Gabapentin Enacarbil (Horizant): A Comprehensive Medical SEO Guide
Welcome to this in-depth guide on Gabapentin Enacarbil, commonly known by its brand name Horizant. As an expert in medical SEO and orthopedic conditions, we aim to provide a definitive resource for patients, caregivers, and healthcare professionals seeking authoritative information on this unique medication. Horizant is an extended-release prodrug of gabapentin, specifically formulated to provide consistent therapeutic levels for the management of two distinct neurological conditions: Restless Legs Syndrome (RLS) and Postherpetic Neuralgia (PHN).
This guide will delve into the intricacies of Horizant, from its innovative mechanism of action and pharmacokinetic profile to detailed clinical indications, precise dosage guidelines, potential risks, and crucial safety information. Our goal is to empower you with comprehensive knowledge to understand Horizant's role in improving quality of life for those suffering from RLS and PHN.
Understanding Gabapentin Enacarbil (Horizant): Introduction & Overview
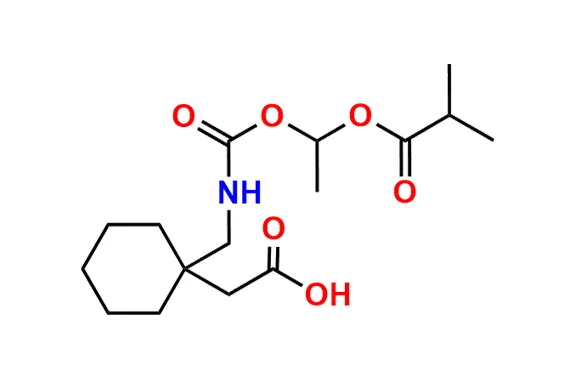
Gabapentin Enacarbil (Horizant) represents a significant advancement in the therapeutic landscape for certain chronic pain and neurological disorders. Unlike immediate-release gabapentin, Horizant is a prodrug designed to enhance bioavailability and maintain stable drug concentrations throughout the day or night, depending on the indication. This extended-release formulation minimizes fluctuations in plasma levels, which can contribute to more consistent symptom control and potentially reduce side effects associated with peak concentrations.
Approved by the U.S. Food and Drug Administration (FDA), Horizant offers a targeted approach for:
- Moderate-to-severe primary Restless Legs Syndrome (RLS): A neurological disorder characterized by an irresistible urge to move the legs, often accompanied by uncomfortable sensations, typically worsening in the evening or night.
- Postherpetic Neuralgia (PHN): A persistent, often debilitating neuropathic pain condition that can develop after a shingles (herpes zoster) rash has cleared.
The development of Horizant addresses some of the pharmacokinetic limitations of conventional gabapentin, providing a more predictable and sustained therapeutic effect. This guide will meticulously explore these aspects, ensuring a thorough understanding of its clinical utility.
Deep-Dive into Technical Specifications & Mechanisms
Understanding how Horizant works at a molecular level is crucial for appreciating its therapeutic benefits.
Mechanism of Action
Gabapentin Enacarbil is a prodrug that is rapidly hydrolyzed in the body to its active moiety, gabapentin. The mechanism of action of gabapentin itself is not fully elucidated but is believed to involve several pathways. Critically, gabapentin does not bind to GABA-A or GABA-B receptors, nor does it affect GABA uptake or degradation. Instead, its primary mechanism is thought to involve high-affinity binding to the alpha-2-delta (α2δ) subunit of voltage-gated calcium channels in the central nervous system.
This binding is hypothesized to modulate the release of excitatory neurotransmitters from presynaptic nerve terminals. By reducing the influx of calcium into these terminals, gabapentin can decrease the release of neurotransmitters such as glutamate, norepinephrine, and substance P. This effect is believed to contribute to its analgesic and anticonvulsant properties.
In the context of its approved indications:
- Restless Legs Syndrome (RLS): While the exact pathophysiology of RLS is complex and involves dopaminergic and iron dysregulation, the modulation of neurotransmitter release by gabapentin in specific brain regions may help to normalize neuronal excitability and alleviate the sensory and motor symptoms of RLS.
- Postherpetic Neuralgia (PHN): PHN is characterized by hyperexcitability of damaged sensory neurons. By reducing the overactivity of these neurons through its interaction with α2δ subunits, gabapentin can help to attenuate the chronic neuropathic pain signals.
Pharmacokinetics
The pharmacokinetic profile of Gabapentin Enacarbil is specifically engineered to overcome the variable and saturable absorption of conventional gabapentin.
- Absorption:
- Horizant is absorbed from the gastrointestinal tract via the L-amino acid transporter 1 (LAT1), an active transport system present in the small intestine. This transporter-mediated absorption allows for more efficient and less variable absorption compared to the saturable amino acid transport system utilized by conventional gabapentin.
- Following absorption, gabapentin enacarbil is rapidly and extensively hydrolyzed by esterases in the intestine and liver to gabapentin and enacarbil. Enacarbil is then eliminated.
- Peak plasma concentrations of gabapentin occur approximately 5 to 8 hours post-dose.
- Food Effect: Administration with food significantly increases the bioavailability of gabapentin from Horizant, making it a critical aspect of dosing instructions. Taking it with food increases the absorption rate and extent.
- Distribution:
- Gabapentin distributes widely throughout the body, including the central nervous system.
- It does not bind to plasma proteins.
- Metabolism:
- Gabapentin enacarbil is a prodrug; its active form is gabapentin. Gabapentin itself is not significantly metabolized in humans.
- Elimination:
- Gabapentin is eliminated from the systemic circulation exclusively by renal excretion as unchanged drug.
- The mean elimination half-life of gabapentin following Horizant administration ranges from approximately 6 to 9 hours. This extended half-life, combined with its sustained-release formulation, allows for once-daily or twice-daily dosing while maintaining stable therapeutic levels.
- Linearity:
- Gabapentin exposure (AUC and Cmax) from Horizant increases proportionally with dose, indicating linear pharmacokinetics over the therapeutic range.
This unique pharmacokinetic profile ensures a smoother, more predictable therapeutic effect, which is particularly beneficial for conditions requiring sustained symptom control.
Extensive Clinical Indications & Usage
Horizant is indicated for the treatment of specific neurological conditions, with clear guidelines for its administration.
Approved Indications
1. Moderate-to-Severe Primary Restless Legs Syndrome (RLS)
- Description: RLS is a chronic sensorimotor neurological disorder characterized by an irresistible urge to move the legs, usually accompanied by uncomfortable and unpleasant sensations (e.g., creeping, crawling, tingling, pulling, aching, or itching). These symptoms typically begin or worsen during periods of rest or inactivity, are partially or totally relieved by movement, and are worse in the evening or night.
- Horizant's Role: Horizant helps to alleviate the bothersome sensory symptoms and the urge to move the legs, leading to improved sleep quality and overall quality of life for RLS patients.
- Dosage for RLS:
- The recommended dose for RLS is 600 mg once daily, taken orally with food at approximately 5:00 PM.
- Initial Dose: Begin with 600 mg once daily.
- Titration: No titration is required.
- Administration: It is crucial to take Horizant with food to optimize absorption. Taking it in the evening aligns with the nocturnal worsening of RLS symptoms.
2. Postherpetic Neuralgia (PHN)
- Description: PHN is a long-lasting neuropathic pain complication of herpes zoster (shingles). It is defined as pain that persists for more than 3 months after the onset of the rash. The pain can be severe, burning, stabbing, or aching, and may be accompanied by allodynia (pain from normally non-painful stimuli) or hyperalgesia (increased pain from painful stimuli).
- Horizant's Role: Horizant effectively reduces the severity of PHN-associated pain, providing significant relief for patients suffering from this chronic condition.
- Dosage for PHN:
- The recommended dose for PHN is 1200 mg once daily (600 mg twice daily), taken orally with food.
- Initial Dose: Start with 600 mg once daily in the morning with food for 3 days.
- Titration:
- On Day 4, increase the dose to 600 mg twice daily (1200 mg per day), taken with food.
- The two doses should be separated by 8 to 12 hours. For example, a morning dose and an evening dose.
- Maximum Dose: The maximum recommended dose is 1200 mg per day.
- Administration: Always take with food to ensure optimal absorption.
General Dosing Guidelines
- Administration:
- Swallow tablets whole. Do not chew, crush, or split tablets. Altering the tablet can affect its extended-release properties and lead to rapid drug release.
- Always take Horizant with food.
- Renal Impairment:
- Dosage adjustment is necessary for patients with impaired renal function, as gabapentin is eliminated renally.
- The following table provides guidance for dose adjustments:
| Renal Function (Creatinine Clearance, mL/min) | RLS Recommended Dose | PHN Recommended Dose |
|---|---|---|
| ≥ 60 | 600 mg once daily at 5:00 PM | 600 mg twice daily (1200 mg/day) |
| 30 to 59 | 600 mg once daily at 5:00 PM | 600 mg once daily |
| < 30 | 300 mg once daily at 5:00 PM | 300 mg once daily |
| End-Stage Renal Disease (ESRD) on Hemodialysis | Not recommended due to lack of specific dosage studies | Not recommended due to lack of specific dosage studies |
- Missed Doses: If a dose is missed, take the next dose as scheduled. Do not take an extra dose to make up for the missed one.
- Discontinuation: If Horizant is discontinued, it should generally be tapered gradually over at least 1 week to minimize the potential for withdrawal symptoms.
Risks, Side Effects, & Contraindications
Like all medications, Horizant can cause side effects and carries certain risks. A thorough understanding of these is essential for safe and effective use.
Contraindications
Horizant is contraindicated in patients with a known hypersensitivity to gabapentin, gabapentin enacarbil, or any of the inactive ingredients in the formulation.
Warnings and Precautions
- Suicidal Thoughts and Behaviors: Antiepileptic drugs (AEDs), including gabapentin, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with Horizant should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, or any unusual changes in mood or behavior.
- Drowsiness and Sedation: Horizant can cause significant drowsiness and sedation. Patients should be cautioned about engaging in activities requiring complete mental alertness, such as driving or operating machinery, until they know how Horizant affects them.
- Dizziness and Somnolence: These are common dose-related side effects, particularly during the initial phase of treatment.
- Anaphylaxis and Angioedema: Rare but serious hypersensitivity reactions, including anaphylaxis and angioedema (swelling of the face, lips, tongue, and throat), have been reported with gabapentin. Patients should be instructed to seek immediate medical attention if they experience signs of these reactions.
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): DRESS, also known as multi-organ hypersensitivity, is a severe, potentially life-threatening systemic reaction. It typically presents with fever, rash, lymphadenopathy, and eosinophilia, often with involvement of other organ systems. Prompt recognition and discontinuation of the drug are crucial.
- Respiratory Depression: Serious, life-threatening, or fatal respiratory depression has been reported with gabapentin, particularly when co-administered with central nervous system (CNS) depressants like opioids, or in patients with underlying respiratory impairment or elderly patients. Patients should be monitored for signs of respiratory depression.
- Withdrawal Precipitated Seizure, Status Epilepticus: Abrupt discontinuation of gabapentin in patients with epilepsy may precipitate status epilepticus. Although Horizant is not indicated for epilepsy, withdrawal symptoms, including anxiety, insomnia, nausea, pain, and sweating, can occur with abrupt discontinuation. A gradual taper is recommended.
- Risk of Tumors in Animal Studies: In carcinogenicity studies, gabapentin caused an increase in the incidence of pancreatic acinar cell adenomas and carcinomas in rats. The clinical significance of these findings for humans is unknown.
- Effect on Diagnostic Tests: False positive results have been reported in the Ames-2000 urine protein test when gabapentin was added to other anticonvulsant agents.
Common Side Effects
The most frequently reported adverse reactions (≥5% and greater than placebo) in clinical trials for Horizant are listed below:
| Condition Treated | Common Side Effects |
|---|---|
| RLS | Somnolence, Dizziness, Nausea, Fatigue, Dry Mouth, Diarrhea |
| PHN | Dizziness, Somnolence, Nausea, Headache, Constipation, Edema Peripheral |
These side effects are generally mild to moderate and often diminish with continued treatment. However, if they persist or worsen, patients should consult their healthcare provider.
Serious Side Effects
While less common, some side effects require immediate medical attention:
- Severe allergic reactions (rash, hives, difficulty breathing, swelling of face/lips/tongue/throat)
- Suicidal thoughts or actions
- Changes in mood or behavior (e.g., aggression, hostility, agitation)
- Unusual weakness or tiredness
- Severe dizziness or loss of coordination
- Unexplained muscle pain or weakness
- Signs of DRESS (fever, rash, swollen lymph nodes, swelling in the face, yellowing of skin or eyes)
- Signs of respiratory depression (slow, shallow breathing)
Drug Interactions
- Opioids and Other CNS Depressants (e.g., benzodiazepines, alcohol, sedating antihistamines): Co-administration with opioids or other CNS depressants can significantly increase the risk of respiratory depression, profound sedation, coma, and death. If co-administration is necessary, the lowest effective dose of Horizant and the CNS depressant should be used, and patients should be closely monitored for signs of respiratory depression and sedation.
- Antacids: Unlike immediate-release gabapentin, the absorption of gabapentin from Horizant is not significantly affected by co-administration with antacids containing aluminum and magnesium.
- Alcohol: Concomitant use with alcohol can intensify CNS side effects such as drowsiness and dizziness. Patients should be advised to avoid or limit alcohol consumption while taking Horizant.
Pregnancy and Lactation
- Pregnancy: There are no adequate and well-controlled studies of Horizant in pregnant women. Animal studies have shown developmental toxicity (e.g., skeletal abnormalities, increased fetal loss) at doses higher than those used clinically. Horizant should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Pregnancy Exposure Registry: A pregnancy exposure registry is available for women exposed to gabapentin during pregnancy. Healthcare providers are encouraged to register patients by calling the North American Antiepileptic Drug (NAAED) Pregnancy Registry at 1-888-233-2334.
- Lactation: Gabapentin is excreted into human milk. The effect of gabapentin on the breastfed infant and on milk production is unknown. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. Potential for sedation and poor feeding in the infant exists.
Overdose Management
Experience with Horizant overdose is limited. However, overdose with gabapentin has resulted in:
- Symptoms: Dizziness, double vision, slurred speech, drowsiness, lethargy, and diarrhea. In some cases, coma or death has been reported, particularly when combined with other CNS depressants.
- Treatment: There is no specific antidote for Horizant overdose. Management should be supportive, including general supportive measures and close monitoring of vital signs. Gabapentin can be removed by hemodialysis, which may be indicated in severe cases or in patients with significant renal impairment.
Massive FAQ Section
Q1: What is the difference between Gabapentin and Gabapentin Enacarbil (Horizant)?
A1: Gabapentin Enacarbil (Horizant) is a prodrug of gabapentin, meaning it is converted into active gabapentin in the body. The key difference lies in its unique absorption mechanism (L-amino acid transporter 1) and extended-release formulation. This allows Horizant to deliver gabapentin more efficiently and maintain stable drug levels for longer periods, often allowing for once or twice-daily dosing, compared to immediate-release gabapentin which typically requires multiple daily doses due to saturable absorption.
Q2: How should I take Horizant?
A2: Horizant tablets should be swallowed whole with food. Do not chew, crush, or split them, as this can alter the extended-release properties. For Restless Legs Syndrome, take 600 mg once daily with food at approximately 5:00 PM. For Postherpetic Neuralgia, you'll start with 600 mg once daily for 3 days, then increase to 600 mg twice daily (1200 mg/day) with food, with doses separated by 8 to 12 hours.
Q3: Can Horizant be crushed or chewed?
A3: No, Horizant tablets must not be crushed, chewed, or split. They are designed as an extended-release formulation, and altering the tablet can lead to a rapid release of the medication, potentially increasing side effects and reducing its effectiveness.
Q4: How long does it take for Horizant to start working?
A4: While some patients may notice improvement within a few days, the full therapeutic effects of Horizant for RLS or PHN may take several weeks to become apparent as your body adjusts to the medication. It's important to continue taking it as prescribed, even if you don't feel immediate relief.
Q5: What are the most common side effects of Horizant?
A5: Common side effects include dizziness, somnolence (sleepiness), nausea, fatigue, headache, dry mouth, constipation, and diarrhea. These are usually mild to moderate and may lessen over time. If they persist or are bothersome, consult your doctor.
Q6: Is Horizant addictive?
A6: Horizant is not considered an opioid and does not typically cause the same type of physical dependence associated with opioid medications. However, like other gabapentinoids, there have been reports of misuse and dependence, particularly in individuals with a history of substance abuse. Abruptly stopping Horizant can lead to withdrawal symptoms (e.g., anxiety, insomnia, nausea), so it should be tapered under medical supervision.
Q7: Can I drink alcohol while taking Horizant?
A7: It is generally advised to avoid or limit alcohol consumption while taking Horizant. Alcohol can enhance the central nervous system depressant effects of Horizant, leading to increased drowsiness, dizziness, and impaired coordination.
Q8: What if I miss a dose of Horizant?
A8: If you miss a dose, take your next dose as scheduled. Do not take an extra dose to make up for the missed one. It's important to maintain your prescribed dosing schedule.
Q9: Is Horizant safe during pregnancy or breastfeeding?
A9: Horizant is not extensively studied in pregnant women. Animal studies suggest potential risks to the fetus at high doses. It should only be used during pregnancy if the potential benefit outweighs the potential risk. Gabapentin is excreted into breast milk, and its effects on a breastfed infant are unknown. Discuss the risks and benefits with your doctor if you are pregnant, planning to become pregnant, or breastfeeding.
Q10: Does Horizant interact with other medications?
A10: Yes, Horizant can interact with other medications, particularly central nervous system depressants such as opioids, benzodiazepines, and certain antidepressants or antihistamines. These interactions can increase the risk of severe drowsiness and respiratory depression. Always inform your doctor and pharmacist about all medications, supplements, and herbal products you are taking.
Q11: What should I do in case of an overdose?
A11: In case of a suspected overdose, seek immediate medical attention or call emergency services. Symptoms of overdose may include severe dizziness, double vision, slurred speech, extreme drowsiness, lethargy, or diarrhea.
Q12: How does Horizant treat Restless Legs Syndrome?
A12: Horizant, through its active component gabapentin, binds to specific sites (alpha-2-delta subunits) on voltage-gated calcium channels in the brain. This action helps to modulate the release of certain neurotransmitters, which in turn can reduce the neuronal hyperexcitability and abnormal sensory signals that contribute to the uncomfortable sensations and irresistible urge to move characteristic of RLS.
Q13: How does Horizant treat Postherpetic Neuralgia?
A13: For PHN, Horizant works by the same mechanism of binding to the alpha-2-delta subunit of voltage-gated calcium channels. In PHN, damaged nerves become hypersensitive and generate abnormal pain signals. By modulating calcium influx and neurotransmitter release, Horizant helps to calm these overactive nerves, thereby reducing the severity and frequency of the neuropathic pain associated with PHN.