Comprehensive Guide to Orthopedic Osteomyelitis: Classification, Epidemiology, & Biomechanics
Key Takeaway
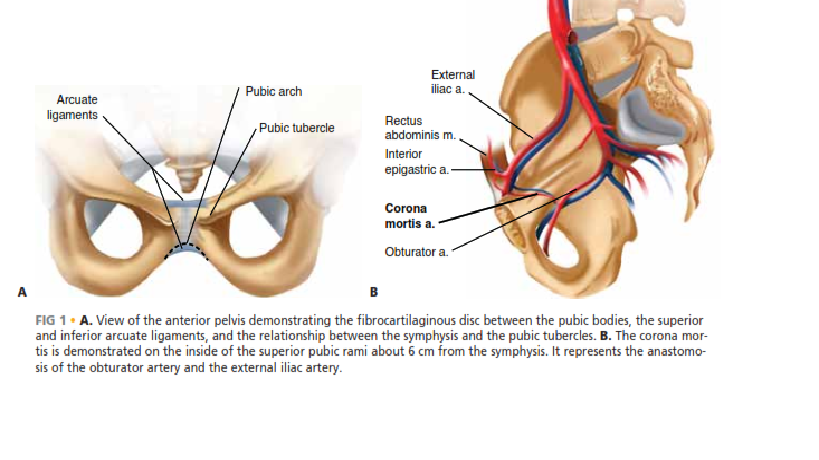
Osteomyelitis is classified by its infection mechanism and chronicity. Mechanism includes Exogenous (direct inoculation from trauma/surgery) and Hematogenous (bloodstream spread). Chronicity defines it as Acute (rapid onset) or Chronic (persistent, often involving necrotic bone, new bone formation, and bacterial biofilms).
Osteomyelitis: Your Guide to Symptoms, Diagnosis, and Treatment
Introduction & Epidemiology
Osteomyelitis represents an inflammatory process of bone and bone marrow, typically initiated by an infecting organism. This condition remains a significant challenge in orthopedic surgery, often necessitating a multidisciplinary approach due to its protean manifestations and the complexity of eradication. The classification of osteomyelitis is primarily based on the mechanism of infection and chronicity, each influencing its epidemiology, presentation, and management.
Classification and Etiology
-
Exogenous Osteomyelitis:
- Prevalence: This is the most common form of osteomyelitis encountered in adult populations.
- Mechanism: Infection typically results from direct inoculation of microorganisms into bone tissue.
-
Common Scenarios:
- Acute Post-Traumatic/Post-Surgical: Direct contamination from open fractures, gunshot wounds, or surgical procedures, including internal fixation of fractures or arthroplasty. The presence of foreign material (implants) significantly lowers the inoculum required for infection.
-
Chronic Contiguous Focus:
Prolonged exposure to infection from neglected wounds, soft tissue ulcers, or chronic sinus tracts.
- Diabetic Foot Infections: A prevalent and severe manifestation, often complicated by peripheral neuropathy and vascular insufficiency, leading to deep tissue and bone involvement without overt signs of acute infection.
- Decubitus Ulcers: Pressure sores that penetrate to bone, particularly over bony prominences such as the sacrum, ischium, and greater trochanter.
- Vascular Insufficiency Ulcers: Similar to diabetic foot infections, compromised blood supply limits healing and antibiotic penetration.
-
Hematogenous Osteomyelitis:
- Prevalence: This is the most common form of osteomyelitis observed in pediatric patients.
- Mechanism: Pathogens disseminate through the bloodstream from a distant primary source of infection.
-
Pediatric Patients:
- Immature Immune System: Contributes to increased susceptibility.
- Anatomical Predilection: The metaphyseal or epiphyseal regions of long bones are common sites due to their rich, slow-flowing sinusoidal capillary networks, which are conducive to bacterial lodging and proliferation.
- Location: Lower extremity bones (femur, tibia) are affected more frequently than upper extremity bones.
-
Adult Patients:
- Risk Factors: Immunocompromised states are significant predispositions. This includes the elderly, individuals undergoing chemotherapy, transplant recipients on immunosuppressive medications, and patients with chronic systemic illnesses.
-
Anatomical Predilection:
- Vertebrae: The most common site for adult hematogenous osteomyelitis, often presenting as discitis-osteomyelitis.
- Rib and Spine: Frequently observed in patients undergoing dialysis, likely due to repeated vascular access and compromised immune function.
- Clavicle: Medial or lateral clavicle osteomyelitis is pathognomonic for intravenous drug abusers (IVDA), often associated with septic emboli from contaminated injection sites.
Chronicity
- Acute Osteomyelitis: Characterized by a rapid onset and a relatively short duration, typically less than two weeks. If recognized and treated promptly, it often responds well to medical and surgical interventions.
- Chronic Osteomyelitis: Persists for weeks, months, or even years. It is characterized by the presence of necrotic bone (sequestrum), new bone formation (involucrum), and often draining sinus tracts. Chronic forms frequently require aggressive surgical debridement in conjunction with prolonged antimicrobial therapy.
Surgical Anatomy & Biomechanics
Understanding the intricate interplay between bone anatomy, vascular supply, and the biomechanical response to infection is paramount for effective management of osteomyelitis.
Bone Anatomy and Vascularity in Infection
- Cortical vs. Cancellous Bone: Cancellous (trabecular) bone, found in the metaphysis and epiphysis, is highly vascular and metabolically active, making it a common site for hematogenous seeding. Cortical bone, denser and less vascular, can become necrotic more easily when its blood supply is interrupted by infection or trauma.
- Periosteum: In children, the periosteum is loosely attached and robustly vascularized, allowing significant subperiosteal abscess formation which can Strip the periosteum from the underlying cortex, leading to cortical necrosis. In adults, the periosteum is more firmly adhered, limiting subperiosteal abscess expansion but making cortical stripping more damaging to vascularity.
- Metaphyseal Vascular Loops (Children): The unique hairpin turns of the metaphyseal arterial supply create turbulent, slow blood flow, providing an ideal environment for bacterial adherence and proliferation, contributing to the high incidence of hematogenous osteomyelitis in this region.
- Vertebral Vascularity (Adults): The vertebral bodies receive segmental arterial supply, with rich anastomoses. However, infection often localizes in the highly vascularized anterior vertebral body adjacent to the intervertebral disc, leading to discitis-osteomyelitis, which can then spread to adjacent vertebrae.
- Biofilm Formation: A critical biomechanical aspect of chronic osteomyelitis, particularly around foreign implants. Bacteria within a biofilm are encased in an extracellular polymeric substance (EPS) matrix, which protects them from host immune responses and reduces antibiotic penetration and efficacy. This renders the infection significantly more resistant to systemic antibiotics alone.
Biomechanical Consequences of Infection
- Bone Weakening: Infection directly degrades bone matrix and impairs osteocyte viability, leading to focal areas of osteolysis. This weakening increases the risk of pathological fractures, particularly in weight-bearing bones or the spine.
-
Sequestrum and Involucrum:
- Sequestrum: Necrotic bone that has been deprived of its blood supply, often appearing denser on radiographs. It acts as a nidus for persistent infection, as antibiotics cannot penetrate devitalized tissue. Surgical removal is crucial.
- Involucrum: New bone formation attempting to wall off the infection, often encasing the sequestrum. While a host response, it can hinder surgical access and create internal compartments that harbor bacteria.
- Compromised Stability: In long bones, extensive debridement of infected and necrotic bone can create significant segmental defects, severely compromising mechanical stability and predisposing to nonunion or malunion if not adequately reconstructed. In the spine, vertebral body destruction can lead to kyphotic deformity and neurological compromise.
- Soft Tissue Envelope: The quality of the surrounding soft tissue is critical. Scarring, fibrosis, and chronic inflammation due to long-standing infection can compromise local vascularity, hindering healing and increasing the risk of wound dehiscence or flap failure after surgical reconstruction.
Indications & Contraindications
The decision for operative intervention in osteomyelitis is complex, balancing the necessity for source control with patient comorbidities and the extent of infection.
Indications for Surgical Intervention
- Failure of Medical Management: Persistent infection despite adequate, pathogen-directed antibiotic therapy.
- Presence of Sequestrum: Necrotic bone that acts as a nidus for infection and cannot be penetrated by antibiotics.
- Intramedullary Abscess: Walled-off collection of pus within the medullary canal.
- Chronic Draining Sinus Tract: Indicative of persistent infection and necrotic tissue.
- Instability / Pathological Fracture: Bone destruction leading to structural compromise or impending fracture.
- Joint Involvement: Septic arthritis secondary to osteomyelitis or impending joint penetration.
- Presence of Foreign Body / Implant Infection: Implants provide a surface for biofilm formation, necessitating removal for infection eradication, especially in chronic cases. Acute infections around stable implants may sometimes be managed with debridement and implant retention (DAIR) in specific circumstances.
- Large Subperiosteal or Soft Tissue Abscess: Requiring drainage to relieve pressure, reduce bacterial load, and improve antibiotic penetration.
- Neurological Deficit (Spinal Osteomyelitis): Compression of the spinal cord or nerve roots necessitating urgent decompression.
- Progressive Disease: Worsening local or systemic symptoms despite conservative measures.
Contraindications for Surgical Intervention
Absolute contraindications are rare, but relative contraindications require careful risk-benefit analysis:
- Uncontrolled Systemic Sepsis/Shock: Requires stabilization prior to definitive surgical intervention, though emergent debridement may be life-saving.
- Severe Uncorrectable Co-morbidities: Advanced cardiac, pulmonary, or renal disease that significantly increases surgical risk without adequate benefit.
- Extremely Frail Patients / Limited Life Expectancy: Consideration of palliative care options over aggressive surgical debridement and reconstruction.
- Diffuse, Untreatable Infection: Extremely rare, but situations where surgical debridement would lead to unacceptable morbidity (e.g., limb amputation without prosthetic potential).
- Early, Uncomplicated Acute Osteomyelitis: Often successfully managed with antibiotics alone, especially in hematogenous cases without significant abscess or necrosis.
Operative vs. Non-Operative Indications
The decision matrix for osteomyelitis management is stratified based on the chronicity, extent, and patient factors.
| Indication Category | Operative Management | Non-Operative Management |
|---|---|---|
| Acute Osteomyelitis |
* Large subperiosteal abscess
Intramedullary abscess Failure of initial medical therapy (e.g., persistent bacteremia, worsening local signs) Pathological fracture or instability Joint involvement (septic arthritis) * Spinal osteomyelitis with neurological deficit |
* Early presentation without abscess or sequestrum
Responsive to systemic antibiotics (e.g., resolution of fever, decreasing inflammatory markers, improving pain) Acute hematogenous osteomyelitis in children without significant abscess or bone destruction * Positive response to image-guided aspiration and antibiotic treatment |
| Chronic Osteomyelitis |
* Presence of sequestrum/involucrum
Chronic draining sinus tract Persistent pain and functional limitation Nonunion or malunion secondary to infection Infected implants (often requires removal and exchange) Soft tissue defects requiring coverage and debridement Recurrent infection after prior conservative management |
* Patients deemed unsuitable for surgery due to severe comorbidities
* Limited, well-controlled infection in select cases where debridement would lead to severe functional deficit (e.g., short segment of minor bone, minimal symptoms, often in conjunction with long-term suppressive antibiotic therapy - a salvage option, not curative) |
| Specific Scenarios |
* Diabetic foot osteomyelitis with bone destruction, abscess, or deep ulceration
Vertebral osteomyelitis with epidural abscess or spinal instability Post-traumatic osteomyelitis with hardware infection (often staged approach with hardware removal, debridement, and delayed reconstruction) |
* Superficial diabetic foot infection without bone involvement
Vertebral osteomyelitis without neurological compromise and stable spine responding to antibiotics Early post-operative superficial surgical site infection without deep bone involvement |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is crucial to optimize outcomes, minimize complications, and ensure comprehensive eradication of osteomyelitis.
Pre-Operative Planning
-
Diagnosis Confirmation and Extent Assessment:
- Clinical Evaluation: Detailed history, physical examination, assessment of neurological status (especially spine).
-
Laboratory Studies:
- CRP (C-reactive protein): Highly sensitive, rapid rise and fall, excellent for monitoring treatment response. An increase in ≈97% of acute cases.
- ESR (Erythrocyte Sedimentation Rate): Elevated in ≈90% of cases; slower to normalize than CRP but useful for monitoring chronic inflammation.
- CBC (Complete Blood Count): Leukocytosis (increased WBCs) in only ≈33% of cases, thus not a sensitive indicator alone.
- Procalcitonin: May be useful in differentiating bacterial from non-bacterial inflammation and for prognosis.
-
Imaging Modalities:
- Plain Radiographs: Initial assessment, may show lucency, periosteal reaction, sequestrum, involucrum in chronic cases. Less sensitive in acute phase (takes 10-14 days for changes).
- MRI (Magnetic Resonance Imaging): Highly sensitive (90-100%) and specific (80-90%) for early detection of osteomyelitis, delineating soft tissue involvement, abscesses, and intramedullary pathology. Crucial for spinal osteomyelitis.
- CT (Computed Tomography): Excellent for assessing cortical bone destruction, sequestrum, involucrum, and planning surgical approaches in complex anatomical regions. Useful for guiding biopsy.
-
Nuclear Medicine Scans:
- Technetium-99m bone scan: Highly sensitive but lacks specificity.
- Indium-111 labeled leukocyte scan or FDG-PET/CT: More specific for infection, especially useful in chronic cases or around implants where MRI artifacts are problematic.
-
Microbiological Confirmation:
- Aspiration and Biopsy Cultures: The most specific diagnostic test. Should be performed before initiating antibiotics if possible, to maximize yield. Multiple samples (at least 3-5) from infected bone and soft tissue are recommended. Histopathology (bony spicules with live osteocytes surrounded by inflammatory cells) confirms osteomyelitis.
- Intraoperative Cultures: Essential for guiding definitive antibiotic therapy; often yields higher rates of positive cultures than pre-operative percutaneous methods.
-
Multidisciplinary Team Involvement:
- Infectious Disease Specialists: For antibiotic selection, duration, and monitoring.
- Plastic/Reconstructive Surgeons: For complex soft tissue defects or flap coverage.
- Vascular Surgeons: If peripheral vascular disease contributes to the infection (e.g., diabetic foot).
- Nutritionists: To optimize nutritional status, critical for wound healing and immune function.
-
Antibiotic Management:
- Pre-operative Antibiotics: Generally deferred until cultures are obtained. If sepsis is present or delay is unavoidable, broad-spectrum empiric antibiotics are started and later narrowed based on culture results.
- Intraoperative Antibiotics: Often administered prior to incision and sometimes locally via antibiotic-impregnated materials.
-
Surgical Planning:
- Approach: Determine the optimal surgical approach to gain wide access for debridement while minimizing damage to neurovascular structures and preserving potential flap options.
- Extent of Debridement: Plan for aggressive debridement of all non-viable bone and soft tissue.
- Reconstruction Strategy: Consider options for dead space management (antibiotic cement spacers/beads, muscle flaps), bone reconstruction (bone graft, vascularized fibula, allograft), and soft tissue coverage.
- Stabilization: Plan for temporary or definitive stabilization (external fixator, internal fixation) depending on bone defect and stability.
Patient Positioning
Patient positioning must allow for optimal exposure of the affected area, access for debridement, potential graft harvesting (e.g., iliac crest), and simultaneous work by other surgical teams (e.g., plastic surgery).
-
General Principles:
- Padding: Protect all pressure points to prevent decubitus ulcers and nerve palsies.
- Sterile Field: Ensure adequate draping to allow for extensive debridement and potential distant donor sites.
- Tourniquet: May be used for extremity surgery to provide a bloodless field, but consideration must be given to the duration of ischemia and potential for further tissue damage in compromised limbs. Release tourniquet before definitive closure to assess for bleeders.
-
Specific Examples:
- Femur/Tibia: Supine or lateral decubitus, depending on the aspect requiring access.
- Spine: Prone position for posterior approaches; lateral decubitus or anterior approach for anterior vertebral body lesions.
- Pelvis/Hip: Lateral decubitus for posterior approaches; supine for anterior/ilioinguinal.
- Foot/Ankle: Supine or prone, depending on location of infection.
Detailed Surgical Approach / Technique
The cornerstone of surgical treatment for osteomyelitis is thorough debridement, aiming for eradication of infected and necrotic tissue. This is often followed by dead space management, stabilization, and soft tissue coverage.
Fundamental Principles of Surgical Debridement
- Radical Excision of All Infected and Non-Viable Tissue: This is the most critical step. All bone that does not bleed or appears devitalized (e.g., grayish, discolored, fragmented) must be removed. This includes sequestrum, chronic granulation tissue, and foreign bodies.
- Wide Surgical Exposure: Adequate incision and soft tissue dissection are required to visualize the full extent of the infection and ensure complete debridement. The incision must consider potential future flap coverage.
- Multiple Intraoperative Cultures: Obtain at least 3-5 distinct samples from different sites of infection (bone, pus, soft tissue interface) for aerobic, anaerobic, and fungal cultures. This guides specific antibiotic therapy.
- Mechanical Cleansing: Aggressive pulsatile lavage with copious amounts of saline helps remove debris and reduce bacterial load.
- Dead Space Management: After debridement, the resulting cavity must be filled to prevent hematoma formation, which can serve as a new nidus for infection. This can involve antibiotic-impregnated polymethylmethacrylate (PMMA) beads or cement spacers, muscle flaps, or sometimes cancellous bone graft once infection is controlled.
- Stabilization: The bone often requires stabilization after debridement, especially if significant bone resection leads to instability. External fixation is frequently preferred in the acute setting to avoid introducing new hardware into an infected field. Internal fixation may be considered in chronic, well-debrided cases.
- Soft Tissue Coverage: Adequate, well-vascularized soft tissue coverage is essential. This may involve primary closure if tension-free, delayed primary closure, local flaps, or free tissue transfer, often in conjunction with plastic surgery.

Step-by-Step Surgical Technique (General)
-
Incision:
- Plan the incision to provide wide access to the entire infected area, potentially incorporating existing sinus tracts or wounds. Avoid vital neurovascular structures.
- Consider future reconstructive needs; parallel incisions for muscle flaps may be required.
-
Soft Tissue Dissection:
- Carefully dissect through skin, subcutaneous tissue, and muscle layers. Identify internervous planes where possible to minimize muscle damage.
- Resect all chronically infected or devitalized soft tissue, including sinus tracts, bursae, and fibrous capsules.
-
Access to Bone:
- Expose the infected bone. The periosteum may be thickened, inflamed, or absent over areas of necrosis.
- Identify and remove any loose sequestra or foreign material.
-
Bone Debridement:
- Using osteotomes, rongeurs, and high-speed burrs, meticulously remove all non-viable bone. The principle is to remove bone until healthy, punctate-bleeding bone is encountered.
- For cortical bone, saucerization (beveling the edges of the cortical defect) can facilitate soft tissue adherence and vascularization.
- For intramedullary infections, reaming the medullary canal may be necessary to remove infected marrow and necrotic bone.
-
Intraoperative Cultures:
- Before extensive lavage, collect multiple bone and soft tissue samples for culture and histopathology. This step is paramount.
-
Pulsatile Lavage:
- Copiously irrigate the wound with saline or antiseptic solutions (e.g., povidone-iodine, chlorhexidine) under pressure to mechanically debride loose debris and reduce bacterial load.
-
Dead Space Management:
- Antibiotic-Impregnated PMMA: After thorough debridement, mold PMMA cement (often mixed with heat-stable antibiotics like vancomycin, gentamicin, or tobramycin) into beads or a custom spacer to fill the defect. This provides high local antibiotic concentrations.
- Muscle Flaps: For larger defects or to bring vascularized tissue into compromised areas, local or free muscle flaps may be transposed.
- VAC (Vacuum-Assisted Closure) Therapy: Can be used to manage large wounds, promote granulation tissue formation, and prepare the wound bed for delayed closure or flap coverage.
-
Stabilization:
- External Fixation: Preferred for initial stabilization in contaminated fields. Provides rigid fixation while allowing for wound access and soft tissue management.
- Internal Fixation: May be used in selected chronic cases after radical debridement, typically with antibiotic coverage and in conjunction with a "clean" wound. Biodegradable implants might offer advantages.
-
Soft Tissue Coverage:
- Primary Closure: Achieved only if the wound is clean, tension-free, and healthy, well-vascularized tissue can be approximated.
- Delayed Primary Closure: After 24-72 hours, once the wound is deemed clean and infection-free.
- Local/Regional Flaps: For moderate defects, using adjacent muscle or fasciocutaneous flaps.
- Free Flaps: For large, complex defects, particularly in lower extremities or highly vascularized areas, requiring microvascular anastomosis.
-
Wound Dressing:
- Apply appropriate dressings, which may include sterile non-adherent dressings, impregnated gauze, or advanced wound care products.
Special Considerations
- Vertebral Osteomyelitis: Anterior surgical approaches (thoracotomy, retroperitoneal) may be required for direct debridement of vertebral bodies and discectomy, followed by reconstruction with structural bone grafts or cages, often with supplemental posterior stabilization.
- Diabetic Foot Osteomyelitis: Often involves partial foot amputation (toe, ray resection), transmetatarsal amputation, or even ankle disarticulation/below-knee amputation, depending on the extent of infection and vascular compromise. Reconstruction often involves careful attention to wound healing and offloading.
- Prosthetic Joint Infection (PJI): Often requires a two-stage exchange arthroplasty (removal of components, debridement, antibiotic spacer, then reimplantation), though one-stage exchange or DAIR may be considered in highly selected cases.
Complications & Management
Osteomyelitis management is fraught with potential complications, both related to the infection itself and the aggressive surgical and medical treatments. Proactive identification and skilled management are essential.
| Complication | Incidence (Approximate) | Salvage Strategies / Management | Recurrence (after surgery and antibiotics) | 5-20% | * Repeat radical surgical debridement.
Revision stabilization (if implants infected or loose).
Prolonged targeted antibiotic therapy.
Consider reconstructive options (e.g., vascularized bone graft for large defects, soft tissue flap).
Amputation as a last resort for chronic, intractable infection with significant functional impairment. |
|
Pathological Fracture
| Variable, dependent on bone destruction | * Surgical stabilization (external fixation, internal fixation in a clean environment).
Bone graft (autograft, allograft) or vascularized fibula transfer for bone reconstruction.
May require extensive soft tissue coverage. |
|
Nonunion / Pseudoarthrosis
| Significant after bone trauma or surgery | * Revision osteosynthesis.
Bone grafting (autograft/allograft) for deficient or poorly vascularized bone.
Infection workup if chronic nonunion suspected as related to low-grade infection.
Limb salvage procedures if extensive bone loss. | Nonunion / Pseudoarthrosis | Significant after bone trauma or surgery | * Revision osteosynthesis.
Bone grafting (autograft/allograft) for deficient or poorly vascularized bone.
Infection workup if chronic nonunion suspected as related to low-grade infection. |
|
Functional Limitation (Chronic)
| Often leads to significant functional impairment | * Amputation for intractable pain or non-functional limb.
Limb salvage with extensive reconstruction (bone/soft tissue transfer) to preserve function. |
|
Soft Tissue Issues / Defects
| Significant incidence with deep infections | * Meticulous wound care, débridement, and infection control.
Local rotational flaps for smaller defects.
Free tissue transfer (microvascular flaps) for larger, complex soft tissue defects, to bring in highly vascularized tissue. |
|
Fatigue/Weakness
| Variable, often due to infection and chronic conditions | * Nutritional support and optimization.
Aggressive osteomyelitis management.
Physical therapy to restore strength and function.
Correction of anemia or other systemic causes. |
|
Implant-Related Infection
| High (if hardware present) | * Removal of infected implant (if clinically feasible and stable without it).
Thorough debridement of bone and soft tissues.
Staged reconstruction (e.g., explantation, spacer, reimplantation).
Long-term suppressive antibiotics in select cases where implant retention is unavoidable. |
|
Neurovascular Injury
| Rare, but possible with aggressive surgical dissection | * Careful identification and preservation of neurovascular structures.
Intraoperative mapping or angiography if anatomical variations or complex anatomy is encountered.
Post-operative neurological assessment and prompt intervention for deficits. |
|
Malnutrition/Cachexia
| ~30-50% in hospitalized patients with chronic infection | * Aggressive nutritional support (oral, enteral, parenteral).
Correction of underlying systemic inflammatory state. |
|
Organ failure (e.g., renal, hepatic)
| Variable, depends on severity of sepsis | * Intensive supportive care.
Optimization of fluid and electrolyte balance.
Dialysis or liver support as indicated.
Aggressive infection source control and appropriate antibiotics. |
|
Other (e.g., DVT, PE, pressure ulcers) | Variable, often dependent on immobility and comorbidities | * Prophylactic DVT/PE measures (mechanical and pharmacological).
Frequent repositioning and specialized mattresses to prevent pressure ulcers. |
| *
O.R. Time | Dependent on procedure and patient factors | * Efficient surgical planning and execution.
Minimizing unnecessary delays.
* Consideration of patient comorbidities and tolerance for prolonged surgery. |
|
|
Source of Bite
|
Organism
|
Primary Antimicrobial Regimen
|
|
|
*Human
|
Streptococcus viridans (100%)
Bacteroides spp. (82%)
Staphylococcus aureus
(30%)
Eikenella corrodens
(15%) |
Initial Regimen (Oral)
: Amoxicillin-clavulanate (Augmentin) or Clindamycin + Fluoroquinolone (for penicillin allergy)
Initial Regimen (IV)
: Ampicillin-sulbactam (Unasyn) or Piperacillin-tazobactam (Zosyn) or Ceftriaxone + Metronidazole (for penicillin allergy)
Duration
: Typically 6 weeks total. Adjust based on clinical response and organism sensitivities. |
|
* |
*Cat
|
Pasteurella multocida (80%)
Staphylococcus aureus
(15%)
Bartonella henselae
(cat scratch disease) |
Initial Regimen (Oral)
: Amoxicillin-clavulanate or Doxycycline (for
Bartonella
)
Initial Regimen (IV)
: Ampicillin-sulbactam or Piperacillin-tazobactam
Duration
: Typically 6 weeks total. Adjust based on clinical response and organism sensitivities. |
| |
Dog
|
Staphylococcus intermedius
(50-60%)
_Pasteurella multocida
(40-50%)
Capnocytophaga canimorsus (in asplenic) |
Initial Regimen (Oral)
: Amoxicillin-clavulanate or Clindamycin + Fluoroquinolone (for penicillin allergy)
Initial Regimen (IV)
: Ampicillin-sulbactam or Piperacillin-tazobactam or Ceftriaxone + Metronidazole (for penicillin allergy)
Duration
: Typically 6 weeks total. Adjust based on clinical response and organism sensitivities. |
|
Human
|
Streptococcus viridans (100%)
Bacteroides spp. (82%)
Staphylococcus aureus
(30%)
Eikenella corrodens
(15%) |
Initial Regimen (Oral)
: Amoxicillin-clavulanate (Augmentin) or Clindamycin + Fluoroquinolone (for penicillin allergy)
Initial Regimen (IV)
: Ampicillin-sulbactam (Unasyn) or Piperacillin-tazobactam (Zosyn) or Ceftriaxone + Metronidazole (for penicillin allergy)
Duration
: Typically 6 weeks total. Adjust based on clinical response and organism sensitivities. |
|
Cat
|
Pasteurella multocida (80%)
Staphylococcus aureus
(15%)
Bartonella henselae
(cat scratch disease) |
Initial Regimen (Oral)
: Amoxicillin-clavulanate or Doxycycline (for
Bartonella
)
Initial Regimen (IV)
: Ampicillin-sulbactam or Piperacillin-tazobactam
Duration
: Typically 6 weeks total. Adjust based on clinical response and organism sensitivities. |
|
Dog
|
Staphylococcus intermedius
(50-60%)
_Pasteurella multocida
(40-50%)
Capnocytophaga canimorsus (in asplenic) |
Initial Regimen (Oral)
: Amoxicillin-clavulanate or Clindamycin + Fluoroquinolone (for penicillin allergy)
Initial Regimen (IV)
: Ampicillin-sulbactam or Piperacillin-tazobactam or Ceftriaxone + Metronidazole (for penicillin allergy)
Duration
: Typically 6 weeks total. Adjust based on clinical response and organism sensitivities. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is a critical component of osteomyelitis management, aiming to restore function, prevent recurrence, and integrate the patient back into daily activities. Protocols are highly individualized, dependent on the site of infection, extent of surgical debridement, and method of reconstruction and stabilization.
-
Immobilization vs. Mobilization:
- Initial Immobilization: Often necessary to protect the surgical site, facilitate soft tissue healing, and prevent stress on newly reconstructed bone or fixation. This may involve casts, splints, or external fixators.
- Early Controlled Mobilization: Once initial wound healing and stability are achieved, controlled range of motion exercises may begin to prevent joint stiffness and muscle atrophy, particularly in non-weight-bearing joints.
- Weight-Bearing Restrictions: Strict non-weight-bearing is often mandated after extensive bone debridement or reconstructive procedures, gradually progressing to partial and then full weight-bearing based on radiographic healing and clinical stability. This can extend for several months.
-
Wound Care and Monitoring:
- Dressing Changes: Regular wound dressing changes, particularly if VAC therapy is utilized. Monitoring for signs of infection, wound dehiscence, or seroma/hematoma.
- Drain Management: Surgical drains are typically removed when output is minimal (e.g., <25-50 mL/24h) and serous.
- Infection Surveillance: Regular clinical checks for signs of local infection (erythema, swelling, discharge, tenderness) and systemic signs (fever, malaise).
- Laboratory Monitoring: Serial CRP and ESR measurements are essential to track the inflammatory response and assess the efficacy of antibiotic therapy. A decreasing trend is favorable.
-
Antibiotic Therapy:
- Duration: Systemic, pathogen-directed antibiotic therapy is typically continued for a prolonged period, commonly 4-6 weeks intravenously, followed by oral antibiotics for several more weeks or months, depending on the organism, extent of debridement, and host factors. The total duration often extends to 6-12 weeks, with some chronic cases requiring longer or suppressive therapy.
- Monitoring for Adverse Effects: Close monitoring for antibiotic-related side effects (renal, hepatic toxicity, gastrointestinal intolerance, C. difficile infection).
- Compliance: Emphasize strict adherence to the antibiotic regimen to prevent recurrence and resistance.
-
Physical and Occupational Therapy:
- Pain Management: Multimodal analgesia to facilitate early participation in rehabilitation.
- Strength and Range of Motion: Targeted exercises to regain strength, flexibility, and endurance.
- Gait Training: For lower extremity involvement, progressive gait training with assistive devices.
- Functional Training: Re-educate patients on activities of daily living and functional movements specific to their needs.
- Orthotics/Prosthetics: Fabrication of custom orthoses for support or prosthetics following amputation.
-
Nutritional Support:
- Optimizing Healing: Adequate protein, calorie, vitamin, and mineral intake is critical for wound healing, immune function, and bone regeneration. Nutritional assessment and intervention are often required.
-
Psychological Support:
- Chronic Nature: Patients with osteomyelitis often face a prolonged and challenging recovery. Psychological support, addressing anxiety, depression, and frustration, is important for overall well-being and compliance.
-
Long-Term Follow-up:
- Recurrence: Patients require long-term follow-up for several years to monitor for signs of recurrence. This includes clinical evaluation, inflammatory markers, and periodic imaging.
Summary of Key Literature / Guidelines
Management of osteomyelitis is a continually evolving field, guided by accumulating evidence and consensus guidelines.
-
Infectious Diseases Society of America (IDSA) Guidelines: The IDSA provides comprehensive guidelines for the diagnosis and management of various orthopedic infections, including native vertebral osteomyelitis, diabetic foot infections, and prosthetic joint infections. These guidelines offer evidence-based recommendations on diagnostic criteria, microbiological work-up, empirical and pathogen-directed antimicrobial therapy, and the role of surgical intervention. They emphasize the importance of identifying the causative organism and tailoring antibiotic regimens based on susceptibility profiles.
-
European Society of Clinical Microbiology and Infectious Diseases (ESCMID) Guidelines: Similar to IDSA, ESCMID provides robust guidelines, often with a focus on specific scenarios such as post-surgical bone and joint infections or osteomyelitis in specific patient populations. These guidelines frequently address the nuances of biofilm-related infections and strategies for implant retention versus removal.
-
Orthopedic Trauma Association (OTA) and American Academy of Orthopaedic Surgeons (AAOS): These organizations publish clinical practice guidelines and educational materials related to the surgical management of osteomyelitis, particularly in the context of open fractures, nonunions, and chronic post-traumatic infections. They emphasize principles of radical debridement, stable fixation, and soft tissue coverage.
-
Key Literature Themes:
- Multidisciplinary Approach: Consensus in the literature highlights that optimal outcomes are achieved through a collaborative team including orthopedic surgeons, infectious disease specialists, plastic surgeons, and rehabilitation therapists.
- Importance of Cultures: The critical role of deep tissue and bone cultures in guiding specific antibiotic therapy is consistently underscored. Empiric therapy should be broad but rapidly refined once culture results are available.
- Radical Debridement: All guidelines stress that surgical debridement of all non-viable bone and soft tissue is paramount for successful eradication of osteomyelitis, especially in chronic forms or those involving implants.
- Antibiotic-Impregnated Materials: The use of local antibiotic delivery systems (e.g., PMMA beads, cement spacers) has been shown to achieve high local concentrations, supplementing systemic therapy and improving success rates, particularly in dead space management.
- Soft Tissue Coverage: Adequate vascularized soft tissue coverage is recognized as vital for preventing recurrence and promoting healing, often necessitating advanced reconstructive techniques.
- Emerging Concepts: Ongoing research focuses on novel antimicrobial agents, anti-biofilm strategies, advanced imaging techniques (e.g., PET-CT, specialized MRI sequences), and biological adjuncts to enhance bone healing in infected fields. The role of bacteriophages and newer surgical technologies also represents an area of active investigation.
In summary, the management of osteomyelitis remains a complex and challenging endeavor that demands a systematic, evidence-based, and patient-centered approach. Integrating radical surgical debridement with targeted antimicrobial therapy and comprehensive rehabilitation is key to achieving durable infection control and restoring functional outcomes.
Clinical & Radiographic Imaging

You Might Also Like