Upper Extremity Compartment Syndrome: Pathophysiology, Historical Context, and Operative Management
Key Takeaway
Compartment syndrome of the upper extremity is a surgical emergency characterized by elevated intracompartmental pressure that compromises tissue perfusion. Left untreated, it progresses to irreversible muscle necrosis and Volkmann ischemic contracture. This comprehensive guide details the historical evolution of our understanding, the underlying pathophysiology, and the definitive step-by-step surgical management via decompressive fasciotomy to preserve limb function and prevent devastating ischemic sequelae.
DEFINITION AND INTRODUCTION
Acute compartment syndrome (ACS) is a devastating orthopedic emergency defined as a condition in which the microcirculation within a closed, unyielding fibro-osseous compartment is critically compromised by an abnormal elevation in intracompartmental pressure. This pathological pressure gradient obstructs venous outflow, subsequently collapsing the arterial microcirculation and leading to profound tissue hypoxia. If surgical decompression is not achieved expeditiously, the resulting ischemia causes irreversible necrosis of the highly metabolically active skeletal muscles and peripheral nerves. Ultimately, the overlying skin may also succumb to necrosis due to excessive, unremitting swelling and vascular compromise.
Volkmann ischemic contracture represents the catastrophic, end-stage sequela of an untreated or inadequately decompressed compartment syndrome. In this condition, the necrotic muscle and nerve tissues undergo fibrotic degeneration, being replaced by dense, inelastic scar tissue. This results in severe, fixed contractures of the affected limb, rendering it functionally useless and often insensate.
In the upper extremity, compartment syndrome most frequently afflicts the forearm, typically secondary to high-energy trauma, crush injuries, or fractures of the radius and ulna. However, the intrinsic muscle compartments of the hand are also highly susceptible to ischemic insult. While less common, compartment syndrome of the upper arm (brachium) has been well-documented, particularly following severe humerus fractures, prolonged immobilization, or massive fluid extravasation.
HISTORICAL PERSPECTIVES
The evolution of our understanding of compartment syndrome is a testament to over a century of meticulous clinical observation and surgical inquiry.
In 1881, Richard von Volkmann published his seminal paper describing the rapid onset of paralytic contractures that could develop within mere hours following a limb injury. Volkmann astutely deduced that these contractures were not of neurogenic origin, but rather the direct result of arterial insufficiency and profound muscle ischemia. However, he erroneously postulated that extrinsic compression—specifically, the application of overly tight bandages and splints—was the sole etiology of this vascular insufficiency. This concept of extrinsic pressure as the primary, if not exclusive, cause of paralytic contracture dominated the English medical literature for decades.
The paradigm began to shift in 1909 when Thomas conducted a comprehensive study of 107 cases of paralytic contracture. He observed that several severe contractures developed following blunt contusions to the forearm in the complete absence of fractures, splints, or tight bandages. This pivotal observation established the concept that extrinsic pressure was not the sole harbinger of ischemia.
In 1914, Murphy advanced the pathophysiological understanding significantly. He reported that internal hemorrhage and inflammatory effusion into the muscle bellies could cause a rapid increase in internal pressures within the unyielding, deep fascial compartments of the forearm. Murphy correctly identified that this internal pressure surge led to the subsequent obstruction of venous return, initiating the ischemic cascade.
By 1928, Jones synthesized these theories, concluding definitively that Volkmann contracture could be precipitated by pressure from within (hemorrhage, edema), from without (casts, tight dressings), or a synergistic combination of both. Building upon this established pathophysiology, Eichler and Lipscomb later outlined the early, rudimentary techniques of surgical fasciotomy, cementing it as the primary, indispensable surgical treatment for impending or established compartment syndrome.
PATHOPHYSIOLOGY AND BIOMECHANICS
The biomechanics of compartment syndrome are governed by the principles of fluid dynamics within a closed container. The fascial envelopes of the upper extremity are inherently inelastic. When the volume of the compartment increases (via hemorrhage, edema, or reperfusion injury) or the capacity of the compartment decreases (via tight casts or circumferential burns), the intracompartmental pressure (ICP) rises exponentially.
Clinical Pearl: The critical threshold for muscle ischemia is not an absolute pressure value, but rather the perfusion pressure (Delta P). Delta P is calculated as the Diastolic Blood Pressure minus the Intracompartmental Pressure (Delta P = DBP - ICP). A Delta P of less than 30 mmHg is universally recognized as the absolute indication for emergent fasciotomy.
The ischemic cascade proceeds as follows:
1. Venous Hypertension: As ICP rises, it first exceeds the low pressure of the venous outflow tract, causing venous collapse and congestion.
2. Capillary Transudation: Venous congestion leads to increased hydrostatic pressure within the capillary beds, forcing plasma into the interstitial space, further increasing ICP.
3. Arteriolar Collapse: Once the ICP exceeds the critical closing pressure of the precapillary arterioles, arterial inflow ceases.
4. Cellular Hypoxia: Deprived of oxygen, muscle cells switch to anaerobic metabolism, producing lactic acid. The resulting acidosis disrupts the sodium-potassium pump, leading to massive intracellular swelling and eventual cell death.
5. Nerve Ischemia: Peripheral nerves are highly sensitive to hypoxia. Sensory changes (paresthesia) occur within 30 minutes of ischemia, while irreversible nerve damage begins after 12 to 24 hours.
CLINICAL PRESENTATION AND DIAGNOSIS
The diagnosis of acute compartment syndrome is primarily clinical, demanding a high index of suspicion. The classic "6 Ps" (Pain, Pallor, Pulselessness, Paresthesia, Paralysis, and Poikilothermia) are frequently taught; however, relying on them can be a dangerous pitfall.
Surgical Warning: Pallor, pulselessness, and paralysis are late signs of compartment syndrome. If a surgeon waits for the radial pulse to disappear before diagnosing forearm compartment syndrome, irreversible muscle necrosis has likely already occurred.
Early Clinical Signs:
* Pain out of proportion: The hallmark symptom. The pain is deep, unremitting, and poorly localized, often unresponsive to potent intravenous opioids.
* Pain with passive stretch: The most sensitive early clinical finding. In the forearm, passive extension of the fingers (stretching the flexor compartment) will elicit excruciating pain.
* Palpable tenseness: The affected compartment feels "wood-like" or rigidly tense compared to the contralateral limb.
Objective Diagnostics:
In obtunded, intubated, or polytrauma patients where clinical examination is unreliable, objective measurement of intracompartmental pressure is mandatory. Using a solid-state transducer or a slit-catheter technique, pressures must be measured in all suspected compartments. Measurements should be taken within 5 cm of the fracture site, as pressure gradients exist within the compartment.
SURGICAL ANATOMY OF THE FOREARM
A thorough mastery of forearm anatomy is non-negotiable for safe and effective decompression. The forearm is divided into three primary compartments:
- Volar (Flexor) Compartment: The most frequently involved compartment. It is subdivided into superficial and deep layers.
- Superficial: Pronator teres, flexor carpi radialis (FCR), palmaris longus, flexor carpi ulnaris (FCU), and flexor digitorum superficialis (FDS).
- Deep: Flexor digitorum profundus (FDP), flexor pollicis longus (FPL), and pronator quadratus. The deep flexors are the most severely affected during ischemia due to their proximity to the interosseous membrane and their specific vascular supply.
- Dorsal (Extensor) Compartment: Contains the extensor digitorum communis, extensor digiti minimi, extensor carpi ulnaris, supinator, and the outcropping muscles of the thumb.
- Mobile Wad (Lateral) Compartment: Contains the brachioradialis, extensor carpi radialis longus (ECRL), and extensor carpi radialis brevis (ECRB).
INDICATIONS FOR SURGERY
Absolute Indications:
* Unequivocal clinical signs of acute compartment syndrome (pain out of proportion, severe pain on passive stretch).
* Intracompartmental pressure measurements yielding a Delta P (Diastolic BP - ICP) of less than 30 mmHg.
* Absolute ICP consistently greater than 30-40 mmHg in an unresponsive or unreliable patient.
Contraindications:
* Missed compartment syndrome (presentation > 48-72 hours post-injury with established necrosis and absent motor/sensory function). Fasciotomy in this late stage significantly increases the risk of severe infection and systemic sepsis without providing functional benefit.
PREOPERATIVE PREPARATION AND POSITIONING
- Resuscitation: Ensure the patient is hemodynamically stable. Hypotension drastically lowers the diastolic blood pressure, thereby reducing the Delta P and exacerbating the ischemic insult.
- Positioning: The patient is placed supine on the operating table with the affected arm extended on a radiolucent hand table.
- Tourniquet: A pneumatic tourniquet is applied to the upper arm but is not inflated routinely. Inflating the tourniquet exacerbates ischemia and prevents the surgeon from assessing muscle viability and bleeding during the decompression. It is reserved strictly for catastrophic, uncontrollable hemorrhage.
- Preparation: The entire upper extremity, from the axilla to the fingertips, is prepped and draped in a standard sterile fashion.
SURGICAL APPROACH: FOREARM FASCIOTOMY
The gold standard for forearm decompression is the combined volar and dorsal approach. The volar approach must decompress the superficial flexors, deep flexors, and the carpal tunnel.
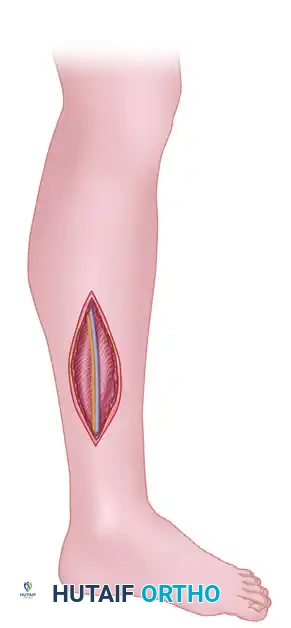
Step 1: The Volar Approach (Extended Henry)
- Incision Planning: Begin the incision just proximal to the medial epicondyle of the humerus. Extend it distally and laterally across the antecubital fossa, curving gently toward the radial aspect of the forearm. Continue distally along the volar forearm, curving ulnarly to cross the wrist crease obliquely (to prevent flexion contractures of the scar). Finally, extend the incision into the palm, paralleling the thenar crease to access the carpal tunnel.
- Superficial Dissection: Incise the skin and subcutaneous tissue. Identify and protect the medial antebrachial cutaneous nerve proximally and the palmar cutaneous branch of the median nerve at the wrist.
- Carpal Tunnel Release: Distally, incise the transverse carpal ligament completely to decompress the median nerve. This is a critical step, as forearm swelling invariably compresses the median nerve at the wrist.
- Superficial Fasciotomy: Proceed proximally, incising the superficial antebrachial fascia over the flexor carpi radialis (FCR) and flexor digitorum superficialis (FDS). Use dissecting scissors to open the fascia completely from the wrist to the antecubital fossa.
- Deep Fasciotomy: Retract the FCR and the radial artery laterally, and the FDS medially. This exposes the deep compartment containing the flexor digitorum profundus (FDP) and flexor pollicis longus (FPL). Incise the deep fascia overlying these muscles.
- Epimysiotomy: Inspect the muscle bellies. If the muscle remains tight and pale despite fascial release, perform an epimysiotomy (incising the epimysium directly over the muscle belly) to ensure complete decompression.
- Assessment of Viability: Assess muscle viability using the "4 Cs": Color (red/pink vs. dark/pale), Consistency (soft/elastic vs. firm/friable), Contractility (twitches with electrocautery), and Capacity to bleed. Do not aggressively debride muscle during the index procedure unless it is unequivocally necrotic and liquefying.
Pitfall: Failing to release the deep fascial layer over the FDP is the most common cause of inadequate volar decompression, leading directly to Volkmann's contracture of the deep flexors.
Step 2: The Dorsal Approach
While the volar incision decompresses the flexors, a separate dorsal incision is often required to decompress the extensor compartment and the mobile wad.
- Incision Planning: Make a straight longitudinal incision on the dorsal aspect of the forearm, starting 2 cm distal to the lateral epicondyle and extending distally toward the midline of the wrist.
- Fascial Release: Incise the subcutaneous tissue to expose the dorsal antebrachial fascia. Incise the fascia longitudinally over the extensor digitorum communis.
- Mobile Wad Decompression: Undermine the skin laterally from the same incision to access the fascia overlying the mobile wad (brachioradialis, ECRL, ECRB). Incise this fascia longitudinally.
- Extensor Retinaculum: It is generally not necessary to release the extensor retinaculum at the wrist unless there is specific evidence of localized compartment syndrome in the dorsal wrist compartments.
Step 3: Hand Compartment Decompression (If Indicated)
If the hand is tense and intrinsic minus posturing is present, the hand compartments (10 in total: 4 dorsal interosseous, 3 volar interosseous, thenar, hypothenar, and adductor) must be released.
* Interossei: Two longitudinal dorsal incisions over the 2nd and 4th metacarpals allow access to all interosseous compartments.
* Thenar/Hypothenar: Separate longitudinal incisions along the glabrous borders of the thenar and hypothenar eminences are used to decompress these respective compartments.
POSTOPERATIVE PROTOCOL AND WOUND MANAGEMENT
The management of the fasciotomy wound is as critical as the decompression itself.
- Wound Closure: Never attempt primary closure of a fasciotomy wound. The wounds must be left open to accommodate the inevitable postoperative swelling.
- Dressings: Apply a sterile, non-adherent dressing. Negative Pressure Wound Therapy (NPWT / VAC dressing) at 75 to 125 mmHg continuous pressure is the current gold standard. NPWT manages exudate, reduces interstitial edema, and promotes granulation tissue formation.
- Alternative Techniques: The "shoelace" technique using vessel loops interlaced through skin staples can be applied to provide gentle, continuous dynamic traction on the skin edges, facilitating delayed primary closure.
- Immobilization: Splint the extremity in a functional position (wrist in 20 degrees of extension, MCP joints in 70 degrees of flexion, IP joints fully extended). Elevate the limb strictly above the level of the heart to promote venous and lymphatic drainage.
- Second Look: Return the patient to the operating room at 48 to 72 hours for a "second look" procedure. At this stage, re-evaluate muscle viability, perform necessary debridement of definitively necrotic tissue, and attempt delayed primary closure.
- Skin Grafting: If the wound edges cannot be approximated without tension by 5 to 7 days, a split-thickness skin graft (STSG) should be harvested (typically from the anterior thigh) to cover the remaining defect.
COMPLICATIONS AND SEQUELAE
The most devastating complication of missed or inadequately treated compartment syndrome is Volkmann Ischemic Contracture.
Tsuge classified Volkmann's contracture into three types based on severity:
* Mild: Localized to the deep flexors (usually FDP of the middle and ring fingers). Treated with dynamic splinting or localized tendon lengthening.
* Moderate: Involves both deep and superficial flexors, often with median and ulnar nerve sensory deficits. Requires radical flexor muscle slide (e.g., Page-Scaglietti procedure), neurolysis, and potential tendon transfers.
* Severe: Extensive necrosis of all flexors and often extensors, with profound neurological deficit. The limb is severely contracted and insensate. Management requires radical excision of all fibrotic scar tissue, extensive nerve grafting, and free functional muscle transfer (e.g., gracilis free flap) to restore basic prehension.
Other complications include chronic regional pain syndrome (CRPS), permanent peripheral nerve palsy, infection, and in extreme cases of myoglobinuria secondary to massive muscle necrosis, acute renal failure requiring hemodialysis.
Vigilance, rapid clinical assessment, and decisive surgical intervention remain the cornerstones of preventing these catastrophic outcomes in upper extremity compartment syndrome.
You Might Also Like