Comprehensive Introduction and Patho-Epidemiology
Cerebral palsy (CP) represents a heterogeneous group of permanent, non-progressive disorders of movement and posture resulting from a non-progressive disturbance that occurred in the developing fetal or infant brain. Among the myriad of musculoskeletal manifestations associated with spastic diplegic and quadriplegic CP, the development of a progressive crouch gait remains one of the most debilitating. Crouch gait is biomechanically defined by excessive sagittal-plane knee flexion during the stance phase of the gait cycle, almost universally accompanied by compensatory hip flexion and variable degrees of ankle involvement, ranging from equinus to calcaneus depending on the integrity of the plantarflexor-knee extension couple. The management of this complex gait deviation requires a profound understanding of dynamic muscle overactivity, myostatic contracture evolution, and the principles of Single-Event Multilevel Surgery (SEMLS).
The natural history of crouch gait in the spastic patient is a predictable cascade of biomechanical failures. In the ambulatory child typically under the age of six, the initial presentation is often a "jump gait." This is characterized by dynamic spasticity of the hamstrings and hip flexors, combined with ankle equinus. At this stage, the muscle fibers maintain relatively normal resting lengths, but their stretch reflex thresholds are pathologically lowered. As the child grows, the skeletal system lengthens at a rate that the spastic, under-stimulated muscle-tendon units cannot match. This discrepancy transitions the dynamic overactivity into a fixed, myostatic contracture. The hamstrings, particularly the medial group, become permanently foreshortened, tethering the knee in flexion and dragging the pelvis into a compensatory posterior tilt if the contracture is severe enough, or exacerbating anterior pelvic tilt if hip flexor contractures dominate.
As the knee flexion contracture solidifies, usually between the ages of six and ten, the biomechanical demands on the lower extremity skyrocket. The ground reaction force vector falls progressively posterior to the center of rotation of the knee joint. This generates an exponentially increasing external flexion moment that must be counteracted by an internal extension moment generated by the quadriceps. The rectus femoris and the vasti are forced to fire continuously and at near-maximal capacity throughout stance to prevent collapse. This chronic over-activation leads to early fatigue, severely diminished walking endurance, and devastating patellofemoral joint reaction forces. Over time, this unrelenting pressure results in patella alta, fragmentation of the inferior pole of the patella, chondromalacia, and intractable anterior knee pain, often culminating in the complete loss of independent ambulation by early adulthood.
Concurrently, we frequently observe a "stiff-knee gait" during the swing phase, primarily driven by spasticity and inappropriate firing of the rectus femoris. Normally, the rectus femoris should be silent during early swing to allow for adequate knee flexion for foot clearance. In CP, prolonged firing of the rectus acts as a tether, severely limiting peak swing-phase knee flexion, causing the foot to drag and increasing the risk of tripping. Therefore, addressing crouch gait comprehensively often requires a dual approach: distal hamstring lengthening to correct the stance-phase flexion contracture, and a rectus femoris transfer to address the swing-phase stiffness. This intraoperative masterclass will detail the meticulous execution of both procedures within the context of a SEMLS intervention.
Detailed Surgical Anatomy and Biomechanics
A masterful surgical intervention in the spastic lower extremity demands an intimate, three-dimensional understanding of the myoarchitecture and neurovascular topography of the thigh. We are not merely cutting tendons; we are recalibrating biomechanical levers. The hamstrings and the rectus femoris are biarticular muscles, meaning their influence spans both the hip and the knee, making their surgical manipulation highly impactful on global gait kinematics.
The Medial Hamstring Group
The medial hamstrings—comprising the gracilis, semitendinosus, and semimembranosus—are the primary culprits in the development of spastic knee flexion contractures. The gracilis originates from the inferior pubic ramus and inserts onto the anteromedial proximal tibia as part of the pes anserinus. Innervated by the obturator nerve, its myoarchitecture is characterized by a relatively small physiologic cross-sectional area and a very large ratio of muscle fiber length to tendon length. This design indicates a muscle built for maximal excursion rather than massive force generation. Because of this architecture, the gracilis is highly amenable to simple tenotomy or fractional lengthening without significant risk of functional weakness.
The semitendinosus shares a similar excursion-dominant myoarchitecture. Originating from the inferolateral ischial tuberosity and inserting just posterior to the gracilis on the pes anserinus, it is innervated by the tibial division of the sciatic nerve. It possesses a long, distinct, cord-like distal tendon that is easily palpable and visible intraoperatively. Like the gracilis, its long muscle fibers and smaller cross-sectional area make it an ideal candidate for Z-lengthening or tenotomy. During dissection, it is critical to identify and release the fascial bands that frequently tether the semitendinosus to the gastrocnemius fascia, as failure to do so will result in under-correction of the contracture.
The semimembranosus, however, presents a completely different biomechanical and architectural profile. Originating superior to the semitendinosus on the ischium, it inserts via a complex expansion onto the posteromedial tibia and posterior knee capsule. Crucially, its myoarchitecture features a large physiologic cross-sectional area and short muscle fibers, indicating a muscle designed for high force generation and minimal excursion. Its distal aspect is characterized by a broad, thick aponeurosis rather than a discrete cord-like tendon. Because it is a primary force generator for hip extension, completely transecting or aggressively Z-lengthening the semimembranosus can lead to devastating weakness and an iatrogenic anterior pelvic tilt. Therefore, it must strictly be managed with an intramuscular aponeurotic lengthening (fractional lengthening) to preserve its structural integrity and force-generating capacity.
The Lateral Hamstrings and Rectus Femoris
While the lateral hamstrings (biceps femoris) are less frequently targeted in primary SEMLS for crouch gait, their anatomy must be respected. The biceps femoris consists of a long head (originating from the ischium) and a short head (originating from the linea aspera of the femur), inserting conjointly onto the fibular head. The critical neurovascular consideration here is the common peroneal nerve, which intimately hugs the posteromedial border of the biceps tendon before wrapping around the fibular neck. Any surgical approach to the lateral hamstrings requires meticulous neurolysis and protection of this nerve to prevent devastating foot drop.
The rectus femoris is the sole biarticular muscle of the quadriceps complex. Originating from the anterior inferior iliac spine (straight head) and the superior acetabular rim (reflected head), it converges into the common quadriceps tendon to insert on the patella. Innervated by the femoral nerve, it functions as both a hip flexor and a knee extensor. In the spastic patient, inappropriate firing of the rectus femoris during the swing phase prevents the knee from flexing, leading to a stiff-knee gait. By transferring the distal tendon of the rectus femoris—typically posterior to the axis of the knee joint, often to the sartorius or semitendinosus stump—we convert it from a swing-phase knee extensor into a knee flexor, thereby dramatically improving swing-phase kinematics and foot clearance without sacrificing overall stance-phase extension power, which is maintained by the vasti.
Exhaustive Indications and Contraindications
The decision to perform distal hamstring lengthening and rectus femoris transfer must be derived from a rigorous, multi-modal diagnostic algorithm. Clinical examination alone is insufficient; it must be corroborated by standardized functional assessments and, ideally, three-dimensional Quantitative Gait Analysis (QGA). The timing of the intervention is equally critical, aiming to intervene when contractures become fixed but before irreversible bony deformity or joint degeneration occurs.
Indications for Surgery
The primary indication for medial hamstring lengthening is a fixed knee flexion contracture that significantly impairs gait efficiency and cannot be managed conservatively. Clinically, this is defined by a popliteal angle greater than 45 to 50 degrees and a unilateral straight-leg raise of less than 60 degrees. These measurements must be confirmed under anesthesia (EUA) to eliminate the dynamic spastic component and reveal the true myostatic contracture. On QGA, we look for persistent knee flexion greater than 20 degrees at initial contact and throughout the stance phase, coupled with an increased internal knee extension moment.
The indication for a rectus femoris transfer is a stiff-knee gait pattern during the swing phase. Clinically, this may present as a positive Duncan-Ely test, where rapid passive flexion of the knee in the prone position elicits spontaneous hip flexion, indicating rectus spasticity. On QGA, the hallmark of rectus overactivity is diminished peak knee flexion during swing (typically less than 40-45 degrees, whereas normal is roughly 60 degrees) and delayed timing of peak swing knee flexion. Dynamic electromyography (EMG) is highly valuable here, demonstrating prolonged or continuous firing of the rectus femoris throughout the swing phase.
| Parameter | Indication for Hamstring Lengthening | Indication for Rectus Transfer |
|---|---|---|
| Clinical Exam | Popliteal angle > 45°-50°; SLR < 60° | Positive Duncan-Ely test |
| Kinematics (QGA) | Stance-phase knee flexion > 20° | Peak swing knee flexion < 45°; Delayed peak |
| Kinetics (QGA) | Increased internal knee extension moment | N/A |
| Dynamic EMG | Prolonged stance-phase medial hamstring firing | Continuous/prolonged swing-phase rectus firing |
| EUA | Confirmed fixed myostatic contracture | Confirmed rectus tethering |
Contraindications and Cautions
Absolute contraindications include purely dynamic spasticity without fixed contracture in a young child (typically under 6 years old). In these cases, aggressive physical therapy, bracing, and targeted botulinum toxin type A injections are the standard of care. Surgical lengthening of a purely dynamic muscle often leads to devastating over-lengthening, resulting in a severe genu recurvatum (back-knee) deformity and profound weakness.
Relative contraindications include severe, global lower extremity weakness where the patient relies on the spastic tethering for upright stability. Furthermore, if the patient has developed severe, fixed bony deformities—such as a fixed structural knee flexion contracture greater than 15-20 degrees that does not correct under anesthesia—soft tissue lengthening alone will fail. These patients require concomitant distal femoral extension osteotomies (DFEO) and potentially patellar tendon advancements (PTA) to restore the skeletal lever arm, with soft tissue procedures serving only an adjunctive role. Finally, isolated hamstring lengthening in the presence of severe, unaddressed ankle plantarflexion contractures will inevitably drive the patient into profound genu recurvatum, highlighting the absolute necessity of the SEMLS approach.
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock of successful outcomes in pediatric orthopaedic deformity correction. The "diagnostic matrix" must be synthesized to formulate a precise surgical blueprint. This begins with a meticulous review of the patient's clinical history, focusing on functional decline, increasing reliance on assistive devices, and the onset of anterior knee pain.
Diagnostic Imaging and Gait Analysis
Standardized standing radiographs are mandatory. An anteroposterior (AP) pelvis film assesses for hip subluxation or dysplasia, which frequently co-occurs in spastic diplegia and may alter the surgical plan. Standing long-leg alignment films evaluate for coronal plane malalignment. Crucially, a standing lateral radiograph of the knee is scrutinized for patella alta (measured via the Insall-Salvati or Caton-Deschamps ratios) and fragmentation of the inferior pole of the patella. The presence of these radiographic findings confirms chronic patellofemoral overload and solidifies the indication for intervention to rescue the joint.
Quantitative Gait Analysis (QGA) is the gold standard for surgical planning in CP. It provides objective, dynamic data that clinical examination simply cannot yield. By analyzing the kinematic graphs, we can precisely quantify the degree of stance-phase crouch and swing-phase stiffness. The kinetic data reveals the immense forces acting across the knee joint, justifying the need to restore the ground reaction force vector to a more physiological position. Dynamic EMG confirms which specific muscles are firing inappropriately, allowing us to selectively target the medial hamstrings and the rectus femoris while sparing muscles that are functioning normally or compensating appropriately.
Patient Positioning and Operating Room Setup
The patient is brought to the operating theater and placed in the supine position on a standard radiolucent operating table. General endotracheal anesthesia is administered. Prior to prepping, an Examination Under Anesthesia (EUA) is rigorously performed. This is the final, critical checkpoint. With the patient fully relaxed, the popliteal angle and straight-leg raise are re-measured to confirm the degree of fixed myostatic contracture. If the contracture completely resolves under anesthesia, the surgical plan must be aborted or significantly modified to avoid over-lengthening.
A sterile pneumatic tourniquet is placed as proximally as possible on the thigh. While some surgeons prefer to operate without a tourniquet to assess muscle tension dynamically, the use of a tourniquet provides a bloodless field, which is highly advantageous when performing meticulous fascial dissections and isolating neurovascular structures during a complex SEMLS procedure. The entire lower extremity, from the iliac crest to the toes, is prepped and draped free. This extensive draping is non-negotiable; the surgeon must have the ability to freely flex and extend the hip and knee throughout the procedure to dynamically assess the effect of each sequential lengthening and transfer.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution requires a systematic, layered approach, respecting the distinct anatomical characteristics of each targeted muscle-tendon unit. We will begin with the medial hamstring lengthening to address the stance-phase crouch, followed by the rectus femoris transfer to address the swing-phase stiffness.
I. Medial Hamstring Lengthening
1. Incision and Superficial Dissection
A longitudinal incision, approximately 6 to 10 centimeters in length, is made over the posteromedial aspect of the distal thigh. The distal extent of the incision should terminate roughly two to three fingerbreadths proximal to the posterior skin crease of the popliteal fossa. This precise placement avoids the complex neurovascular structures of the popliteal space while providing excellent access to the musculotendinous junctions of the medial hamstrings.
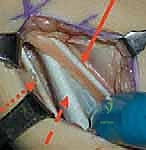
The dissection is carried sharply through the subcutaneous adipose tissue. Extreme vigilance is required here to identify and protect the saphenous vein and the saphenous nerve, which course superficially in the anteromedial thigh. The vein is often prominent and, if injured, can cause significant postoperative hematoma. The nerve must be gently retracted anteriorly to prevent traction neuropraxia, which can lead to painful neuromas or medial leg numbness.
2. Deep Dissection and Muscle Isolation
Upon incising the deep fascia, the sartorius muscle is the first major landmark encountered. It is characterized by its oblique course and distinct fascial sheath. The sartorius is retracted anteriorly, revealing the medial hamstring complex deep and posterior to it.
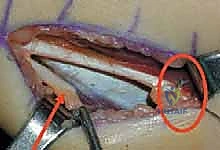
The gracilis is typically the most superficial and medial of the hamstring group. It is identified by its relatively small muscle belly and long, thin tendon. A right-angle clamp is passed deep to the gracilis tendon to isolate it. Given its myoarchitecture (high excursion, low force), the gracilis is commonly managed with a simple transverse tenotomy or a step-cut Z-lengthening if a more controlled elongation is desired.
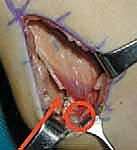
Immediately posterior and slightly deep to the gracilis lies the semitendinosus. It is unmistakable due to its thick, cord-like tendon. The semitendinosus is systematically isolated. It is imperative to identify and transect any aberrant fascial bands connecting the semitendinosus to the medial head of the gastrocnemius, as these will tether the knee even after the main tendon is lengthened.
Once fully mobilized, the semitendinosus tendon undergoes a Z-lengthening. The tendon is split longitudinally for 3 to 4 centimeters, and hemi-transections are made at opposite ends. The knee is then passively extended to achieve the desired length, and the tendon ends are sutured using a non-absorbable braided suture in a figure-of-eight fashion.
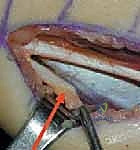
3. Aponeurotic Lengthening of the Semimembranosus
Deep and slightly proximal to the semitendinosus lies the robust semimembranosus muscle. As previously emphasized, this muscle must never be completely transected. We identify its broad, glistening, silvery aponeurosis at the musculotendinous junction.
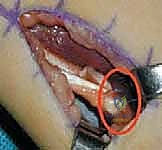
Using a #15 blade, two or three transverse, staggered incisions are made strictly through the aponeurotic fibers, ensuring the underlying muscle fibers are left completely intact. This is the essence of a fractional, or intramuscular, lengthening.
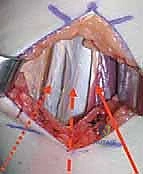
With the hip flexed to 90 degrees, the knee is slowly and firmly extended. The surgeon will feel and see the aponeurotic cuts separate and slide, effectively lengthening the muscle-tendon unit while preserving its continuity and force-generating capability. The goal is to achieve a popliteal angle of approximately 20 to 30 degrees. Over-lengthening to 0 degrees is strictly avoided to prevent postoperative genu recurvatum.

II. Rectus Femoris Transfer
1. Anterior Approach and Rectus Isolation
Attention is then turned to the anterior thigh. A 5 to 7-centimeter longitudinal incision is made over the distal anterior thigh, centered over the quadriceps tendon, ending just proximal to the superior pole of the patella. The subcutaneous tissue is divided, and the deep fascia is incised in line with the skin incision.
The interval between the rectus femoris and the underlying vastus intermedius, as well as the medial/lateral borders with the vastus medialis and lateralis, must be meticulously developed. The rectus femoris tendon is identified; it is the most superficial, distinct tendinous structure of the quadriceps mechanism.

Using blunt and sharp dissection, the rectus tendon is freed from the underlying vasti. It is critical to dissect proximally enough to ensure adequate excursion for the transfer, typically mobilizing the muscle belly for several centimeters proximal to the musculotendinous junction.
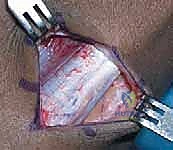
2. Distal Release and Transfer Routing
Once isolated, the distal tendon of the rectus femoris is sharply detached from its insertion on the common quadriceps tendon, as close to the superior pole of the patella as possible to maximize tendon length. A heavy, non-absorbable whipstitch (e.g., #2 FiberWire or Ethibond) is woven into the distal end of the detached tendon to secure it for transfer.
The routing of the transfer depends on the specific kinematic goals, but the most common transfer is to the medial side, attaching it to the sartorius or the stump of the semitendinosus. A large Kelly clamp or a tendon passer is introduced through the medial hamstring incision, tunneled subcutaneously over the anterior aspect of the medial thigh, and brought out through the anterior rectus incision. The pre-placed whipstitch is grasped, and the rectus tendon is pulled through the subcutaneous tunnel into the medial wound.
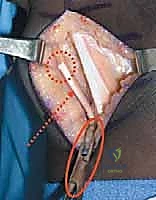
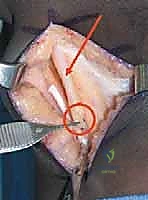
3. Tensioning and Fixation
The knee is positioned in approximately 30 to 45 degrees of flexion, and the hip is placed in neutral extension. The transferred rectus tendon is laid over the chosen recipient site (e.g., the sartorius muscle belly or the semitendinosus).
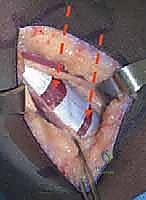
The tendon is pulled under moderate resting tension. It is then securely sutured to the recipient tissue using multiple interrupted, non-absorbable figure-of-eight sutures. The fixation must be robust enough to withstand the forces of early postoperative mobilization.
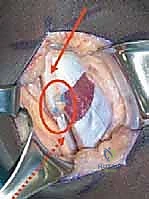
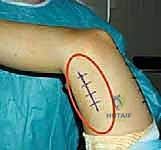
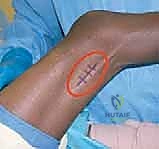
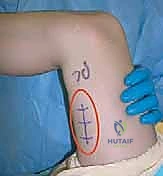
Following rigorous hemostasis upon tourniquet deflation, the wounds are irrigated copiously. The deep fascial layers are closed with absorbable sutures, and the skin is closed with a subcuticular stitch.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, soft tissue interventions in the spastic patient carry inherent risks. The margin between under-correction and devastating over-lengthening is exceptionally narrow, requiring precision and experience. Recognizing and managing these complications promptly is a hallmark of an expert orthopaedic surgeon.
The most feared complication of hamstring lengthening is over-lengthening, leading to iatrogenic genu recurvatum and an anterior pelvic tilt. This occurs when the hamstrings are excessively weakened, destroying their ability to act as posterior tethers. This is particularly disastrous if concomitant ankle equinus is left untreated, as the plantarflexed ankle forcefully drives the knee into hyperextension during stance. The management of severe recurvatum is highly complex, often requiring prolonged bracing, and in refractory cases, salvage bony procedures such as a proximal tibial flexion osteotomy.
Under-correction or recurrence of the crouch gait is also common, particularly during the adolescent growth spurt. Recurrence rates can range from 15% to 30% depending on the severity of the initial pathology and the patient's growth potential. This underscores the importance of delaying surgical intervention until the contractures are truly fixed and the child is older, ideally closer to skeletal maturity, though this must be balanced against the risk of joint damage from prolonged crouch. Management of recurrence often involves revision soft tissue lengthening or, more commonly, transitioning to bony procedures (DFEO) as the patient matures.
Neurovascular injuries, while rare, are highly morbid. Injury to the saphenous nerve during the medial approach can cause painful neuromas, while injury to the common peroneal nerve during a lateral hamstring approach results in foot drop. Meticulous surgical technique and precise anatomical knowledge are the only reliable preventative measures.
| Complication | Estimated Incidence | Prevention Strategy | Salvage Management |
|---|---|---|---|
| Over-lengthening / Genu Recurvatum | 5% - 15% | Avoid complete semimembranosus tenotomy; Address ankle equinus simultaneously; Target 20°-30° popliteal angle post-op. | Ground Reaction AFOs (GRAFOs); Proximal tibial flexion osteotomy in severe, rigid cases. |
| Under-correction / Recurrence | 15% - 30% | Ensure complete release of fascial bands; Delay surgery until fixed contracture is present; Rigorous post-op bracing. | Revision soft tissue lengthening; Distal Femoral Extension Osteotomy (DFEO). |
| Saphenous Nerve Injury | < 2% | Careful superficial dissection; Anterior retraction of the nerve; Avoid aggressive electrocautery near the skin. | Gabapentinoids; Desensitization therapy; Surgical neuroma excision if refractory. |
| Wound Dehiscence / Infection | 1% - 3% | Meticulous hemostasis; |
