Comprehensive Bone Biology: Cellular Mechanisms, Remodeling, & Orthopedic Pathologies
Key Takeaway
Bone biology centers on osteoblasts, bone-forming cells originating from mesenchymal stem cells. They synthesize bone matrix under regulation by RUNX2, BMPs, and Wnt pathways. This cellular orchestration drives bone remodeling, vital for skeletal integrity, mechanical adaptation, and effective orthopedic fracture healing.
Introduction and Epidemiology
Bone, a dynamic and highly specialized connective tissue, serves critical mechanical and metabolic functions within the human body. As orthopedic surgeons, a profound understanding of bone biology, from its cellular constituents to its adaptive responses, is paramount to optimizing patient outcomes. The remarkable capacity of bone for self-repair and remodeling differentiates it from many other tissues, yet this capacity is tightly regulated by intricate cellular and molecular mechanisms. Disruptions to these processes underpin a vast array of orthopedic pathologies, including fracture nonunion, osteoporosis, osteonecrosis, and tumor formation.
The fundamental unit of bone remodeling is the basic multicellular unit (BMU), a coordinated consortium of bone-resorbing osteoclasts and bone-forming osteoblasts. The equilibrium between bone resorption and formation dictates overall bone mass and structural integrity. A comprehensive grasp of this cellular interplay is essential for the effective management of both acute trauma and chronic degenerative conditions. Furthermore, age-related decline in bone repair capabilities, influenced by changes in stem cell niches and hormonal signaling, contributes to the epidemiological burden of fragility fractures in the elderly.
Understanding the precise cellular and molecular pathways that govern bone anabolism and catabolism allows for targeted therapeutic interventions, ranging from enhancing fracture healing to mitigating bone loss. Epidemiologically, impaired bone healing represents a massive burden on the healthcare system. Approximately 5 to 10 percent of the 6.3 million fractures occurring annually in the United States progress to delayed union or nonunion. These complications necessitate complex surgical interventions, prolonged rehabilitation, and substantial socioeconomic costs. Furthermore, osteoporosis affects over 200 million women worldwide, leading to millions of fragility fractures annually. The intersection of bone biology and surgical intervention is most evident in the management of these complex scenarios, where mechanical stability must be perfectly married with biological augmentation.

Surgical Anatomy and Biomechanics
Bone macrostructure comprises cortical (compact) bone, providing strength and rigidity, and cancellous (trabecular) bone, offering porosity and metabolic activity. At the microscopic level, these structures are organized into osteons in cortical bone and trabeculae in cancellous bone. The mechanical properties of bone, including its stiffness, strength, and toughness, are directly influenced by its hierarchical organization, composition, and cellular activity. The bone matrix, primarily composed of type I collagen and a diverse array of non-collagenous proteins, is mineralized by hydroxyapatite crystals, providing the material's compressive strength. The arrangement of collagen fibers and mineral crystals, influenced by mechanical loading and cellular secretion, dictates anisotropic mechanical behavior.
The cellular biology of bone dictates its adaptive response to mechanical stimuli, a principle fundamental to Wolff's Law. Mechanical stress and strain are transduced into biochemical signals, regulating the activity of osteocytes, osteoblasts, and osteoclasts.
Osteoblasts and Bone Formation
Osteoblasts are the primary bone-forming cells, responsible for synthesizing and secreting the organic components of the bone matrix, followed by its mineralization.
- Morphology and Location: They appear as cuboid cells aligned in layers along immature osteoid, typically found on bone surfaces undergoing active formation.
- Origin and Differentiation: Osteoblasts are derived from undifferentiated mesenchymal stem cells (MSCs). These MSCs reside in critical niches such as the haversian canals, endosteum, and periosteum. Their differentiation into osteoblasts is a tightly regulated process, critically influenced by the local mechanical and biochemical environment.
- Under conditions of low strain and increased oxygen tension, MSCs are directed towards the osteoblast lineage.
- The transcription factor RUNX2 (Runt-related transcription factor 2), also known as Core-binding factor alpha 1 (CBF alpha 1), is a master regulator of osteoblast differentiation, essential for skeletal development.
- Bone morphogenetic proteins (BMPs), particularly BMP-2, -4, and -7, are potent osteoinductive factors that signal through SMAD pathways to direct mesenchymal cells toward the osteoblast phenotype.
Osteoclasts and Bone Resorption
Osteoclasts are multinucleated giant cells responsible for bone resorption. They originate from the hematopoietic stem cell lineage, specifically the monocyte macrophage precursor pool.
The differentiation and activation of osteoclasts are governed by the RANKL OPG axis. Receptor Activator of Nuclear factor Kappa-B Ligand (RANKL), expressed by osteoblasts and osteocytes, binds to the RANK receptor on osteoclast precursors, stimulating their fusion, differentiation, and activation. Osteoprotegerin (OPG), also secreted by osteoblasts, acts as a decoy receptor for RANKL, competitively inhibiting its binding to RANK and thereby suppressing osteoclastogenesis.
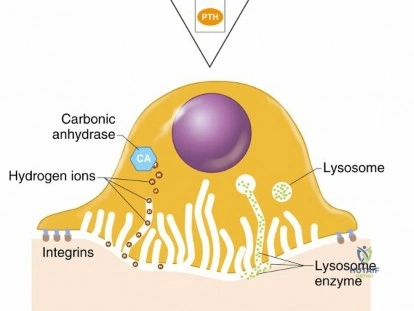
Active osteoclasts form a sealing zone over the bone matrix, creating an isolated microenvironment known as Howship's lacuna. Within this ruffled border, they secrete hydrogen ions via vacuolar-type H-ATPases to dissolve the inorganic hydroxyapatite, followed by the secretion of proteolytic enzymes, predominantly Cathepsin K and Matrix Metalloproteinases (MMPs), to degrade the organic collagenous matrix.

Osteocytes and Mechanotransduction
Osteocytes are terminally differentiated osteoblasts that have become embedded within the mineralized bone matrix. They represent the most abundant cell type in mature bone (comprising over 90 percent of bone cells) and reside within lacunae, communicating with one another and with surface cells via an extensive dendritic network housed within canaliculi.
Osteocytes function as the primary mechanosensors of the skeletal system. Fluid flow through the lacunocanalicular network, driven by mechanical loading, generates shear stress on osteocyte dendrites. This mechanical signal is transduced into biochemical responses, notably the regulation of sclerostin. Sclerostin, a glycoprotein encoded by the SOST gene, is secreted by osteocytes and acts as a potent inhibitor of bone formation by antagonizing the Wnt beta-catenin signaling pathway in osteoblasts. Mechanical loading downregulates sclerostin expression, thereby disinhibiting Wnt signaling and promoting osteogenesis. Conversely, unloading (e.g., microgravity, prolonged bed rest) upregulates sclerostin, leading to bone loss.
Biomechanics of Fracture Healing
Fracture healing involves a complex cascade of events categorized into primary (intramembranous) and secondary (endochondral) ossification. Primary healing requires absolute stability (interfragmentary strain less than 2 percent) and occurs via direct remodeling by cutting cones, bypassing the callus formation phase. This is typically achieved through rigid internal fixation, such as compression plating.
Secondary healing is the most common pathway and occurs under conditions of relative stability (interfragmentary strain between 2 and 10 percent), such as with intramedullary nailing, bridge plating, or external fixation. It proceeds through four overlapping phases: inflammatory response, soft callus formation (chondrogenesis), hard callus formation (woven bone), and remodeling (lamellar bone). The mechanical environment directly dictates the differentiation pathway of the pluripotential tissue within the fracture gap, following Perren's Strain Theory.

Indications and Contraindications
Surgical intervention in the context of bone biology typically involves the management of impaired healing (nonunions, delayed unions), critical-sized bone defects, and the necessity for biologic augmentation in high-risk arthrodesis. The decision to intervene operatively versus non-operatively hinges on the assessment of host biology, mechanical stability, and the presence of infection.
The "Diamond Concept" of fracture healing, proposed by Giannoudis, dictates that successful bone repair requires four mutually inclusive elements: osteogenic cells, an osteoconductive scaffold, osteoinductive growth factors, and mechanical stability. Deficiencies in any of these domains constitute an indication for targeted intervention.
| Clinical Scenario | Operative Indications (Biologic Augmentation and Fixation) | Non Operative Indications (Observation or Biophysical Modalities) |
|---|---|---|
| Delayed Union | Inadequate mechanical stability requiring hardware revision; clear biological failure (e.g., atrophic appearance at 4-6 months). | Stable fixation with progressive, albeit slow, callus formation; optimization of metabolic parameters (Vitamin D, smoking cessation). |
| Atrophic Nonunion | Absolute indication for operative intervention. Requires debridement, rigid fixation, and biologic augmentation (autograft, BMPs). | Contraindicated. Atrophic nonunions lack biological potential and will not heal without surgical delivery of osteoinductive/osteogenic elements. |
| Hypertrophic Nonunion | Indicated for optimization of mechanical stability. The biology is robust, but excessive strain prevents bridging. Requires rigid fixation (e.g., exchange nailing, compression plating). | Contraindicated. Biophysical stimulation (LIPUS) is rarely sufficient if the mechanical environment is grossly unstable. |
| Critical Sized Bone Defect | Segmental defects greater than 2-3 cm. Requires Masquelet technique, bone transport (Ilizarov), or vascularized bone grafting. | Contraindicated. Defects of this magnitude exceed the inherent biological capacity for spontaneous bridging. |
| High Risk Arthrodesis | Patient with multiple risk factors (smoker, diabetic, revision surgery). Indicated for use of autograft, cellular allograft, or BMP-2. | Standard primary arthrodesis in a healthy host may proceed with local autograft/allograft without advanced biologic adjuncts. |
Contraindications to advanced biologic augmentation (e.g., recombinant BMPs) include active infection at the surgical site, history of malignancy in the vicinity of the defect, skeletal immaturity (open physes), and known hypersensitivity to the product. Furthermore, the use of BMP-2 in the anterior cervical spine is generally avoided or used with extreme caution due to the risk of life-threatening prevertebral soft tissue swelling.

Pre Operative Planning and Patient Positioning
Preoperative planning for complex bone reconstruction or nonunion surgery begins with a meticulous assessment of the host's physiological status. The surgeon must identify and optimize any systemic factors that impair bone biology.
Metabolic Evaluation and Optimization
A comprehensive metabolic workup is essential for patients presenting with nonunions or requiring complex fusions. This includes evaluating serum calcium, phosphorus, alkaline phosphatase, parathyroid hormone (PTH), and 25-hydroxyvitamin D levels. Vitamin D deficiency is endemic and profoundly impairs osteoid mineralization; levels should be repleted to greater than 30 ng/mL prior to elective reconstruction.
Endocrine abnormalities, particularly uncontrolled diabetes mellitus and thyroid dysfunction, must be managed. Glycemic control is critical, as advanced glycation end-products (AGEs) disrupt collagen cross-linking and impair osteoblast function. Smoking cessation is paramount; nicotine causes peripheral vasoconstriction, leading to tissue hypoxia, while carbon monoxide competitively inhibits oxygen binding to hemoglobin. Furthermore, toxins in cigarette smoke directly inhibit osteoblastogenesis. Patients should be counseled that active smoking significantly increases the risk of graft failure and recurrent nonunion.
Preoperative Imaging
High-resolution imaging is required to define the structural anatomy of the defect or nonunion.
* Orthogonal Radiographs: Assess overall alignment, hardware integrity, and the morphology of the nonunion (oligotrophic, atrophic, or hypertrophic).
* Computed Tomography: CT with multiplanar reconstruction is the gold standard for assessing the extent of bridging bone, evaluating the size of segmental defects, and planning hardware trajectory.
* Magnetic Resonance Imaging: MRI is utilized to evaluate the viability of the bone ends (e.g., assessing for avascular necrosis) and to delineate associated soft tissue injuries or infection.
* Nuclear Medicine: Indium-111 labeled white blood cell scans combined with Technetium-99m bone scans may be employed to differentiate aseptic nonunion from chronic osteomyelitis.
Patient Positioning
Positioning is dictated by the primary surgical site and the planned autograft harvest locations.
* Anterior Iliac Crest Bone Graft Harvest: The patient is positioned supine. A bump may be placed under the ipsilateral hip to elevate the crest.
* Posterior Iliac Crest Bone Graft Harvest: The patient is positioned prone. This provides the largest volume of autogenous cancellous bone.
* Reamer Irrigator Aspirator Harvest: For RIA harvest from the femur, the patient is positioned supine on a radiolucent flat Jackson table or a fracture table, depending on concurrent procedures. The ipsilateral limb is prepped and draped free to allow for access to the greater trochanter or retrograde entry point.

Detailed Surgical Approach and Technique
The surgical technique for addressing impaired bone biology—specifically in the context of an atrophic nonunion—requires a systematic approach to restore both the mechanical and biological environments. This process embodies the clinical application of the Diamond Concept.
Site Preparation and Debridement
The initial and arguably most critical step is the meticulous takedown of the nonunion site.
1. Exposure: The nonunion is approached through an appropriate internervous plane, minimizing further disruption to the periosteal blood supply.
2. Hardware Removal: Any failed or unstable hardware is removed. Deep tissue cultures are obtained routinely, even in presumed aseptic nonunions, to rule out indolent infections (e.g., Cutibacterium acnes, coagulase-negative staphylococci).
3. Debridement: All intervening fibrous tissue, synovial pseudarthrosis fluid, and necrotic bone must be radically excised.
4. Decortication and Petaling: The sclerotic bone ends are resected back to healthy, bleeding cortical bone. This is confirmed by the "paprika sign"—the presence of punctate cortical bleeding. The medullary canal on both sides of the nonunion must be opened and reamed to restore endosteal blood flow and allow for marrow continuity.
Autologous Bone Graft Harvest Techniques
Autologous bone graft remains the gold standard for biologic augmentation, possessing osteoconductive, osteoinductive, and osteogenic properties.
Iliac Crest Bone Graft Harvest:
1. Incision: For the anterior crest, the incision is made 2 cm posterior to the Anterior Superior Iliac Spine (ASIS) to avoid the Lateral Femoral Cutaneous Nerve (LFCN).
2. Dissection: The fascia is incised over the crest. The gluteal musculature is elevated subperiosteally from the outer table, or the iliacus is elevated from the inner table.
3. Harvest: Corticocancellous blocks can be harvested using osteotomes, or pure cancellous bone can be obtained using gouges and curettes between the inner and outer tables.
4. Closure: Meticulous hemostasis is achieved using bone wax or topical hemostatic agents. The fascial layers are closed tightly over a drain to prevent hematoma formation.
Reamer Irrigator Aspirator Technique:
The RIA system allows for the harvest of a large volume of highly osteogenic intramedullary graft while minimizing donor site morbidity compared to ICBG.
1. Entry: A standard antegrade or retrograde femoral entry point is established under fluoroscopy.
2. Guidewire Placement: A ball-tipped guidewire is passed into the metaphysis.
3. Reaming: The RIA reamer is advanced over the guidewire. The system simultaneously irrigates the canal and aspirates the reamings, collecting the bone graft and marrow elements in a filter.
4. Graft Processing: The harvested effluent is rich in mesenchymal stem cells, osteoprogenitor cells, and growth factors. It is typically mixed with an osteoconductive carrier (e.g., cancellous chips or synthetic matrix) prior to implantation.

Application of Biologics and Scaffolds
Once the site is prepared and the graft is harvested, the biological construct is assembled.
* Osteoinduction: If autograft volume is insufficient or the host biology is severely compromised, recombinant human Bone Morphogenetic Protein 2 (rhBMP-2) may be utilized. The BMP is reconstituted and applied to an absorbable collagen sponge carrier.
* Osteoconduction: Demineralized bone matrix (DBM), tricalcium phosphate, or hydroxyapatite scaffolds may be used as volume expanders. These provide a structural lattice for osteoblast migration and new bone deposition.
* Implantation: The graft material is packed meticulously into the defect, ensuring intimate contact with the bleeding host bone ends.
Fixation Principles
Biological augmentation is futile without absolute mechanical stability in an atrophic nonunion.
1. Compression: If the defect is small or the bone ends can be apposed, compression plating is utilized to minimize interfragmentary strain and promote primary bone healing.
2. Bridging: For segmental defects, rigid bridge plating or statically locked intramedullary nailing is employed. The construct must be robust enough to withstand physiological loads during the prolonged incorporation phase of the bone graft.

Complications and Management
The surgical manipulation of bone biology and the use of orthobiologics carry specific risks that must be anticipated and managed. Complications can arise from the graft harvest site, the application of recombinant proteins, or the failure of the biological construct.
| Complication | Incidence | Etiology and Clinical Presentation | Salvage Strategy and Management |
|---|---|---|---|
| ICBG Donor Site Morbidity | 10 - 30% | Chronic pain, hematoma, infection, or pelvic fracture. LFCN injury causes anterolateral thigh numbness (meralgia paresthetica). | Prevention via meticulous technique (staying 2cm posterior to ASIS). Management includes gabapentinoids for nerve pain, drainage of large hematomas. |
| RIA Femur Fracture | 1 - 2% | Eccentric reaming or over-reaming of the femoral diaphysis leading to iatrogenic fracture during or after harvest. | Prophylactic intramedullary nailing if cortical thinning is severe. Immediate internal fixation if fracture occurs intraoperatively. |
| BMP Induced Swelling | 5 - 15% | Robust inflammatory response secondary to BMP application. In the cervical spine, this can cause life-threatening airway compromise. | Avoid BMP in anterior cervical spine. In extremities, manage with elevation, delayed closure, or short-course corticosteroids if severe. |
| Heterotopic Ossification | 5 - 10% | Ectopic bone formation due to BMP leakage into surrounding soft tissues or robust local stem cell activation. | Meticulous containment of BMP/graft within the bony defect. Excision of HO after maturation if it causes mechanical block to joint motion. |
| Persistent Nonunion | 5 - 15% | Failure of the biological construct to incorporate due to inadequate stability, infection, or poor host metabolic status. | Comprehensive re-evaluation (Diamond Concept). Rule out infection (ESR, CRP, biopsy). Revise fixation and repeat biologic augmentation. |

Post Operative Rehabilitation Protocols
Postoperative rehabilitation following biologic reconstruction is a delicate balance between protecting the surgical construct and applying controlled mechanical stress to stimulate bone healing via mechanotransduction pathways.
The initial phase (0 to 6 weeks) typically focuses on protecting the fixation and allowing the acute inflammatory and soft callus phases of healing to commence. Weight-bearing is generally restricted, particularly if large segmental defects were grafted or if the fixation relies entirely on bridging constructs. Joint mobilization of adjacent joints is initiated early to prevent arthrofibrosis and to facilitate local blood flow.
The intermediate phase (6 to 12 weeks) incorporates the principles of Wolff's Law. As radiographic evidence of early graft incorporation or callus formation appears, progressive mechanical loading is introduced. Mechanical strain across the healing site is critical for downregulating osteocytic sclerostin and upregulating Wnt/beta-catenin signaling, thereby driving osteoblast-mediated bone formation. Patients are transitioned from touch-down weight-bearing to partial and eventually full weight-bearing based on radiographic progression and clinical symptoms.
Metabolic support continues postoperatively. Patients must maintain adequate calcium (1000-1200 mg daily) and Vitamin D (1000-2000 IU daily) intake. In specific refractory cases, such as atypical femoral nonunions or severe osteoporotic fractures, the off-label use of anabolic agents like Teriparatide (recombinant human PTH 1-34) may be considered to stimulate osteoblast activity and accelerate remodeling, though this requires careful patient selection and endocrinology consultation.
Summary of Key Literature and Guidelines
The evolution of our understanding of bone biology and its surgical application is rooted in several landmark studies and conceptual frameworks.
- The Diamond Concept (Giannoudis et al., 2007): This conceptual framework revolutionized the approach to fracture nonunions by emphasizing the equal importance of osteogenic cells, osteoconductive scaffolds, osteoinductive factors, and mechanical stability. It remains the guiding principle for complex bone reconstruction.
- Discovery of BMPs (Urist, 1965): Marshall Urist's seminal discovery that demineralized bone matrix could induce ectopic bone formation laid the foundation for the isolation and eventual recombinant production of Bone Morphogenetic Proteins, fundamentally altering the landscape of biologic augmentation.
- The BESTT Study (Govender et al., 2002): This prospective, randomized multicenter trial demonstrated that the application of rhBMP-2 on an absorbable collagen sponge significantly reduced the frequency of secondary interventions and accelerated fracture and wound healing in open tibial shaft fractures compared to standard intramedullary nailing alone.
- Mechanotransduction and Sclerostin (Robling et al., 2008): Research elucidating the role of the osteocyte network in sensing mechanical load and regulating bone mass via sclerostin inhibition has provided the molecular basis for Wolff's Law and paved the way for novel therapeutics like Romosozumab (anti-sclerostin antibody) in the treatment of severe osteoporosis.
A thorough comprehension of these biological principles, coupled with meticulous surgical technique, allows the orthopedic surgeon to predictably restore skeletal integrity even in the most challenging clinical scenarios.
Clinical & Radiographic Imaging








