Transfemoral Amputation: An Oncologic Masterclass for Fellows
Key Takeaway
This masterclass guides orthopaedic fellows through oncologic transfemoral amputation. We meticulously cover surgical anatomy, preoperative planning, and granular intraoperative steps from incision to closure. Learn precise techniques for neurovascular management, bone transection, myodesis, and nerve handling. Extensive pearls, pitfalls, and postoperative strategies for optimal patient outcomes and complication management are emphasized, ensuring a comprehensive understanding of this complex procedure.
Introduction and Epidemiology
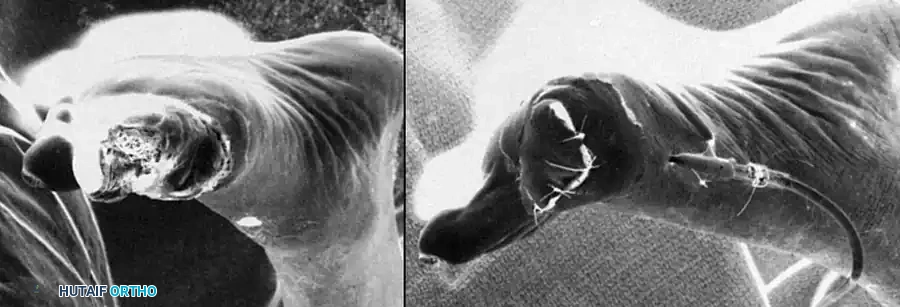
Although many bone and soft tissue sarcomas of the femur and thigh can be treated with limb-sparing techniques, some aggressive tumors are complicated by neurovascular involvement or extensive soft tissue contamination and thus require an above-knee amputation. An above-knee amputation, or transfemoral amputation, of the lower extremity is a profound reconstructive procedure. It must not be viewed merely as an ablative failure, but rather as a definitive surgical reconstruction designed to eradicate disease while creating a functional, well-balanced residual limb capable of enduring the biomechanical demands of prosthetic wear.
Historically, above-knee amputations were often performed as emergent, life-saving measures without the benefit of modern advanced cross-sectional imaging. For instance, in the early 1960s, a surgeon treating an osteosarcoma of the distal femur lacked accurate imaging modalities to ascertain extraosseous extension. The extent of the soft tissue component and the relation of the tumor to the neurovascular bundle were assessed intraoperatively using an extensile incision; only then was the decision to proceed with an amputation finalized. Today, advanced magnetic resonance imaging (MRI), computed tomography (CT) angiography, and the consensus of multidisciplinary tumor boards dictate these decisions preoperatively with high precision.
Beyond oncologic indications, the vast majority of transfemoral amputations in modern practice are performed for end-stage peripheral vascular disease (PVD), often complicated by poorly controlled diabetes mellitus and superimposed, ascending polymicrobial infections. Trauma, including severe crush injuries, blast trauma, or unsalvageable mangled extremities, represents another significant demographic, particularly in younger, high-energy patient populations. Regardless of the underlying etiology, the overarching goal of the procedure remains consistent: absolute eradication of the pathology, meticulous handling of the soft tissue envelope, and the biomechanical optimization of the residual limb to facilitate postoperative mobility and minimize energy expenditure.
Surgical Anatomy and Biomechanics
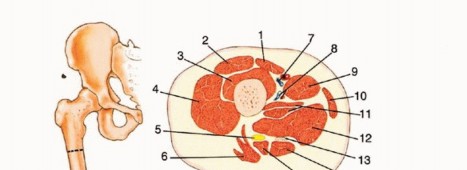
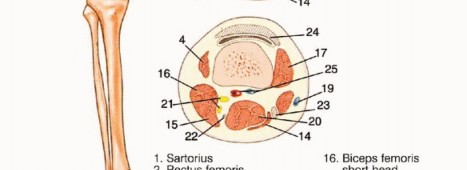
The operating surgeon must possess an intimate familiarity with the major neurovascular structures of the thigh, as these structures dictate the dissection planes and require meticulous identification, isolation, and secure ligation. The femoral artery serves as the primary arterial supply to the anterior thigh and femur. Its course and relationship to surrounding musculature change dynamically throughout the length of the femur, meaning its exact anatomical location varies depending on the elected level of amputation.
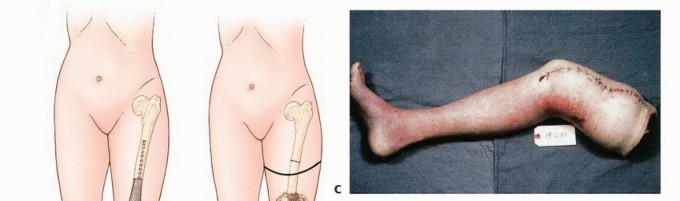
Above-knee amputations are generally classified by their anatomical level: high (just below the lesser trochanter), standard or midfemur (diaphyseal), or low distal femur (supracondylar). In principle, transfemoral amputations preserving 50% to 70% of the residual bone length (measured from the tip of the greater trochanter to the lateral femoral condyle) provide the optimal lever arm for prosthetic fitting and biomechanical advantage. However, in the oncologic setting, the length of the remaining femur is strictly dictated by the proximal extent of the tumor and the necessity of achieving wide, negative oncologic margins. Generally, if a minimum of 3 to 5 cm of bone distal to the lesser trochanter is preserved, a standard transfemoral prosthesis can still be utilized, albeit with significantly increased energy expenditure for the patient.
High Above Knee Amputation
At the level of a high above-knee amputation, the proximal femoral artery lies beneath the sartorius muscle, anterior to the adductor longus muscle, and anterior to the femur within the femoral triangle. The profunda femoris artery, responsible for supplying the posterior and lateral compartments, lies deep and posterior to the adductor longus muscle. At this proximal level, the femoral artery is positioned lateral to the femoral vein. The sciatic nerve lies posterior to the adductor magnus and anterior to the long head of the biceps femoris. Meticulous, controlled dissection in this region is critical to avoid the catastrophic retraction of these large-caliber vessels into the pelvis, which can result in life-threatening, uncontainable hemorrhage.
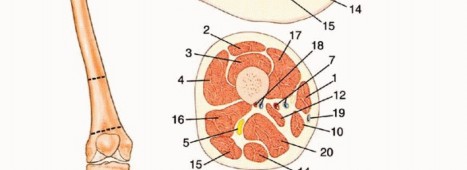
Midfemur Above Knee Amputation
In the midthigh area (diaphyseal level), the neurovascular anatomy transitions. The femoral artery enters the adductor (Hunter's) canal, lying between the vastus medialis anteriorly and the adductor magnus posteriorly, positioned medial to the femur. In this region, the femoral vein has typically rotated to lie lateral and slightly posterior to the artery. The sciatic nerve maintains its posterior position, lying between the short head of the biceps femoris and the semimembranosus.
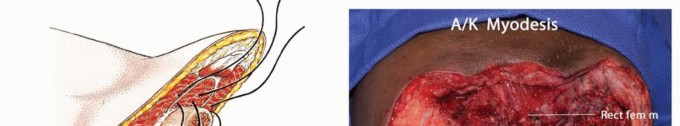
Biomechanical Considerations
The biomechanics of the transfemoral amputee are fundamentally and permanently altered due to the loss of the knee joint and the disruption of normal muscular insertions. The adductor magnus is the primary adductor of the thigh, inserting along the entire length of the linea aspera and the adductor tubercle. When the femur is transected, the adductor magnus loses its distal mechanical attachment. If it is not surgically reattached under physiological tension (myodesis), the powerful hip abductors (gluteus medius and minimus) will overpower the remaining adductor musculature. This imbalance inevitably leads to a classic abduction contracture.
Furthermore, the unopposed pull of the iliopsoas will lead to a severe hip flexion contracture. Energy expenditure during ambulation for a unilateral transfemoral amputee is approximately 65% higher than that of a non-amputee (and up to 120% higher for vascular amputees). This underscores the critical need for a surgically optimized, well-balanced residual limb that preserves the adductor moment arm.
Indications and Contraindications
The decision to proceed with a transfemoral amputation must be weighed carefully against the potential for limb salvage, taking into account the patient's physiological reserve and rehabilitation potential. Absolute indications include unsalvageable ischemic limbs with diffuse, non-reconstructable vascular disease, aggressive malignant tumors where limb-sparing surgery would compromise oncologic margins or leave a flail, functionless extremity, and life-threatening infections such as ascending necrotizing fasciitis or overwhelming sepsis originating from the lower extremity.
Relative indications include severe crush injuries with massive soft tissue and bone loss (often quantified by a Mangled Extremity Severity Score [MESS] greater than 7), chronic intractable osteomyelitis of the femur or knee joint, and severe, rigid knee flexion contractures in non-ambulatory patients where nursing care and perineal hygiene are severely compromised.
Contraindications are primarily relative and center on the patient's overall physiologic inability to survive the surgical insult (e.g., profound cardiopulmonary collapse). However, in cases of ascending wet gangrene or systemic sepsis, amputation is an emergent life-saving measure, and medical optimization must occur concurrently with surgical intervention. A primary contraindication to an above-knee amputation is the clinical viability of a below-knee amputation (BKA). Preserving the anatomical knee joint should always be prioritized if the soft tissue envelope and vascular supply permit, as a BKA drastically reduces the energy cost of ambulation and significantly improves the likelihood of independent household or community ambulation.
| Clinical Scenario | Indication for Primary Above Knee Amputation | Indication for Limb Salvage or Below Knee Amputation |
|---|---|---|
| Oncology | Neurovascular bundle encasement; massive extraosseous extension preventing negative margins; fungating tumors. | Tumor resectable with wide negative margins; reconstructable bone/soft tissue defects (e.g., endoprosthesis). |
| Vascular | Non-reconstructable arterial disease with profound ischemia extending proximal to the tibial plateau. | Patent popliteal artery; adequate distal perfusion for below-knee flap healing (TcPO2 > 30 mmHg). |
| Trauma | Complete sciatic nerve transection with massive femoral bone loss, severe crush, and prolonged warm ischemia. | Intact plantar sensation; reconstructable vascular injury; viable, sensate soft tissue envelope. |
| Infection | Ascending necrotizing fasciitis reaching the knee joint; chronic knee sepsis with catastrophic, uncontained bone loss. | Localized osteomyelitis amenable to aggressive debridement, dead space management, and targeted antibiotic therapy. |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is dictated by the specific etiology of the amputation. In patients with peripheral vascular disease, non-invasive vascular studies are mandatory. Ankle-Brachial Indices (ABI) and transcutaneous oxygen tension (TcPO2) measurements provide objective data regarding healing potential. A TcPO2 greater than 30 mmHg at the planned level of incision is generally predictive of successful wound healing, whereas levels below 20 mmHg are associated with a high rate of flap necrosis. CT angiography or standard angiography may be required to delineate the exact level of arterial occlusion and rule out proximal aortoiliac disease that might compromise even a transfemoral stump.
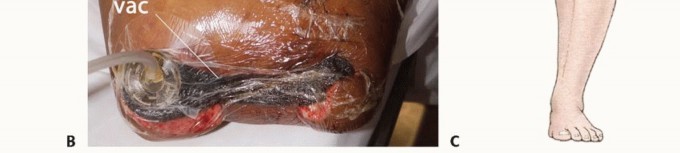
In the oncologic setting, advanced cross-sectional imaging is paramount. A dedicated MRI of the entire femur is required to identify intraosseous skip lesions and delineate the exact proximal extent of the tumor marrow edema. The planned bone resection level should be measured precisely from a fixed radiographic landmark (such as the joint line or greater trochanter) to ensure adequate oncologic margins while maximizing residual limb length.
Patient Positioning and Preparation
The patient is placed in the supine position on a standard radiolucent operating table. A bump or sandbag is often placed under the ipsilateral hemipelvis to internally rotate the leg to a neutral position, counteracting the natural external rotation of the hip and facilitating ergonomic access to the medial, anterior, and lateral compartments. If the amputation is performed for an infectious or oncologic etiology, the limb should be handled with extreme care to prevent the systemic dissemination of septic emboli or tumor cells.
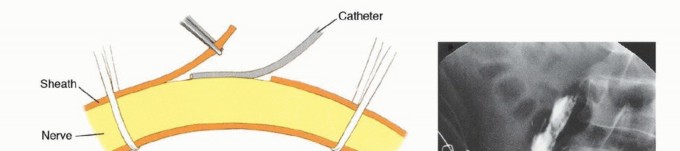
A sterile pneumatic tourniquet is placed as proximally as possible on the thigh to minimize blood loss. In cases of high above-knee amputations, proximal tumors, or severe proximal vascular calcification, a sterile tourniquet may not be feasible. In such scenarios, the surgeon must be prepared for proximal vascular control, occasionally requiring a separate inguinal incision to isolate and control the common femoral artery and vein with vessel loops before addressing the thigh. The entire lower extremity, including the hemipelvis, is prepped and draped free to allow for circumferential access and dynamic manipulation of the limb during the procedure.
Detailed Surgical Approach and Technique
The surgical technique for a transfemoral amputation must prioritize meticulous tissue handling, absolute hemostasis, secure vascular control, and the creation of a robust myodesis. The standard approach utilizes a fish-mouth incision, creating equal anterior and posterior myocutaneous flaps. However, in cases of severe vascular disease, trauma, or specific oncologic resections, atypical flaps (such as a long posterior flap or skewed sagittal flaps) may be necessitated by the availability of viable, well-perfused tissue.
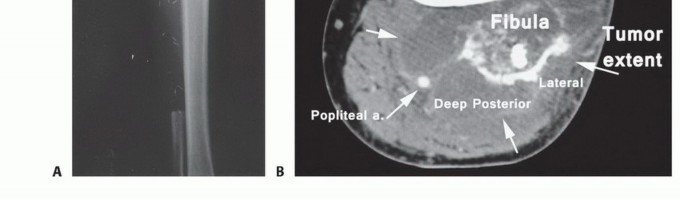
Incision and Dissection
The skin incisions are marked based on the anticipated level of bone transection. The flaps must be designed so that their combined length is slightly greater than the diameter of the thigh at the level of bone resection; this ensures a tension-free closure over the distal femur. The incision is carried down cleanly through the skin, subcutaneous tissue, and deep investing fascia in a single plane to prevent devascularization of the skin edges.
The anterior compartment is addressed first. The quadriceps muscle group (rectus femoris, vastus lateralis, vastus intermedius, vastus medialis) is transected, deliberately beveling the cut distally so that the bulky muscle can later be mobilized over the distal end of the femur. As the dissection proceeds medially, the superficial femoral artery and vein are identified within the adductor canal (in midfemur amputations) or beneath the sartorius (in high amputations).
Vascular and Neural Management
The femoral artery and vein must be carefully isolated from their surrounding adventitia, doubly ligated with heavy non-absorbable suture (e.g., 0-Silk or 1-0 Prolene), and transected. Suture ligation or transfixation sutures are strongly recommended for the artery to prevent catastrophic postoperative hemorrhage, particularly when the patient's blood pressure normalizes postoperatively. The profunda femoris vessels are similarly identified, isolated, and ligated as they are encountered posterior to the adductor longus.
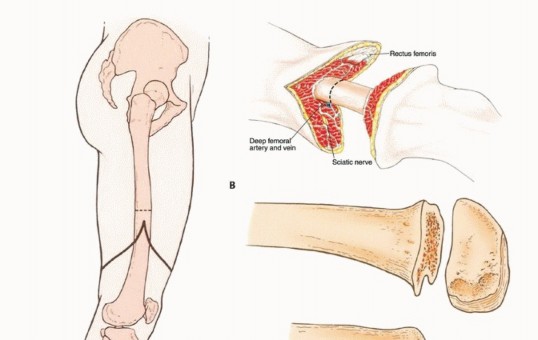
The sciatic nerve is identified in the posterior compartment. Proper management of the sciatic nerve is critical to minimize the risk of debilitating, symptomatic neuroma formation. The nerve is gently mobilized, drawn distally under mild tension, ligated sharply, and allowed to retract deeply into the proximal muscle beds, well away from the distal stump and the planned surgical scar. Modern techniques increasingly utilize Targeted Muscle Reinnervation (TMR), where the transected nerve stumps are surgically coapted to nearby redundant motor nerve branches. TMR provides a physiological target for regenerating axons, significantly reducing neuroma formation and potentially providing high-fidelity signals for advanced myoelectric prostheses.
Bone Transection and Myodesis
The femur is cleared of periosteum circumferentially at the planned osteotomy site. A Gigli saw or an oscillating power saw is used to transect the femur, ensuring continuous saline irrigation to prevent thermal necrosis of the bone. The sharp cortical edges, particularly the prominent linea aspera posteriorly and the anterior cortex, must be meticulously rasped, beveled, and contoured to prevent pressure points against the overlying soft tissue and the future prosthetic socket.
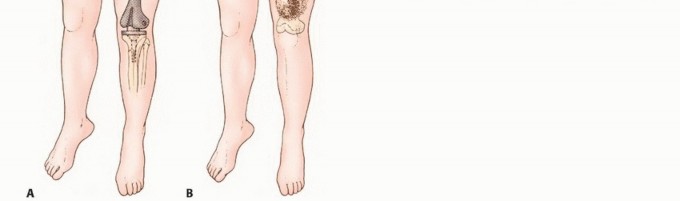
Myodesis is arguably the most critical step for ensuring a functional, biomechanically sound outcome. The Gottschalk technique is highly recommended. Drill holes are placed through the distal lateral and anterior cortices of the femur. The adductor magnus is brought across the distal end of the femur under physiological tension and secured rigidly to the lateral cortex using heavy non-absorbable sutures. This vital step restores the adductor moment arm, counteracting the hip abductors and preventing an abduction contracture. The quadriceps tendon is then brought distally and sutured to the posterior femur, and the posterior hamstrings are sutured to the anterior fascia, creating a robust, well-padded distal soft-tissue envelope.
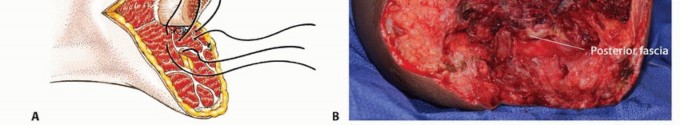
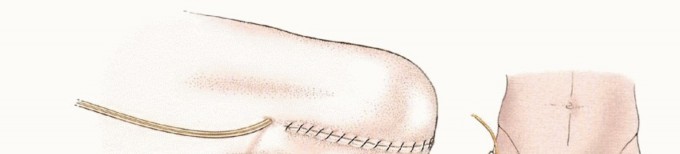
Closure
Following copious irrigation with sterile saline and meticulous hemostasis (which must be achieved and verified after tourniquet deflation), a deep subfascial closed-suction drain is typically placed to prevent hematoma formation. The deep fascia is closed with interrupted absorbable sutures, ensuring absolutely no tension is placed on the overlying skin. The subcutaneous layer and skin are closed meticulously, often using non-absorbable nylon sutures or surgical staples depending on skin quality. A well-padded, rigid or semi-rigid dressing is applied in the operating room to control edema, protect the surgical site from trauma, and prevent early flexion contractures.
Complications and Management
Transfemoral amputations carry a significant risk of perioperative and long-term complications, largely dependent on the patient's baseline physiologic status, nutritional reserve, and the underlying etiology. Vascular and diabetic patients face exponentially higher risks of wound dehiscence and infection compared to younger trauma or oncology patients.
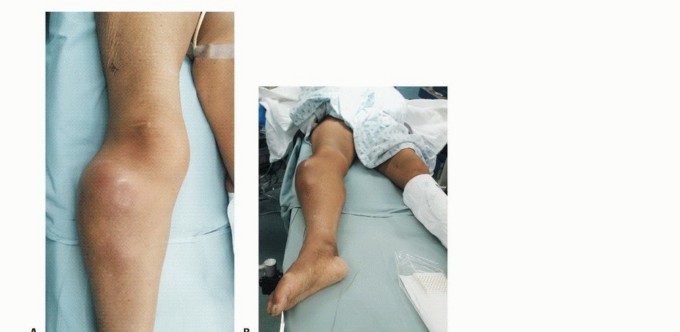
Postoperative hematoma is a frequent complication that can compromise flap viability by increasing local tissue tension and can serve as an ideal nidus for deep infection. Meticulous hemostasis prior to closure, careful ligation of muscular bleeders, and the judicious use of closed suction drains mitigate this risk. Wound dehiscence or full-thickness flap necrosis usually results from inadequate vascular supply to the chosen flaps, excessive tension during fascial closure, or early postoperative trauma to the residuum.
Long-term complications include hip flexion and abduction contractures, which severely limit prosthetic fitting and ambulation. These are prevented by rigorous surgical myodesis and early, aggressive postoperative physical therapy. Phantom limb pain and symptomatic neuromas are complex neuro-pathologic phenomena that require a multimodal approach, including pharmacological management (gabapentinoids, SNRIs, tricyclic antidepressants) and potentially surgical revision or secondary Targeted Muscle Reinnervation.
| Complication | Estimated Incidence | Etiology and Risk Factors | Prevention and Salvage Strategies |
|---|---|---|---|
| Wound Dehiscence / Necrosis | 10% - 25% | Poor perfusion (low TcPO2), excessive closure tension, early mechanical trauma to stump. | Optimize pre-op vascular status; ensure tension-free fascial closure; utilize rigid dressings; salvage via wedge excision or revision to a higher level. |
| Deep Infection | 10% - 20% | Contamination (trauma/open fractures), immunocompromise (diabetes), hematoma formation. | Preoperative weight-based antibiotics; meticulous debridement; deep drains; salvage via operative washout, debridement, and delayed primary closure. |
| Flexion/Abduction Contracture | 15% - 30% | Failure to perform rigid myodesis; prolonged wheelchair use; lack of early physical therapy. | Rigid adductor myodesis (Gottschalk); early prone positioning; aggressive physical therapy; salvage via surgical release (rarely successful). |
| Symptomatic Neuroma | 10% - 25% | Superficial nerve transection; entrapment of nerve ends in scar tissue or bone. | High traction neurectomy; burying nerve ends deep in healthy muscle; Targeted Muscle Reinnervation (TMR); salvage via surgical neuroma excision. |
| Heterotopic Ossification | 5% - 15% | Bone dust left in wound bed; severe blast injuries or high-energy trauma. | Meticulous, copious lavage of bone dust after osteotomy; salvage via surgical excision once the bone is mature, if it interferes with prosthetic socket fit. |

Post Operative Rehabilitation Protocols
Rehabilitation following a transfemoral amputation is a continuous, phased process requiring a dedicated multidisciplinary team comprising orthopedic surgeons, physiatrists, prosthetists, and physical therapists. The acute postoperative phase focuses strictly on wound healing, edema control, pain management, and contracture prevention.
Patients are encouraged to avoid prolonged sitting in wheelchairs or beds with the hip flexed, and are instructed to spend time in the prone position daily to actively stretch the hip flexors. Rigid removable dressings or immediate postoperative prostheses (IPOP) can be utilized to protect the residuum, significantly reduce edema, and psychologically benefit the patient by allowing early simulated weight-bearing and upright positioning.
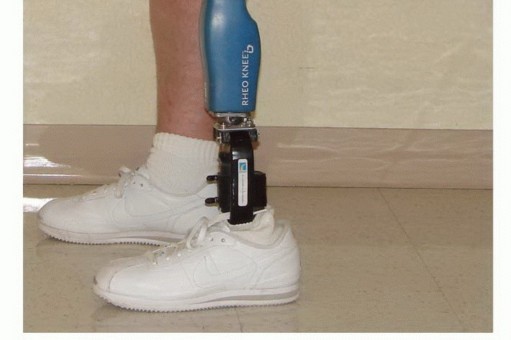
The pre-prosthetic training phase focuses on core strengthening, upper extremity conditioning, and progressive desensitization of the residuum. Once the surgical wound is completely healed and the stump volume has stabilized (typically 4 to 8 weeks postoperatively), the patient is casted for a preparatory (temporary) prosthesis. Gait training with a transfemoral prosthesis is highly energy-demanding; therefore, cardiovascular conditioning is a critical component of the rehabilitation protocol. The transition to a definitive prosthesis occurs once the residual limb has undergone maximal atrophy and volumetric stabilization, which can take up to 12 to 18 months post-amputation.