Primary and Secondary Bone Lymphoma: A Comprehensive Surgical and Clinical Guide
Key Takeaway
Lymphoma of bone presents a unique diagnostic and therapeutic challenge for the orthopedic surgeon. While primary treatment relies on systemic chemotherapy and localized radiation, surgical intervention is critical for managing impending or actual pathological fractures. This guide details the clinical evaluation, advanced imaging, immunohistochemical profiling, and step-by-step surgical techniques—including prophylactic intramedullary nailing—required to optimize functional outcomes in patients with osseous lymphoma.
INTRODUCTION TO OSSEOUS LYMPHOMA
Lymphoma involving the skeletal system may manifest either as a primary bone malignancy (Primary Bone Lymphoma, PBL) or as secondary osseous involvement from systemic disease. Primary bone lymphoma is a rare entity, accounting for approximately 3% to 5% of all primary bone tumors and less than 2% of all lymphomas in adults.
The disease can present at any age but exhibits a distinct peak in incidence during the sixth and seventh decades of life. There is a documented male predominance, with a male-to-female ratio of approximately 1.5:1. The appendicular skeleton is most frequently affected, with the femur being the most common site of involvement. This is followed in frequency by the pelvis, the axial spine, and the ribs.
Understanding the clinical behavior, radiographic subtleties, and histopathological characteristics of bone lymphoma is paramount for the orthopedic oncologist and general orthopedic surgeon. While the cornerstone of treatment is medical (chemotherapy and radiation), the orthopedic surgeon plays a vital role in diagnostic biopsy, staging, and the biomechanical stabilization of compromised bone.
CLINICAL PRESENTATION
The clinical presentation of bone lymphoma is often insidious, leading to delayed diagnosis. Most patients report localized, deep, aching bone pain that is often unremitting and may awaken them at night. Localized swelling or a palpable soft-tissue mass may be present, particularly in advanced stages where the tumor has breached the cortex.
In patients with spinal involvement, the presentation can be neurologically catastrophic. Epidural extension or pathological vertebral body collapse can lead to nerve root impingement (radiculopathy) or frank spinal cord compression (myelopathy), necessitating emergent decompression and stabilization.
Clinical Pearl: In stark contrast to patients with multiple myeloma or metastatic carcinoma—who frequently present with cachexia, fatigue, and systemic illness—patients with primary bone lymphoma often feel otherwise entirely healthy. A patient presenting with a massive, destructive bone lesion but lacking systemic symptoms should immediately raise clinical suspicion for lymphoma.
Symptom duration can vary drastically; some patients endure mild, progressive symptoms for several years before seeking definitive medical attention, while others present acutely following a pathological fracture.
DIAGNOSTIC IMAGING
The radiographic evaluation of bone lymphoma is notoriously deceptive. The extent of marrow involvement is frequently vastly disproportionate to the subtle findings on plain radiography.
Plain Radiography
Radiographically, lymphoma typically presents as an ill-defined area of bone destruction. It exhibits a predilection for the diaphyseal or metadiaphyseal regions of long bones. The classic appearance is a "permeative" or "moth-eaten" pattern of osteolysis, indicative of a rapidly infiltrating round cell tumor.
While the cortex may appear thickened due to chronic endosteal scalloping and reactive bone formation, a florid periosteal reaction is rarely observed unless a pathological microfracture has occurred. Frequently, a massive segment of the bone—or even the entire diaphysis—is involved.

Anteroposterior radiograph of the right humerus in a 36-year-old man with primary lymphoma of bone. Note the extensive permeative bone destruction and cortical thinning. Despite this massive structural compromise, the patient’s symptoms were surprisingly mild.
Advanced Imaging: MRI and Bone Scintigraphy
Because plain radiographs can appear entirely normal despite extensive intramedullary disease, Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the local extent of the tumor. MRI exquisitely delineates marrow replacement, cortical breakthrough, and the associated soft-tissue mass, which can be surprisingly extensive.
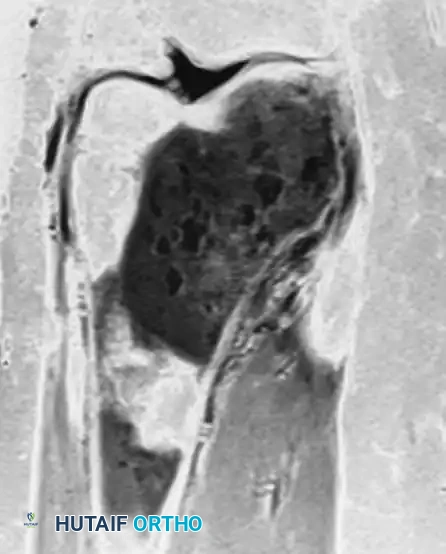
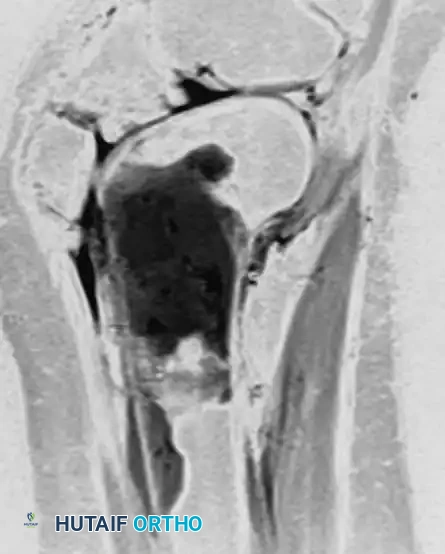
Anteroposterior and lateral radiographs of the right knee in a 51-year-old woman. The osseous structures appear relatively normal, belying the underlying pathology.
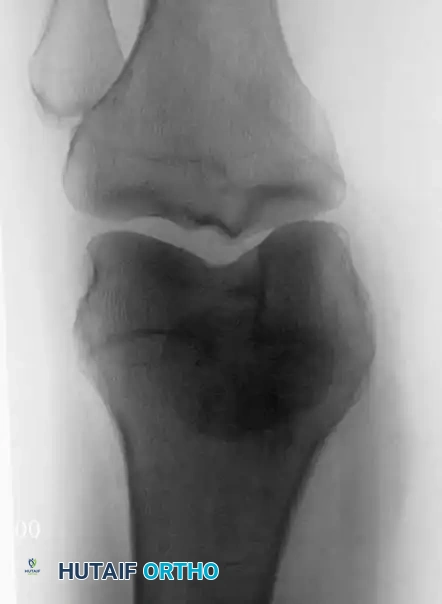
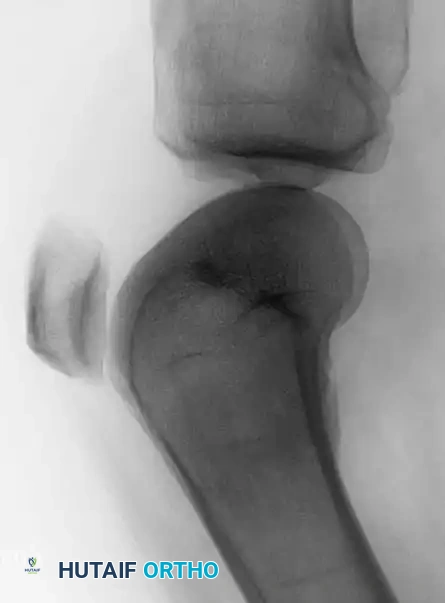
Coronal and sagittal MRI sequences of the same 51-year-old woman. The MRI reveals a massive, marrow-replacing lesion in the distal femur. Incisional biopsy confirmed a B-cell lymphoma. This highlights the critical rule: Lymphoma must be in the differential diagnosis for patients with severe bone pain and normal radiographs but abnormal MRI or bone scans.
DIFFERENTIAL DIAGNOSIS AND STAGING
The differential diagnosis for a permeative, diaphyseal bone lesion in an adult includes:
1. Metastatic Carcinoma: (Breast, lung, thyroid, renal, prostate).
2. Multiple Myeloma / Solitary Plasmacytoma: Often presents with similar destructive lesions but usually with systemic symptoms, hypercalcemia, and renal issues.
3. Ewing Sarcoma: Typically seen in a younger demographic (first and second decades) but can occasionally present in adults.
4. Osteomyelitis: Can mimic the permeative destruction and MRI edema of a round cell tumor.
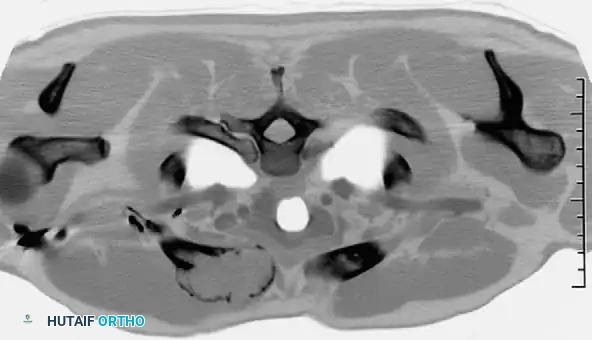
CT scan of a 39-year-old man demonstrating a destructive lesion of the right medial clavicle. Biopsy revealed a solitary plasmacytoma with abundant amyloid. While amyloid in bone is highly associated with myeloma, plasmacytoma and lymphoma share overlapping radiographic features and require definitive tissue diagnosis.
Staging Protocol
Once a diagnosis of osseous lymphoma is suspected, comprehensive staging is mandatory to differentiate Primary Bone Lymphoma (PBL) from systemic disease with secondary bone involvement. Staging studies must include:
* Complete Blood Count (CBC) with differential.
* Comprehensive metabolic panel (including LDH, which is often elevated and serves as a prognostic marker).
* Whole-body bone scintigraphy (Technetium-99m) or PET-CT (highly sensitive for lymphomatous activity).
* CT scans of the chest, abdomen, and pelvis to evaluate for lymphadenopathy or organomegaly.
* Bilateral bone marrow aspiration and biopsy (typically from the posterior iliac crest).
HISTOPATHOLOGY AND IMMUNOHISTOCHEMISTRY
Definitive diagnosis relies on an adequate tissue biopsy. Core needle biopsy is often sufficient, though an open incisional biopsy may be required if core samples are non-diagnostic.
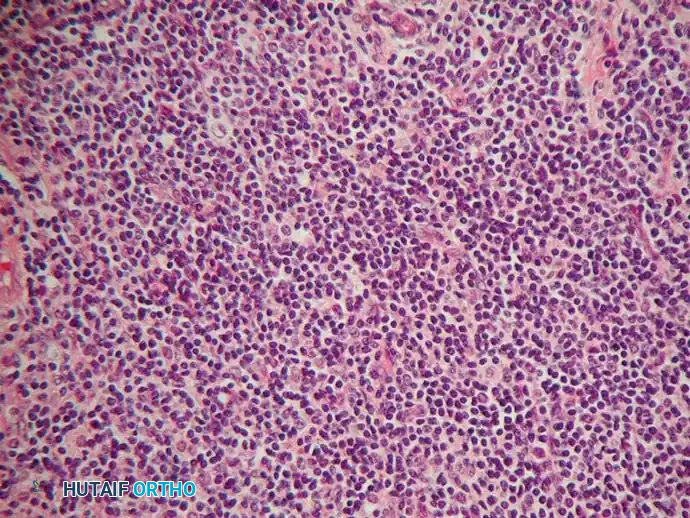
Microscopically, osseous lymphomas are predominantly Non-Hodgkin Lymphomas (NHL), with Diffuse Large B-Cell Lymphoma (DLBCL) being the most common subtype. The cellular infiltrate is composed of a dense, monotonous sheet of large and small lymphoid cells. The nuclei may be cleaved or non-cleaved, with prominent nucleoli and scant cytoplasm.
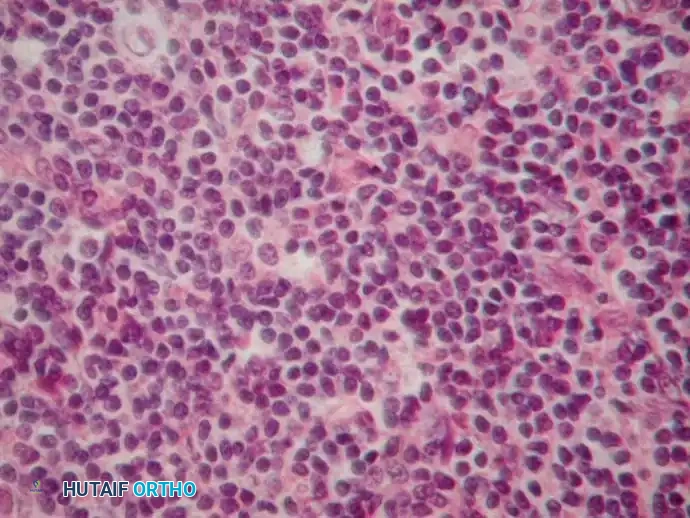
High-power microscopic appearance of bone lymphoma demonstrating a dense infiltrate of atypical lymphoid cells with varying degrees of nuclear cleavage.
Morphologically, lymphoma can appear sarcomatoid, making it exceedingly difficult to distinguish from Ewing sarcoma, undifferentiated pleomorphic sarcoma, or poorly differentiated metastatic carcinoma based on hematoxylin and eosin (H&E) staining alone.
The Role of Immunohistochemistry (IHC)
Immunohistochemistry is the linchpin of diagnosis. A standard panel will differentiate lymphoma from other small blue round cell tumors:
* Positive Markers: CD45 (Leukocyte Common Antigen - LCA), CD20 (B-cell marker, positive in DLBCL), CD3 (T-cell marker).
* Reticulin Stain: Typically positive, demonstrating a dense reticulin network surrounding individual cells.
* Negative Markers: Cytokeratin (rules out carcinoma), S-100 (rules out melanoma), CD99 (usually negative, helping rule out Ewing sarcoma, though weak positivity can occasionally occur), and Periodic Acid-Schiff (PAS) is typically negative (unlike Ewing sarcoma which is PAS positive due to intracellular glycogen).
Surgical Warning: Crush artifact during biopsy can render lymphoid cells unreadable. The surgeon must handle the tissue with extreme care, avoiding excessive forceps compression, and ensure samples are sent fresh (in saline or RPMI medium) for flow cytometry, in addition to formalin fixation.
For contrast, multiple myeloma (which shares clinical overlap) presents with sheets of monoclonal plasma cells, often with a "clock-face" chromatin pattern and a perinuclear halo.
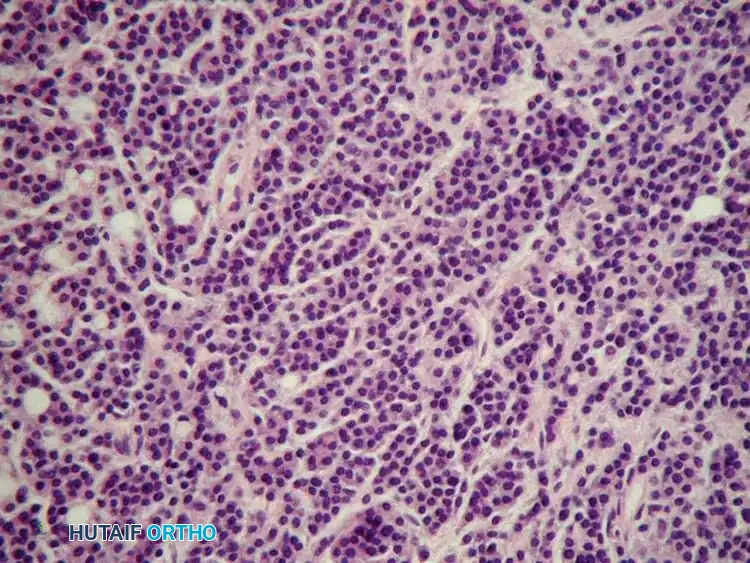
Typical microscopic appearance of multiple myeloma, characterized by dense sheets of malignant plasma cells. Differentiating this from lymphoma via IHC (CD138+, CD38+ for myeloma) is critical for directing medical therapy.
PROGNOSIS AND MEDICAL MANAGEMENT
The prognosis hinges heavily on whether the disease is primary to the bone or systemic. Patients with Primary Bone Lymphoma (PBL) enjoy a significantly better prognosis, with an approximate 55% to 60% 5-year survival rate. Conversely, patients with systemic lymphoma and secondary bone involvement have a much poorer prognosis, with a 5-year survival rate of less than 25%.
Non-Operative Treatment
The primary treatment for lymphoma is systemic chemotherapy, typically utilizing the R-CHOP regimen (Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, Prednisone) for B-cell lineages. Local control of the osseous lesion is highly successful with adjuvant external beam radiation therapy (EBRT). Because lymphoma is exquisitely chemo- and radiosensitive, wide surgical resection (as performed for osteosarcoma) is contraindicated and unnecessary.
OPERATIVE MANAGEMENT: PROPHYLACTIC AND THERAPEUTIC FIXATION
While surgical resection is rarely indicated, surgical stabilization is frequently required. The primary indications for orthopedic intervention are:
1. Actual Pathological Fracture: Requiring rigid internal fixation to allow mobilization and pain relief.
2. Impending Pathological Fracture: Evaluated using Mirels' Criteria. A score of 9 or greater strongly indicates prophylactic fixation.
Preoperative Optimization and Embolization
Highly cellular marrow-replacing tumors (including lymphoma, myeloma, and renal/thyroid metastases) can be massively vascular. If open reduction or extensive exposure is anticipated, preoperative angiography and embolization should be strongly considered to mitigate catastrophic intraoperative hemorrhage.
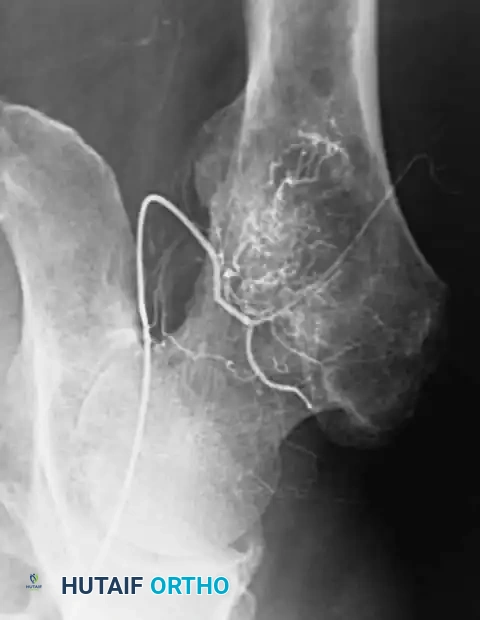
Preoperative angiogram demonstrating a highly vascular tumor blush in the proximal femur.
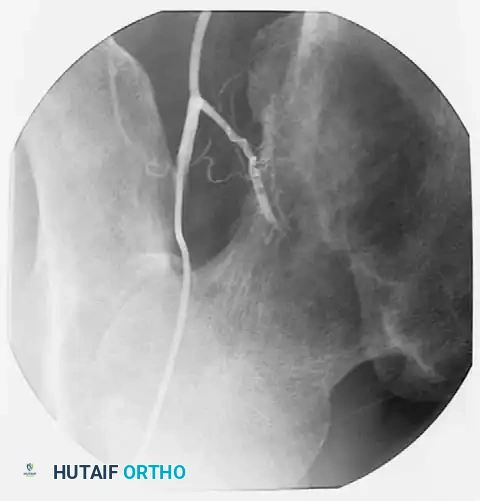
Post-embolization angiogram showing successful occlusion of the tumor's feeding vessels. This critical step minimizes intraoperative blood loss, which can otherwise be extensive and life-threatening.
Biomechanics of Fixation
The goal of surgery in oncologic bone disease is to provide immediate, durable, full weight-bearing stability that will outlast the patient's lifespan or bridge them until radiation/chemotherapy induces bone healing.
* Intramedullary (IM) Nailing: The gold standard for diaphyseal and metadiaphyseal lesions. IM nails offer superior load-sharing biomechanics compared to plates. They protect the entire length of the bone, which is crucial since radiation therapy will temporarily weaken the surrounding osseous structures.
* Plate Osteosynthesis: Reserved for periarticular fractures where an IM nail cannot achieve adequate purchase. If plates are used, polymethylmethacrylate (PMMA) bone cement is often required to fill cavitary defects and augment screw purchase.
Step-by-Step Surgical Technique: Prophylactic Cephalomedullary Nailing of the Femur
When lymphoma involves the proximal or diaphyseal femur, a long cephalomedullary nail (e.g., Gamma nail) is the implant of choice.
1. Patient Positioning and Preparation:
* Place the patient supine on a fracture table or a radiolucent flat Jackson table, depending on surgeon preference and the presence of an actual fracture.
* Ensure high-quality fluoroscopic visualization of the entire femur, from the femoral head to the distal condyles.
2. Surgical Approach:
* A 3-5 cm longitudinal incision is made proximal to the greater trochanter.
* The fascia lata is split in line with its fibers. The gluteus medius is split to access the piriformis fossa or the tip of the greater trochanter (depending on the specific nail design).
3. Guidewire Insertion and Reaming:
* An awl or cannulated opening reamer is used to breach the cortex.
* A ball-tipped guidewire is passed down the medullary canal, traversing the lymphomatous lesion, and centered in the distal femoral epiphysis.
* Surgical Pearl: Reaming must be performed carefully. In marrow-replacing tumors, the canal is often pressurized. Venting the femur (via a small drill hole distal to the lesion) or reaming slowly can reduce the risk of fat or tumor embolization to the lungs.
4. Nail Insertion:
* A long cephalomedullary nail is advanced over the guidewire. The nail must span the entire length of the bone to protect against future fractures, especially if the entire femur is to be irradiated.
5. Proximal and Distal Locking:
* Proximal fixation is achieved with a large lag screw directed into the center of the femoral head, providing rotational stability and preventing varus collapse of the proximal femur.
* Distal locking screws are placed via a perfect-circle fluoroscopic technique to secure the distal segment.
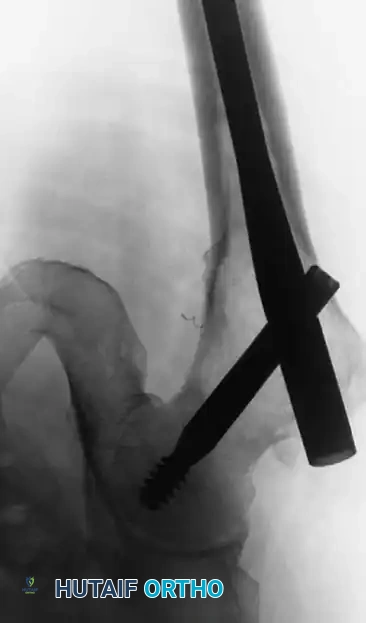
Intraoperative anteroposterior fluoroscopic view demonstrating prophylactic fixation of the proximal femur with a long cephalomedullary (Gamma) nail. Note the trajectory of the lag screw into the femoral head.
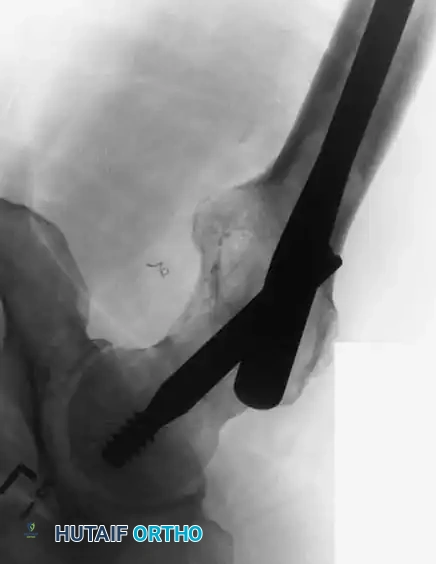
Lateral fluoroscopic view confirming the central placement of the cephalomedullary lag screw within the femoral head and neck.
Postoperative Protocol
- Weight Bearing: Patients are typically allowed immediate weight-bearing as tolerated to prevent the complications of prolonged recumbency (DVT, pneumonia, deconditioning).
- Wound Healing: Surgical incisions must be monitored closely. Radiation therapy should ideally be delayed for 2 to 3 weeks postoperatively to allow for primary soft-tissue healing, preventing wound dehiscence.
- Multidisciplinary Care: Postoperative management requires seamless coordination with medical and radiation oncology to initiate or resume systemic R-CHOP chemotherapy and localized EBRT as soon as surgically permissible.
CONCLUSION
Osseous lymphoma is a complex pathology that demands a high index of clinical suspicion, particularly when radiographic findings are subtle but MRI reveals extensive marrow replacement. While the primary curative modalities are chemoradiation, the orthopedic surgeon's role in obtaining pristine diagnostic tissue and executing biomechanically sound, prophylactic, or therapeutic stabilization is indispensable for preserving the patient's mobility, independence, and quality of life.
📚 Medical References
- bone lymphoma in 24 patients treated between 1955 and 1999, Clin Orthop Relat Res 397:271, 2002.
- De Santis E, Di Giovanni C, Pallotta F: Primary reticulosarcoma of bone (non-Hodgkin’s lymphoma): a study of 12 cases, Ital J Orthop Traumatol 13:477, 1987.
- Dosoretz DE, Raymond AK, Murphy GF, et al: Primary lymphoma of bone: the relationship of morphologic diversity to clinical behavior, Cancer 50:1009, 1982.
- Durr HR, Muller PE, Hiller E, et al: Malignant lymphoma of bone, Arch Orthop Trauma Surg 122:10, 2002.
- Forest M, Tomeno B, Vanel D, eds: Orthopedic surgical pathology: diagnosis of tumors and pseudotumoral lesions of bones and joints, Edinburgh, 1998, Churchill Livingstone. Giudici MAI, Eggli KD, Moser RP Jr, et al: Case report 730, Skeletal Radiol 21:260, 1992.
- Griffi ths H, Conaway JR: Radiologic case study, Orthopedics 22:265, 1999.
- Lands R, Karnad A: Non-T-cell lymphoblastic lymphoma with extensive osteolytic lesions and hypercalcemia, South Med J 84:1405, 1991.
- Ostrowski ML, Unni KK, Banks PM, et al: Malignant lymphoma of bone, Cancer 58:2646, 1986.
- Ruzek KA, Wenger DE: The multiple faces of lymphoma of the musculoskeletal system, Skeletal Radiol 33:1, 2004.
- Sweet DL, Mass DP, Simon MA, et al: Histiocytic lymphoma (reticulum-cell sarcoma) of bone: current strategy for orthopaedic surgeons, J Bone Joint Surg 63A:79, 1981.
- Unni KK: Dahlin’s bone tumors: general aspects and data on 11,087 cases, 5th ed, Philadelphia, 1996, Lippincott-Raven. Vaughan JJ, Winter RB, Lonstein JE, et al: Non-Hodgkin lymphoma of the spine: a report of three cases with a minimum ten-year follow-up, J Bone Joint Surg 80A:104, 1998.
- Wold LE, McLeod RA, Sim FH, et al: Atlas of orthopedic pathology, Philadelphia, 1990, Saunders.
You Might Also Like