation and Stability
Palpation over the anterolateral bowing of the right tibia revealed a firm, non-tender bony prominence. There was no gross instability or crepitus indicative of an acute fracture or frank pseudarthrosis, but the bone felt distinctly irregular and thickened at the apex of the curve. This thickening represents the hypertrophic, dysplastic periosteum—often termed a fibrous hamartoma—that aggressively constricts the underlying cortex, leading to localized ischemia and subsequent osteoclastic resorption.
Neurovascular Assessment
Distal pulses, including the dorsalis pedis and posterior tibial arteries, were symmetrical and strong bilaterally. Capillary refill was brisk in all digits, occurring in under two seconds. Sensation to light touch was intact throughout the right foot and ankle, following expected dermatomal distributions. Motor strength was graded 5/5 in all major muscle groups of the right lower extremity, including tibialis anterior, extensor hallucis longus, gastrocnemius, and the peroneal complex. There was no evidence of foot drop or peroneal nerve palsy, which can occasionally be associated with severe proximal fibular deformities or plexiform neurofibromas in the popliteal fossa.
Range of Motion and Joint Mechanics
Full, pain-free range of motion was demonstrated in the bilateral hips and knees. Ankle dorsiflexion and plantarflexion were full and symmetrical. Subtle stiffness in the subtalar joint was noted on the right, but global ankle range of motion was preserved. It is critical to carefully assess the ankle and subtalar joints in these patients, as the distal tibial deformity often induces compensatory valgus at the ankle joint, and prolonged bracing or altered biomechanics can lead to rigid hindfoot deformities over time.
Systemic Examination
Gross neurological examination was unremarkable, with physiological deep tendon reflexes and negative upper motor neuron signs. The historical finding of Lisch nodules was consistent with an NF1 diagnosis. No proptosis or signs of optic pathway glioma were observed on gross inspection. Cardiac and abdominal examinations were within normal limits.
Imaging and Diagnostics
Radiographic Evaluation
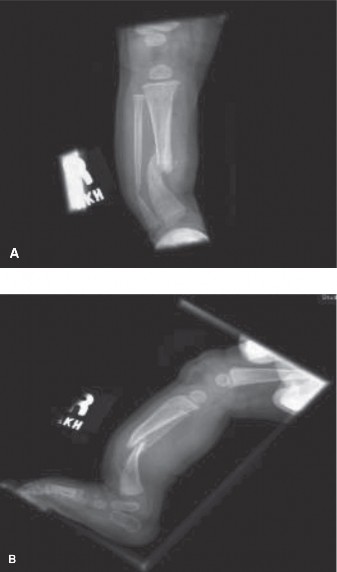
Initial anteroposterior and lateral radiographs of the right tibia and fibula were obtained. These revealed significant anterolateral bowing of the right tibia and fibula, centered at the junction of the middle and distal thirds.

The radiographic analysis demonstrated profound diaphyseal narrowing at the apex of the deformity. The medullary canal was completely obliterated by dense sclerotic bone at the apex of the bow, a classic radiographic hallmark of impending congenital pseudarthrosis of the tibia. According to the Crawford Classification system, this presentation aligns with a Type II deformity, characterized by anterolateral bowing with medullary sclerosis, though early cystic changes were visible on the lateral projection, suggesting a transition toward a Type III lesion.
The fibula also demonstrated concomitant anterolateral bowing, which is a critical prognostic factor. Fibular involvement dictates the necessity of addressing both bones during surgical reconstruction to prevent recurrent deformity and ensure adequate stabilization of the ankle mortise. The mechanical axis of the lower extremity was significantly deviated laterally and anteriorly, resulting in a severe malalignment that exponentially increases the bending moments across the dysplastic tibial diaphysis during the stance phase of gait.
Advanced Cross Sectional Imaging
To further delineate the extent of the periosteal dysplasia and to aid in preoperative templating, a non-contrast Magnetic Resonance Imaging scan of the right lower extremity was ordered. The MRI is invaluable in these cases, not necessarily for the bony architecture, which is well-visualized on plain films, but for the characterization of the surrounding soft tissue envelope.
The T1 and T2-weighted axial and sagittal sequences revealed a distinct, circumferential collar of thickened, intermediate-to-low signal intensity soft tissue encasing the apex of the tibial deformity. This tissue represents the fibromatous hamartoma. The MRI confirmed that this dysplastic tissue extended several centimeters proximal and distal to the radiographic apex of the deformity. Understanding the exact proximal and distal extent of this hamartoma is a mandatory prerequisite for surgical intervention, as complete resection of this biologically hostile tissue down to healthy, bleeding periosteum is the absolute cornerstone of achieving a successful osseous union. Failure to resect this tissue entirely results in an unacceptably high rate of recurrent pseudarthrosis.
Preoperative Templating
Orthogonal full-length standing radiographs were utilized for precise preoperative templating. The center of rotation of angulation was mapped. The true leg length discrepancy was calculated radiographically to be 18 millimeters, factoring in the shortening induced by the angular deformity. Templating focused on determining the exact osteotomy levels required to achieve acute correction of the mechanical axis, sizing the intramedullary device, and mapping the wire and half-pin trajectories for the external fixation construct.
Differential Diagnosis
When evaluating a pediatric patient with lower extremity bowing, it is crucial to differentiate between physiologic conditions, metabolic bone diseases, and localized dysplasias. The anterolateral direction of the bowing is highly specific and immediately narrows the differential diagnosis to pathological entities, as opposed to physiologic bowing which is typically varus, or posteromedial bowing which carries a completely different natural history.
| Pathological Entity | Clinical Presentation | Radiographic Findings | Natural History and Prognosis |
|---|---|---|---|
| Congenital Pseudarthrosis of the Tibia | Anterolateral bowing, associated with NF1 in 50-80% of cases. LLD present. High risk of spontaneous fracture. | Anterolateral angulation, medullary sclerosis, cystic changes, diaphyseal narrowing. Fibular involvement common. | Poor without aggressive surgery. High rate of refracture. Requires combined biological and mechanical surgical intervention. |
| Posteromedial Bowing of the Tibia | Posteromedial angulation noted at birth. Often associated with calcaneovalgus foot deformity. | Posteromedial apex bowing. Thickened cortex on the concave side. Normal medullary canal. | Excellent for the angular deformity, which typically remodels spontaneously. However, LLD persists and often requires epiphysiodesis or lengthening later in childhood. |
| Osteogenesis Imperfecta | Multiple recurrent fractures, blue sclerae, dentinogenesis imperfecta, positive family history. Global bowing. | Osteopenia, multiple healed fractures, thin cortices, "bowed" or "crumpled" long bones depending on Sillence type. | Chronic fragility. Managed with bisphosphonates and telescopic intramedullary rodding. Healing potential of the bone itself is generally normal, unlike CPT. |
| Fibrous Dysplasia | Can be monostotic or polyostotic. Associated with McCune-Albright syndrome if accompanied by café-au-lait spots (coast of Maine) and endocrinopathy. | "Ground glass" appearance of the medullary canal. Expansile lesions. Shepherd's crook deformity in the proximal femur. | Variable. Lesions may stabilize after puberty. Surgery indicated for impending fractures or severe deformity. Does not typically present as isolated anterolateral tibial bowing in infancy. |
Surgical Decision Making and Classification
The management of anterolateral bowing of the tibia in the setting of Neurofibromatosis Type 1 is one of the most formidable challenges in pediatric orthopedic trauma and reconstructive surgery. The decision-making process hinges on the natural history of the disease, the specific Crawford classification of the deformity, and the profound biological deficit inherent to the dysplastic periosteum.
Classification Framework
The Crawford Classification is the standard framework utilized to stage these lesions and guide intervention:
* Type I: Anterior bowing with increased cortical density and a normal medullary canal.
* Type II: Anterior or anterolateral bowing with a sclerotic, obliterated medullary canal.
* Type III: Anterolateral bowing with cystic changes or a pre-fracture state.
* Type IV: Frank pseudarthrosis with complete discontinuity of the tibia and fibula.
Our patient presented with a lesion transitioning from Type II to Type III. The medullary canal was completely obliterated by sclerosis, and early cystic changes were evident.
Rationale for Operative Intervention
Historically, there was debate regarding prophylactic surgery for pre-pseudarthrotic lesions versus waiting for an inevitable fracture to occur. Non-operative management consists solely of total contact bracing (such as a clamshell ankle-foot orthosis) to protect the limb. However, bracing does not alter the underlying biological pathology, nor does it correct the mechanical axis deviation. Given the severe diaphyseal narrowing and the degree of angulation in this patient, fracture was deemed imminent.
Once a fracture occurs through a Crawford Type II or III lesion, the local biological environment—dominated by the highly cellular, osteoclast-stimulating fibromatous hamartoma—prevents normal fracture callus formation. The resulting pseudarthrosis is notoriously recalcitrant to standard fracture care. Therefore, the contemporary consensus among pediatric limb reconstruction specialists strongly favors prophylactic, aggressive surgical intervention for impending fractures with severe deformity.
The goals of operative intervention are multifaceted:
1. Complete eradication of the dysplastic periosteal hamartoma.
2. Restoration of the mechanical axis of the lower extremity.
3. Establishment of absolute mechanical stability to facilitate osseous union.
4. Biological augmentation to overcome the local osteogenic deficit.
5. Prevention of refracture, which requires lifelong intramedullary protection.
We elected to proceed with a comprehensive reconstruction utilizing the principles popularized by Paley and others: radical resection of the hamartoma, acute deformity correction, intramedullary stabilization with a telescopic rod, application of a circular external fixator for compression, and massive autologous bone grafting to create a tibiofibular cross-union.
Surgical Technique and Intervention
Patient Positioning and Anesthesia
The patient was placed supine on a radiolucent Jackson flat table. General endotracheal anesthesia was administered. A thorough preoperative timeout was conducted. The bilateral lower extremities and the bilateral iliac crests were prepped and draped in a standard sterile fashion to allow for autologous bone graft harvesting and unrestricted access to the entire right lower extremity. A sterile tourniquet was applied to the proximal right thigh but was not inflated initially to allow for accurate identification of vascular structures and assessment of periosteal bleeding during the resection phase.
Surgical Approach and Hamartoma Resection
A longitudinal, anterolateral incision was made over the right leg, centered directly over the apex of the deformity. The incision extended sufficiently proximal and distal to expose healthy, normal-appearing bone. Careful subcutaneous dissection was performed to protect the superficial peroneal nerve, which was identified, mobilized, and gently retracted laterally.
The anterior compartment fascia was incised. The extensor musculature was elevated off the interosseous membrane and the lateral aspect of the tibia. Upon exposing the tibia, the fibromatous hamartoma was immediately evident. It presented as a dense, grayish, avascular-appearing fibrous collar tightly adherent to the underlying sclerotic bone.
The most critical step of the procedure commenced: the radical en bloc resection of this hamartomatous tissue. Using sharp dissection, curettes, and a periosteal elevator, the dysplastic periosteum was meticulously stripped from the tibia and the adjacent fibula. The resection was carried proximally and distally until healthy, bleeding periosteum and normal cortical bone were encountered. The underlying sclerotic, avascular bone at the apex of the deformity was then resected using an oscillating saw under continuous saline irrigation to prevent thermal necrosis. The resection of the tibial apex created a segmental defect of approximately 1.5 centimeters, which simultaneously allowed for acute correction of the angular deformity and shortening of the limb to relieve tension on the neurovascular bundle during realignment.
Intramedullary Fixation Strategy
Following the resection, the medullary canals of the proximal and distal tibial segments were completely obliterated by dense sclerotic bone. Using a combination of high-speed burrs and sequentially larger rigid reamers, the medullary canals were painstakingly opened and expanded. It is vital to ream until healthy, bleeding cancellous bone is encountered to ensure adequate vascularity for subsequent union.
To provide long-term internal splintage and prevent the high risk of refracture associated with CPT, a Fassier-Duval telescopic intramedullary rod was selected. The male component of the rod was introduced retrogradely through the distal tibial segment, traversing the distal tibial physis, and exiting through the plantar aspect of the foot. The proximal tibial segment was then aligned, and the female component of the rod was advanced antegrade from the proximal tibial metaphysis, carefully avoiding the proximal tibial physis. The two components were coupled at the osteotomy site. The rod provides excellent resistance to bending forces while allowing for continued longitudinal growth of the tibia.
External Fixation Application
While the intramedullary rod provides excellent alignment and bending stiffness, it does not provide adequate axial compression or rotational control required for healing a pseudarthrosis. Therefore, a multi-plane circular external fixator (Ilizarov frame) was applied to supplement the internal fixation.
Two rings were utilized: one proximal to the osteotomy and one distal. The proximal ring was secured to the proximal tibial segment using two tensioned 1.5 mm smooth wires and one 4.0 mm hydroxyapatite-coated half-pin. The distal ring was similarly secured to the distal tibial segment, ensuring that the wires and pins did not interfere with the intramedullary rod. The rings were connected with threaded rods. Under fluoroscopic guidance, acute axial compression was applied across the osteotomy site via the external fixator. The compression forces the remaining healthy bone ends into intimate contact, maximizing the mechanical environment for osteogenesis.
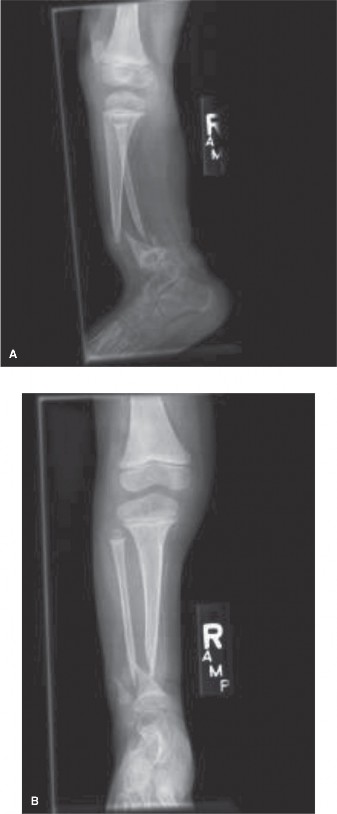
Biologic Augmentation and Cross Union
Given the biologically hostile environment of a NF1-associated pseudarthrosis, mechanical stability alone is insufficient. Massive biologic augmentation is mandatory.
Simultaneous with the tibial preparation, a secondary surgical team harvested a substantial volume of autologous cancellous bone graft from the bilateral anterior and posterior iliac crests.
To maximize the surface area for healing and to create a robust mechanical strut, we employed the tibiofibular cross-union technique. The medial cortex of the fibula and the lateral cortex of the tibia at the level of the osteotomy were decorticated using a high-speed burr to expose bleeding bone. The massive volume of autologous iliac crest bone graft was then meticulously packed into the interosseous space between the tibia and fibula, enveloping the osteotomy site and bridging the two bones. This technique aims to create a solid synostosis between the tibia and fibula, effectively increasing the cross-sectional area of the bone and dramatically enhancing the biomechanical strength of the reconstructed segment. Recombinant human Bone Morphogenetic Protein-2 (rhBMP-2) was utilized off-label, applied to an absorbable collagen sponge, and layered over the autograft to further hyper-stimulate local osteoinduction.
The wound was irrigated copiously. Due to the massive bone grafting, primary closure of the fascia was not attempted to prevent compartment syndrome. The subcutaneous tissues and skin were closed in layers with interrupted sutures. Sterile dressings were applied around the external fixator pin sites.
Post Operative Protocol and Rehabilitation
The postoperative management of congenital pseudarthrosis of the tibia requires extreme vigilance, meticulous pin care, and a long-term commitment to mechanical protection of the limb.
Immediate Postoperative Phase (0-6 Weeks)
The patient was admitted to the pediatric intensive care unit for 24 hours for neurovascular monitoring and pain control. Intravenous antibiotics (Cefazolin) were administered for 24 hours postoperatively. The limb was elevated to minimize edema.
Pin site care was initiated on postoperative day two, utilizing a half-strength hydrogen peroxide and normal saline solution, followed by the application of sterile gauze. The parents were extensively trained in pin site management prior to discharge. The patient was kept strictly non-weight-bearing on the operative extremity. Aggressive physical therapy was initiated to maintain range of motion in the knee and the toes, although ankle motion was restricted by the frame and the trans-articular nature of the distal fixation.
Intermediate Phase (6-12 Weeks)
At the six-week postoperative visit, clinical and radiographic evaluations were performed. Radiographs were scrutinized for early signs of callus formation and incorporation of the massive autologous bone graft into the tibiofibular synostosis. Assuming adequate early consolidation, the patient was transitioned to partial weight-bearing (approximately 20% of body weight) utilizing a custom-modified walker. The micro-motion generated by partial weight-bearing through the tensioned wires of the Ilizarov frame is theorized to stimulate further osteogenesis via the principles of distraction histiogenesis and mechanical transduction.
Frame Removal and Long Term Bracing
The external fixator is typically maintained for a minimum of 4 to 6 months. Removal is entirely dependent on the radiographic evidence of profound, circumferential corticalization of the cross-union mass and obliteration of the osteotomy line. Premature removal of the frame is a catastrophic error that almost invariably leads to refracture.
Once rigid union was confirmed radiographically in at least three out of four cortices, the external fixator was removed under brief general anesthesia in the operating room.
Following frame removal, the patient was immediately placed into a custom-molded, total-contact, clamshell knee-ankle-foot orthosis (KAFO). It is a fundamental tenet of CPT management that the tibia remains mechanically compromised even after achieving a solid radiographic union. The underlying genetic mutation affecting the osteoblasts and osteoclasts remains present; therefore, the bone is perpetually at risk for remodeling into a dysplastic state and refracturing. The patient must wear the clamshell orthosis during all weight-bearing activities until skeletal maturity is reached. The intramedullary telescopic rod is left in place permanently, or until it reaches its maximum excursion and requires exchange, serving as an internal rebar against bending forces.
Clinical Pearls and Pitfalls
The management of pediatric NF1-associated congenital pseudarthrosis of the tibia is fraught with complications. Success requires strict adherence to biomechanical and biological principles.
- Radical Resection is Non-Negotiable: The most common cause of failure and recurrent pseudarthrosis is inadequate resection of the fibromatous hamartoma. The surgeon must resect aggressively until normal, healthy, bleeding bone and periosteum are encountered, regardless of the size of the resulting defect.
- The Charnley Principle: CPT is a biological problem that requires a combined mechanical and biological solution. Neither internal fixation alone, external fixation alone, nor bone grafting alone is sufficient. The combination of intramedullary rodding (for bending control), circular external fixation (for compression and rotational control), and massive autologous bone grafting (for osteoinduction and osteoconduction) represents the gold standard.
- Address the Fibula: The fibula is almost always involved in the dysplastic process. Failure to address the fibular deformity or to incorporate it into a tibiofibular cross-union will result in persistent mechanical axis deviation, progressive valgus deformity of the ankle, and eventual failure of the tibial reconstruction.
- Permanent Internal Splintage: The intramedullary rod must cross the ankle joint initially to provide absolute distal stability and should ideally be a telescoping design to accommodate growth. The rod is never electively removed. If a solid union is achieved, the rod remains in place to protect against the lifelong risk of refracture.
- Lifelong Bracing: The family must be counseled preoperatively that achieving union is only the first hurdle. The patient will require a total-contact orthosis until skeletal maturity. Compliance with bracing is the single most important factor in preventing late refractures after a successful surgical union.
- Manage Expectations: Even in the hands of highly experienced pediatric limb reconstruction surgeons utilizing the most advanced techniques, the refracture rate for CPT remains historically high (approaching 20-30% in some long-term series). Amputation remains a salvage option for multiply recurrent cases with severe, function-limiting deformity and leg length discrepancy, and this possibility must be discussed with the family during the initial consultation.
You Might Also Like