Revascularization and Free Tissue Transfer in Orthopaedic Surgery
Key Takeaway
Revascularization and microvascular free tissue transfer are critical techniques in orthopaedic limb salvage. This comprehensive guide details the indications, biomechanics, and step-by-step surgical protocols for managing devitalized tissues and complex soft-tissue defects. From the "pocket technique" for distal fingertip amputations to the selection of axial pattern free flaps, mastering these microsurgical principles is essential for restoring both form and function in severely traumatized extremities.
REVASCULARIZATION IN ORTHOPAEDIC TRAUMA
Partial amputation or severe devitalization of tissues resulting from serious vascular interruption frequently occurs without the complete detachment of the anatomical part. While some of these tissues with impaired circulation may ultimately survive through collateral flow, persistent ischemia is a critical concern. Chronic ischemia can lead to disabling cold intolerance, severe atrophy, and debilitating contractures of the intrinsic muscles of the hand or foot.
Clinically, digits with impaired circulation demonstrate an extremely slow return of normal pink color following blanching by pressure, indicating profound capillary stasis and endothelial dysfunction. The overarching management of these complex hand and extremity injuries is essentially identical to that of complete replantation. However, because venous and lymphatic channels may be partially intact, a longer interval from the time of the accident to the microvascular anastomosis of the vessels is generally tolerated. Furthermore, unlike multi-level replantations, revascularization procedures can often be efficiently executed by a single surgical team.
When major vessels, such as the radial and ulnar arteries, are severed at the wrist, the standard of care dictates that at least one must be repaired to restore pulsatile flow to the palmar arches. If the viability of the hand remains questionable after single-vessel repair, or if the palmar arch is incomplete, both the radial and the ulnar arteries must be meticulously repaired.

Special Techniques for Distal Amputations
For distal fingertip amputations where the caliber of the vessels renders standard microvascular anastomosis impossible, alternative salvage techniques must be employed to preserve length and function.
The Pocket Technique
Originally described by Brent, the "pocket technique" is utilized for composite graft survival. This method involves meticulously debriding and de-epithelializing the amputated part, reattaching it as a composite graft, and burying it in a well-vascularized subcutaneous pocket. Brent initially utilized a contralateral chest-wall pocket for 3 weeks, after which the digit was removed and the viable tip skin was grafted. Lee et al. subsequently adapted this technique using an abdominal pocket.
However, Muneuchi et al. reported poor functional and aesthetic results with this technique in seven fingers, strongly advising against its use for injuries at or proximal to the lunula. To mitigate the severe shoulder and elbow stiffness associated with prolonged chest or abdominal attachment, Arata et al. modified the procedure by utilizing the ipsilateral palm as the pocket site. In their series of 16 patients, complete survival was achieved in 13, with the remaining three exhibiting only partial necrosis.
🔪 Clinical Pearl: When utilizing the palmar pocket technique, ensure the de-epithelialization is strictly limited to the mid-dermal layer to preserve the subdermal plexus, which is critical for the initial plasmatic imbibition and subsequent inosculation of the composite graft.
Arteriovenous Anastomosis
Another highly specialized technique for replanting distal fingertip amputations involves creating a microvascular arteriovenous (AV) anastomosis. This is achieved by anastomosing a volar radial vein to the proximal digital artery. Venous drainage is subsequently accomplished by creating a transverse tip incision to allow controlled egress of blood, preventing venous congestion. Yabe et al. reported successful survival in all three cases utilizing this technique, though one developed partial necrosis.
SURGICAL TECHNIQUE: THE PALMAR POCKET PROCEDURE
The following is a detailed, step-by-step protocol for the modified palmar pocket technique for distal digit salvage.
First Operation: Preparation and Implantation
- Anesthesia and Positioning: Utilize a regional wrist block or upper arm block anesthesia. Apply a well-padded pneumatic tourniquet to the upper arm.
- Preparation: Wash the amputated part and the amputation stump copiously with normal saline. Carefully extract the nail plate to prevent subungual hematoma and infection.
- Skeletal Stabilization: Reduce any fractured bone segments anatomically. Stabilize the skeleton using fine Kirschner wires (0.028 or 0.035 inch), cutting the wires as short as possible to prevent soft-tissue tethering.
- Reattachment: Reattach the amputated part to the amputation stump without attempting vascular anastomosis.
- De-epithelialization: Using a #15 scalpel blade, meticulously de-epithelialize the amputated part down to the mid-dermal layer. Do not expose the subcutaneous fat.
- Pocket Creation: Make a 2-cm transverse incision in the ipsilateral palm. Bluntly undermine the subcutaneous layer using tenotomy scissors to form a vascularized pocket.
- Insertion and Fixation: Insert the reattached, de-epithelialized part into the palmar pocket. Suture the finger to the palmar skin exactly 2 mm proximal to the reattached level. This precise suturing prevents the inserted digit from inadvertently pulling out of the pocket during the healing phase.
- Dressing: Apply a light, non-adherent compressive gauze dressing. Avoid rigid splinting to allow micro-motion that stimulates vascular ingrowth.
Second Operation: Division and Rehabilitation
- Timing: The second stage is performed 16 to 20 days after the initial operation.
- Extraction: Carefully dissect and remove the replanted part from the palmar pocket.
- Closure: Suture the palmar skin defect primarily.
- Postoperative Care: Change the digital dressing to a wet-to-dry or specialized moist wound healing dressing. Immediately encourage active range of motion (ROM) exercises of the injured finger to prevent tendon adhesions.
- Maturation: At approximately 2 weeks following the second operation, spontaneous epithelialization over the dermis should be complete, and the replanted part will gradually gain structural stability and sensory return.
SINGLE-STAGE TISSUE TRANSFER (FREE FLAPS)
Before the advent of reliable microvascular techniques, remote pedicle flaps (e.g., tubed groin or abdominal flaps) were the gold standard for covering major soft-tissue defects. In 1946, Shaw and Payne detailed their extensive experience with tubed pedicle flaps based on the superficial epigastric and superficial circumflex arterial circulations. Building upon this, and analyzing Bakamjian’s deltopectoral flap, McGregor and Morgan elucidated the critical biomechanical and physiological differences between random pattern and axial pattern flaps.
Flap Biomechanics and Vascular Anatomy
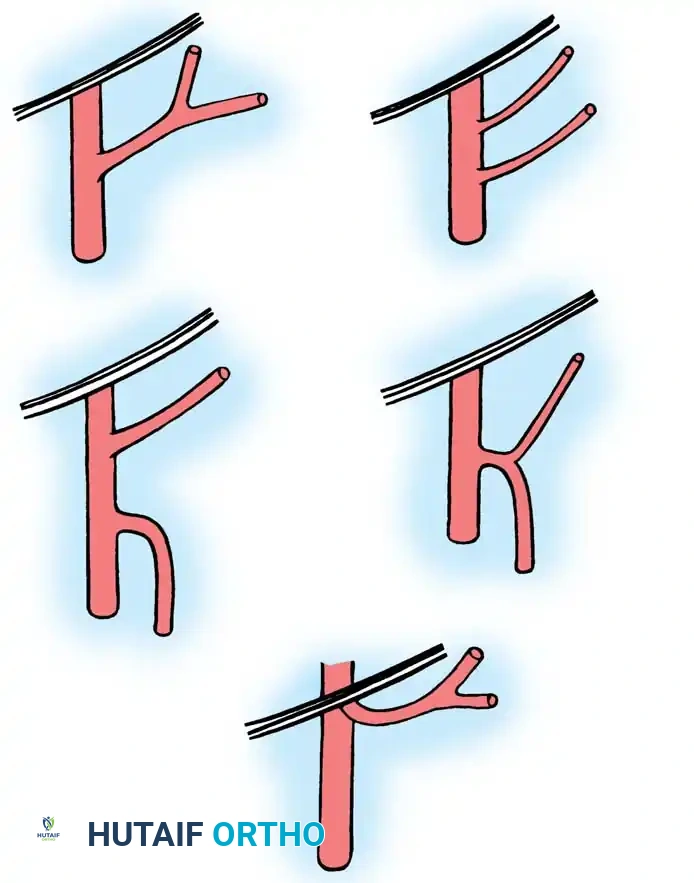
Random Pattern Flaps:
A random pattern flap relies on no specific, anatomically named pattern of circulation. Instead, it depends entirely on the subdermal plexus. Biomechanically, a length-to-width ratio of greater than 2:1 exponentially increases the risk of distal tip necrosis and overall flap failure due to inadequate perfusion pressure.
Axial Pattern Flaps:
An axial pattern flap relies on a definite, anatomically consistent arterial supply centered on one or more named arteries running longitudinally within the flap. Because of this direct arterial inflow, there are no rigid length-to-width ratio requirements, allowing for the design of long, versatile flaps.
These axial flaps are generally classified as cutaneous or myocutaneous, depending on the specific architecture of their arterial circulation:
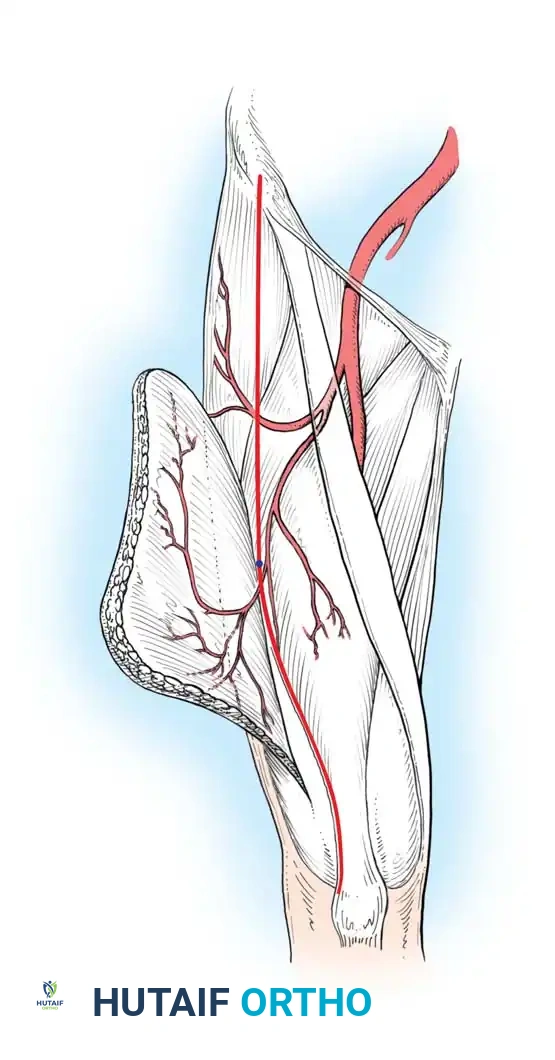
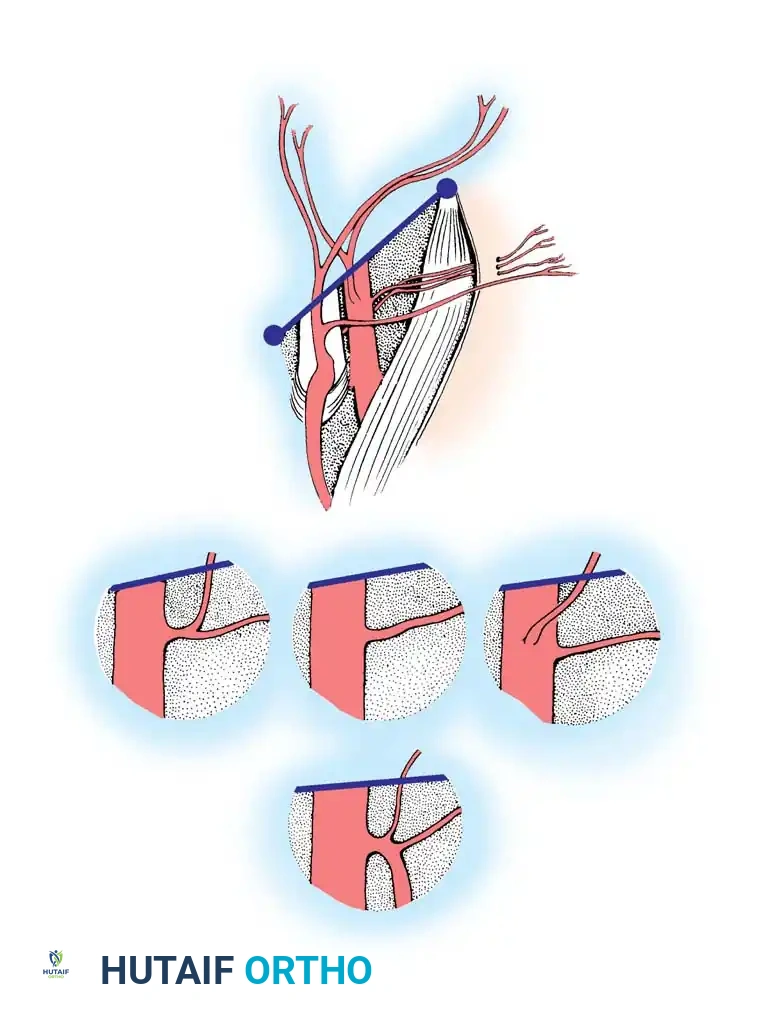
* Cutaneous Flaps: Rely on a constant circulation from a single artery passing through the underlying subcutaneous tissue, supplying the overlying skin directly through the dermal-subdermal vessels.
* Myocutaneous Flaps: Receive their cutaneous arterial supply from deep vessels that perforate the underlying muscle and fascia to reach the skin.
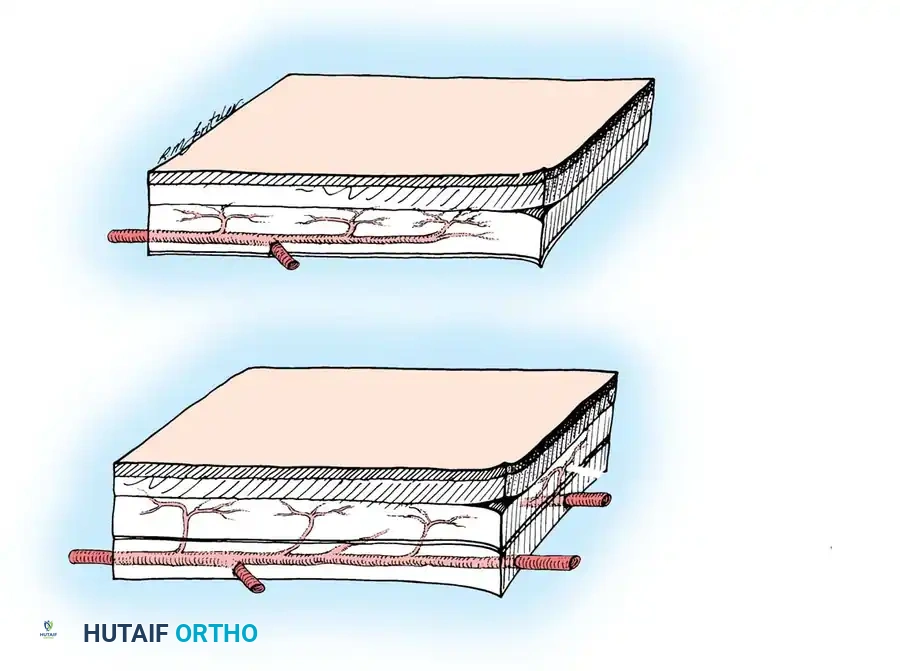
Arterial supply to axial pattern flaps. A, Cutaneous flap, relying on a single artery supplying dermal and subdermal vessels. B, Myocutaneous flap, relying on deep musculocutaneous arteries perforating muscle and fascia to the overlying skin.
Historical Evolution of Microvascular Transfer
Following experimental successes by Goldwyn, Lamb, and White, and the demonstration of successful microvascular free tissue transfer in dogs by Krizek et al., McLean and Buncke reported the first successful clinical free flap in 1972—the coverage of a scalp defect with freely vascularized omentum. By 1973, Daniel and Taylor, alongside O’Brien et al., reported their groundbreaking experiences with the single-stage transfer of groin flaps to the lower extremities. Today, free tissue transfer is a cornerstone of orthopaedic reconstructive surgery.
INDICATIONS AND ADVANTAGES OF FREE FLAPS
While the traditional indications for pedicle flaps share similarities with free flaps, the unparalleled versatility of microvascular transfer vastly expands the reconstructive armamentarium. Each patient must be evaluated individually, but the use of a free flap generally implies that a traditional random or local axial flap is either impossible or biomechanically inappropriate.
Current Indications
- Coverage of Essential Structures: Protection of exposed blood vessels, nerves, tendons, bone devoid of periosteum, and open joints.
- Unsatisfactory Soft-Tissue Beds: Coverage of areas unsuitable for later reconstructive procedures, such as dense scar tissue, chronic draining ulcers, and chronic osteomyelitis. A healthy flap is required before tendon grafting, nerve repair, or bone stabilization can occur.
- Unstable Scars: Replacement of fragile, unstable scars resulting from severe burns, high-dose irradiation, radical oncologic resections, and severe scar contractures.
- Lack of Local Options: Situations where the zone of injury precludes the use of a suitable random or local axial pattern flap.
- Avoidance of Immobilization: Cases where prolonged immobilization of the extremities in awkward positions (e.g., cross-leg flaps) is medically undesirable or physically impossible.
- Functional Restoration: Restoration of specific tissues to satisfy a functional demand. This includes restoring sensibility to the hand or plantar foot, digital reconstruction (toe-to-hand), replacing major skeletal muscle loss, bridging massive bone defects, and correcting congenital deformities (e.g., radial clubhand, congenital pseudarthrosis of the tibia).
Distinct Advantages
Microvascular free flaps offer profound advantages over traditional staged pedicle techniques:
* Single-Stage Procedure: Eliminates the need for multiple surgeries and prolonged attachment to a distant donor site.
* Aesthetic and Functional Matching: Greater versatility in matching the color, texture, thickness, and hair distribution of the donor tissue to the recipient bed.
* Donor Site Morbidity: In many instances, the donor site can be closed primarily without the need for split-thickness skin grafts, leaving an acceptable cosmetic appearance.
* Permanent Vascularity: Well-vascularized tissue with a permanent, robust blood supply replaces ischemic or avascular beds, fundamentally altering the local wound environment.
* Composite Reconstructions: Vascularized bone grafts, functioning joints, physes, and skeletal muscle can be electively included in a single composite graft.
* Early Mobilization: Joints adjacent to the recipient area can be mobilized much earlier, drastically reducing the incidence of joint stiffness and contractures.
* Reduced Hospitalization: Overall hospital stays and rehabilitation timelines are significantly shortened.
CONTRAINDICATIONS AND DISADVANTAGES
Relative and Absolute Contraindications
While absolute contraindications are rare, the reconstructive surgeon must exercise extreme caution in the following scenarios:
* Systemic Illness: Severe cardiovascular, pulmonary, or metabolic diseases that preclude prolonged anesthesia.
* Vascular Anatomy: The presence of only a single major artery supplying the distal limb (e.g., a single-vessel runoff leg). Sacrificing this vessel for an end-to-end anastomosis could result in distal limb ischemia.
* Zone of Injury: Previous severe trauma or high-dose irradiation to the donor or recipient vessels, which causes profound intimal damage and perivascular fibrosis, preventing the performance of a satisfactory, patent anastomosis.
🚨 Surgical Warning: Never perform a microvascular anastomosis within the "zone of injury." Vessels must be traced proximally and distally until healthy, unscarred intima is visualized under the operating microscope. Failure to do so guarantees thrombosis.
Disadvantages and Surgical Risks
- Operative Time: The initial operation is significantly longer than conventional flap procedures, typically ranging from 4 to 10 hours depending on flap complexity and team experience.
- Technical Difficulty: The procedures are technically demanding, tedious, and require specialized microsurgical training.
- Resource Intensive: Two highly trained surgical teams are usually required to work simultaneously (donor and recipient sites) to minimize ischemia time.
- Thrombosis Risk: If vascular thrombosis occurs (arterial or venous), the risk of complete, irreversible loss of the free flap is exceptionally high.
- Failure Rates: Historically, a 10% to 30% failure rate for free flaps was cited by Sharzer et al., though modern high-volume centers report success rates exceeding 95%. Nonetheless, the reoperation rate for hematoma, congestion, or arterial spasm can approach 25%.
- Delayed Complications: While postoperative vascular complications usually occur within the first 24 to 48 hours, delayed thrombosis can be seen up to 10 days post-procedure.
SELECTION OF FREE FLAPS BY ANATOMIC REGION
The selection of a specific free flap is dictated by the defect's size, depth, functional requirements, and the availability of recipient vessels.
Upper Extremity Reconstruction
In the upper extremity, free tissue transfer is invaluable for soft-tissue coverage, sensibility restoration, bony reconstruction, and the replacement of nonfunctioning musculotendinous units.
- Soft-Tissue Coverage: The groin cutaneous flap and the dorsalis pedis cutaneous flap are frequently utilized. The dorsalis pedis flap offers the distinct advantage of being a neurovascular flap; its nerve supply (deep and superficial peroneal nerves) can be coapted to recipient nerves to restore critical sensibility to the hand.
- Dead Space Management: For massive defects with considerable dead space, particularly around the elbow, free muscle transfers such as the latissimus dorsi, serratus anterior, and rectus abdominis are highly effective.
- Functional Muscle Transfer: As popularized by Manktelow, the gracilis, latissimus dorsi, and pectoralis major muscles are transferred with their motor nerves to restore active skeletal muscle function to the forearm (e.g., following Volkmann's ischemic contracture).
- Digital Reconstruction: Portions or all of the great, second, and third toes are routinely used for thumb and finger reconstruction.
- Bone Reconstruction: Vascularized bone grafts utilizing the rib, iliac crest, or fibula are employed for segmental bone loss.
Lister and Scheker have strongly advocated for immediate free flap coverage in the upper extremity. In their landmark series of 31 emergency free flaps, 93.5% survived with only one infection. Coverage was obtained immediately after initial radical debridement, within 24 hours of injury. This aggressive approach requires a well-staffed microvascular team and absolute certainty in the adequacy of the debridement.
Lower Extremity Reconstruction and Osteomyelitis
The lower extremity presents unique challenges, particularly regarding weight-bearing forces, poor local vascularity, and the high incidence of post-traumatic chronic osteomyelitis.
- Soft-Tissue Coverage: Requirements are frequently satisfied using the latissimus dorsi, serratus anterior, rectus abdominis, gracilis, tensor fasciae latae (TFL), free groin, and scapular cutaneous flaps.
- Plantar Sensibility: The dorsalis pedis neurovascular flap is utilized to provide durable, sensate coverage to the weight-bearing plantar surface of the foot.
- Bone Reconstruction: While the rib and iliac crest have been used, their natural curvature and relative biomechanical weakness limit their usefulness in the lower limb. The vascularized fibula is the gold standard for lower extremity bony defects caused by tumor resection, massive trauma, and congenital anomalies like congenital pseudarthrosis of the tibia. It has also been applied to osteonecrosis of the femoral head, though long-term consensus remains under investigation.
Management of Chronic Osteomyelitis
The application of free flaps has revolutionized the treatment of post-traumatic chronic osteomyelitis. Experimental work by Mathes et al. demonstrated that myocutaneous flaps are significantly more resistant to bacterial inoculation than random pattern flaps due to their robust, high-flow vascularity which delivers oxygen, leukocytes, and systemic antibiotics directly to the infected bed.
Clinical results from May et al. and Mathes et al. utilizing microvascular muscle flaps for osteomyelitis have been highly optimistic. May reported zero flap failures in 35 patients, with only a single patient experiencing recurrent drainage. They emphasize a rigid, systematic approach:
1. Precise identification of wound flora via deep tissue cultures.
2. Thorough, radical oncologic-style debridement of all necrotic bone and soft tissue.
3. Application of well-vascularized soft-tissue coverage appropriate to
You Might Also Like