Comprehensive Introduction and Patho-Epidemiology
Posterior cervical decompression and fusion (PCDF) remains a cornerstone in the armamentarium of the modern spine surgeon. This highly versatile approach provides robust access to the dorsal elements of the cervical spine, allowing for expansive neural decompression, deformity correction, and rigid segmental stabilization. Unlike anterior approaches, which are limited by visceral anatomy and multi-level biomechanical constraints, the posterior approach affords the surgeon the ability to address extensive, multi-segmental pathology ranging from the occipitocervical junction down to the cervicothoracic transition zone. Mastery of this approach is not merely a technical prerequisite; it is a fundamental requirement for safely navigating the myriad complex pathologies that afflict the cervical neuroaxis.
The patho-epidemiology of conditions necessitating posterior cervical intervention is vast and increasingly prevalent in our aging population. Cervical spondylotic myelopathy (CSM) stands as the most common cause of spinal cord dysfunction in adults worldwide. The pathophysiology is driven by a cascade of degenerative changes, including intervertebral disc desiccation, facet joint hypertrophy, ligamentum flavum buckling, and osteophyte formation, which collectively result in circumferential stenosis and ischemic injury to the spinal cord. Furthermore, conditions such as ossification of the posterior longitudinal ligament (OPLL), which is particularly prevalent in East Asian demographics but increasingly recognized globally, often dictate a posterior approach due to the high risk of dural tears and catastrophic neurological injury associated with anterior resection of massive, dura-adherent ossified masses.
Beyond degenerative etiologies, the posterior cervical approach is indispensable in the management of high-energy traumatic injuries, neoplastic disease, and destructive infectious processes. Traumatic facet dislocations, burst fractures with severe dorsal element comminution, and highly unstable ligamentous injuries typically require the rigid posterior tension-band construct that pedicle or lateral mass instrumentation provides. In the realm of oncology, posterior stabilization is often required following palliative laminectomy or en bloc resection of metastatic lesions that compromise the structural integrity of the posterior columns. Understanding the diverse epidemiological landscape and the underlying mechanical pathophysiology of these conditions is critical for appropriate patient selection and surgical planning.
Detailed Surgical Anatomy and Biomechanics
Before we even consider making an incision, we must possess an encyclopedic, three-dimensional understanding of posterior cervical anatomy. This region represents a complex, highly dynamic interplay of bone, muscle, ligaments, and critical neurovascular structures. A millimeter of deviation in this territory can mean the difference between a successful reconstruction and a catastrophic, life-altering iatrogenic injury.
Osteology and Bony Landmarks
Our initial exposure relies heavily on identifying key bony landmarks, which serve as our topographic map during dissection. Superiorly, the occiput presents the external occipital protuberance, or inion, a readily palpable bony landmark in the midline. Extending laterally from this prominence is the superior nuchal line, while the inferior nuchal line runs parallel, roughly midway between the inion and the foramen magnum. Descending from the inion towards the foramen magnum is the median nuchal line. These lines serve as critical attachment points for our posterior cervical musculature and dictate our cranial exposure limits.
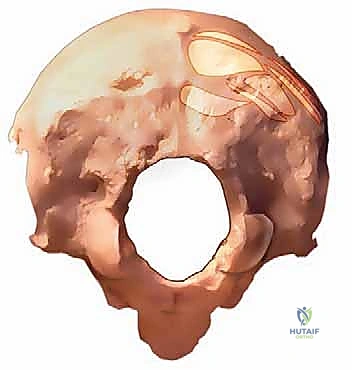
The cervical vertebrae themselves present unique morphological challenges. C1 (Atlas) uniquely lacks a spinous process, possessing instead a posterior tubercle that marks the center of its posterior arch—a crucial landmark for C1 lateral mass screw placement. C2 (Axis) features a distinctive, tall, bifid spinous process, representing the broadest in the cervical spine and serving as a reliable landmark for initial localization. The pars interarticularis of C2 connects the superior and inferior articular processes; its medial margin, along the superior border of the C2 lamina, serves as a vital guide to the medial margin of the C2 pedicle. C3 to C6 possess smaller, bifid spinous processes, while C7 typically boasts the longest, non-bifid spinous process, known as the vertebra prominens.
The lateral masses provide our primary fixation anchors in the subaxial spine. The C1 lateral mass averages 11.6 mm in width, generally accommodating a 3.5-mm screw safely below the posterior arch. In the subaxial spine (C3-C7), the anteroposterior depth of the lateral mass gradually reduces from approximately 8.9 mm at C3 to 6.4 mm at C7. The facet joints themselves dictate our approach to fusion and foraminotomy; while the C1-C2 joint is largely oriented in the axial plane to allow for rotation, the subaxial facet joints are coronally oriented at approximately 45 degrees to the axial plane.
Muscular Architecture and Dissection Planes
The muscles covering the posterior cervical spine are arranged in distinct layers, and understanding these is key to a systematic, relatively avascular dissection. The superficial layer is dominated by the Trapezius muscle, originating from the superior nuchal line, ligamentum nuchae, and upper thoracic spinous processes. During a standard midline approach, we incise through its avascular midline fascial attachment, minimizing muscle fiber transection.
Beneath this lies the intermediate layer, primarily the Splenius Capitis, which arises from the lower ligamentum nuchae and inserts onto the mastoid process. The deep layer is the most critical for our exposure, lying directly over the laminae and facet joints. From superficial to deep, we encounter the Semispinalis Capitis, Semispinalis Cervicis, Multifidus, and the deepest Rotatores. Maintaining a strict subperiosteal dissection plane deep to these muscles, directly on the bone, is imperative to minimize bleeding and preserve the muscular blood supply, which is critical for postoperative healing and the prevention of axial neck pain.
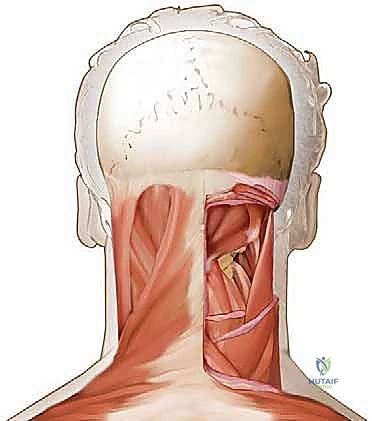
In the upper cervical spine, the suboccipital musculature becomes highly relevant. This includes the Rectus Capitis Posterior Minor and Major, and the Obliquus Capitis Superior and Inferior. The Obliquus Capitis Inferior forms the inferior border of the critical suboccipital triangle. This triangle, bounded by the rectus capitis posterior major and the two oblique muscles, houses the vertebral artery as it courses over the posterior arch of the atlas. Dissection in this region must be exceptionally meticulous to avoid catastrophic vascular injury.
Ligamentous Structures and Spinal Stability
The ligamentous structures of the posterior cervical spine provide dynamic stability and serve as important anatomical boundaries during dissection. The Ligamentum Nuchae is a strong, broad sheet of fibrous tissue extending from the external occipital protuberance to the spinous process of C7, essentially acting as a cranial extension of the supraspinous ligament. It is the primary structure incised during the initial midline approach.
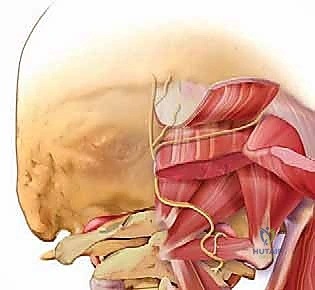
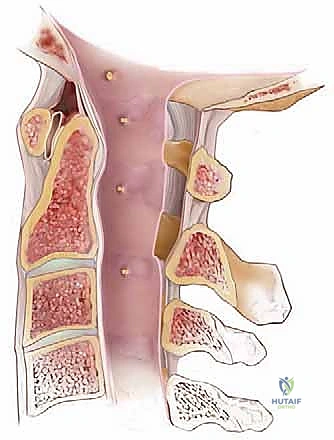
Deeper, we encounter the Posterior Atlanto-occipital Membrane, extending from the foramen magnum to the C1 posterior arch, and the Posterior Atlantoaxial Membrane, connecting C1 to C2. The ligamentum flavum, connecting adjacent laminae from C2 down to the sacrum, is a critical landmark during decompression. In conditions like CSM, the ligamentum flavum hypertrophies and buckles anteriorly into the spinal canal during cervical extension, contributing significantly to dynamic cord compression. Resection of this ligament is a key step in a thorough posterior laminectomy.
Neurovascular Considerations and the Vertebral Artery
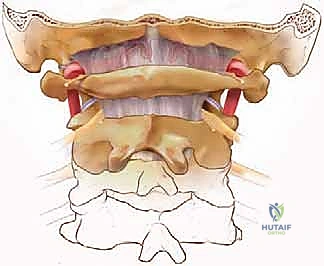
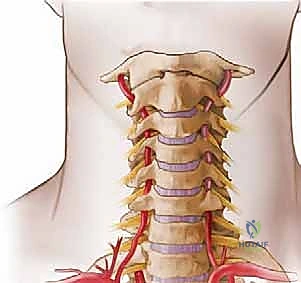
The vertebral artery (VA) is arguably the most critical vascular structure at risk during posterior cervical surgery. Originating from the subclavian artery, it typically enters the transverse foramen at C6 and ascends through the foramina to C1. After emerging from the C2 transverse foramen, it lies lateral to the C1-C2 facet joint before entering the C1 transverse foramen. It then exits C1 and courses posteromedially in a distinct groove on the superior surface of the posterior arch of the atlas (the V3 segment) before piercing the atlanto-occipital membrane to enter the foramen magnum.
To protect the VA during posterior C1 exposure, dissection must be strictly limited to within 12 mm of the midline on the posterior aspect of C1, and within 8 mm of the midline on the superior surface of the posterior arch. Further lateral dissection can be performed more safely on the inferior surface of the C1 arch. The VA is also at risk during lateral mass screw placement if the trajectory is too far anterior and medial, and during pedicle screw placement if the medial cortical wall of the pedicle is breached. Furthermore, the rich venous plexus surrounding the vertebral artery can cause profuse, difficult-to-control hemorrhage if violated during lateral dissection or foraminotomy.
Exhaustive Indications and Contraindications
The decision to proceed with a posterior cervical decompression and fusion must be based on a rigorous evaluation of the patient's clinical presentation, radiographic findings, and overall physiological status. The posterior approach is powerful, but it carries distinct morbidities that must be weighed against the expected benefits.
Primary Surgical Indications
Posterior cervical approaches are generally favored for multi-level pathology, particularly when the pathology spans three or more intervertebral disc levels. In cases of multi-level Cervical Spondylotic Myelopathy (CSM) or severe Ossification of the Posterior Longitudinal Ligament (OPLL), a posterior laminectomy or laminoplasty allows for indirect decompression of the spinal cord by allowing it to drift dorsally away from the anterior compressive elements. This "bowstringing" effect is highly dependent on the preservation of cervical lordosis.
| Pathology Category | Specific Indications for Posterior Approach |
|---|---|
| Degenerative | Multi-level CSM (>3 levels), OPLL, Congenital cervical stenosis, Revision surgery for adjacent segment disease, Foraminal stenosis requiring multi-level foraminotomies. |
| Trauma | Bilateral facet dislocations, Unstable burst fractures with posterior element disruption, Flexion-distraction injuries, Occipitocervical dissociation. |
| Neoplastic | Epidural spinal cord compression from metastatic disease, Primary osseous tumors of the posterior elements, Stabilization following palliative laminectomy. |
| Infectious | Epidural abscess requiring extensive dorsal evacuation, Osteomyelitis/discitis with resultant kyphotic instability requiring posterior tension-band fixation. |
| Deformity | Correction of fixed cervical kyphosis (often in conjunction with anterior releases), Ankylosing spondylitis fractures. |
Absolute and Relative Contraindications
While versatile, the posterior approach has distinct contraindications. A fixed cervical kyphosis is a major relative, and sometimes absolute, contraindication for a stand-alone posterior decompression (like laminectomy alone). If the spine is kyphotic, the spinal cord will remain draped over the anterior osteophytes or discs even after the posterior elements are removed, rendering the decompression ineffective and potentially exacerbating the deformity. In such cases, anterior reconstruction or a combined anterior-posterior approach is mandatory.
Absolute contraindications include active, untreated systemic infection (unless the surgery is specifically for source control of a spinal infection) and severe medical comorbidities that preclude prone positioning or general anesthesia. Relative contraindications include profound osteoporosis, which significantly compromises the pull-out strength of lateral mass and pedicle screws, increasing the risk of construct failure. Furthermore, previous extensive posterior cervical surgery with significant scarring and altered anatomy increases the risk of dural tears and vertebral artery injury, requiring meticulous preoperative planning.
Pre-Operative Planning, Templating, and Patient Positioning
Success in posterior cervical surgery is forged long before the scalpel touches the skin. Meticulous preoperative planning and precise patient positioning are non-negotiable elements of a safe and effective procedure.
Advanced Imaging and Pre-Operative Templating
Standard orthogonal radiographs are essential for assessing overall sagittal alignment, dynamic instability (via flexion-extension views), and bone quality. However, high-resolution Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the neural elements, identifying areas of cord signal change (myelomalacia), and delineating the extent of ligamentous hypertrophy.
For any planned instrumentation, particularly pedicle screws at C2, C7, or the subaxial spine, a fine-cut Computed Tomography (CT) scan is absolutely mandatory. The CT scan allows for precise measurement of pedicle width, trajectory angles, and lateral mass dimensions. Crucially, a CT angiogram (CTA) or MR angiogram (MRA) should be strongly considered if there is any suspicion of vertebral artery anomalies, such as an aberrant course, dominance of one artery, or extension into the C2 pars interarticularis. Knowing the exact location of the vertebral artery dictates our safe zones for drilling and screw placement.
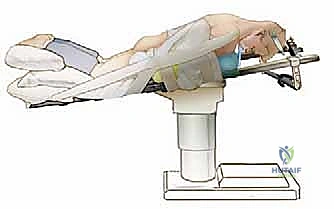
Patient Positioning and Intraoperative Setup
Patient positioning for posterior cervical surgery is a critical, high-risk maneuver. The patient is typically intubated on the transport stretcher and then carefully log-rolled onto the operating table into a prone position. The head is strictly immobilized, most commonly using Mayfield cranial tongs or a Gardner-Wells traction setup. The neck is positioned in a neutral to slightly lordotic alignment; hyperflexion must be avoided as it can exacerbate spinal cord compression, while hyperextension can compromise venous drainage and increase bleeding.
The operating table is often placed in a reverse Trendelenburg position (head elevated 15 to 30 degrees) to decrease venous pressure in the epidural and muscular venous plexuses, thereby significantly reducing intraoperative blood loss. All pressure points, particularly the eyes, face, breasts, and iliac crests, must be meticulously padded. Intraoperative neuromonitoring, including Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs), is considered standard of care to provide real-time feedback on spinal cord integrity during positioning, decompression, and instrumentation. Baseline signals must be obtained prior to positioning and compared immediately after the turn.
Step-by-Step Surgical Approach and Fixation Technique
The execution of a posterior cervical decompression and fusion requires a blend of anatomical respect, technical finesse, and spatial awareness. The procedure can be broadly divided into exposure, decompression, instrumentation, and arthrodesis.
Soft Tissue Exposure and Hemostasis
The skin incision is made strictly in the midline, guided by palpable landmarks (inion, C2 spinous process, C7 vertebra prominens) and fluoroscopy. The dissection proceeds through the subcutaneous tissue to the avascular ligamentum nuchae. Maintaining the dissection strictly in the midline raphe minimizes bleeding and muscle damage. Once the spinous processes are identified, a subperiosteal dissection is performed bilaterally using electrocautery and Cobb elevators.
The key to a bloodless exposure is staying deep to the muscular fascia and sweeping the muscles laterally off the laminae and lateral masses. Self-retaining retractors (e.g., Cerebellar or deep Gelpi retractors) are placed to maintain tension. The lateral exposure should extend to the lateral border of the lateral masses, taking care not to violate the facet joint capsules unless a fusion is planned at that specific level. Hemostasis is achieved meticulously using bipolar electrocautery, bone wax for osseous bleeding, and hemostatic agents (like thrombin-soaked gelatin sponges) for epidural venous oozing.
Decompression Techniques and Foraminotomy
If a laminectomy is indicated, it is typically performed after the lateral mass screws are placed but before the rods are secured, to protect the neural elements from any downward force during screw insertion. The spinous processes are removed with a Leksell rongeur. A high-speed burr (matchstick or diamond tip) is then used to thin the laminae bilaterally at the junction of the lamina and lateral mass (the "gutter").
Once the laminae are paper-thin, a 1mm or 2mm Kerrison rongeur is used to carefully complete the osteotomy and remove the ligamentum flavum, allowing the laminae to be lifted off the dura en bloc. If radiculopathy is present, a posterior cervical foraminotomy is performed. This involves using a high-speed burr to resect the medial one-third of the facet joint (the inferior articular process of the cranial vertebra and the superior articular process of the caudal vertebra) to unroof the exiting nerve root. Excessive resection (greater than 50% of the facet joint) can lead to iatrogenic instability.
Instrumentation and Lateral Mass Fixation
Lateral mass screw fixation is the workhorse of subaxial posterior cervical stabilization. The two most common techniques are the Magerl and the Roy-Camille trajectories. The Magerl technique involves a starting point 1-2 mm medial and superior to the center of the lateral mass. The trajectory is 25-30 degrees lateral and 25-30 degrees cranial (parallel to the facet joint in the sagittal plane). This trajectory maximizes screw length and directs the screw away from the vertebral artery and exiting nerve root.
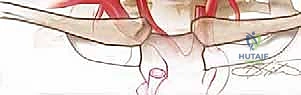
The Roy-Camille technique uses a starting point at the exact center of the lateral mass, with a trajectory that is straight anterior-posterior (perpendicular to the coronal plane) and 10 degrees lateral. Regardless of the technique, the lateral mass is typically decorticated with a burr, a pilot hole is drilled, the tract is palpated with a ball-tipped probe to ensure cortical integrity (especially anteriorly and medially), tapped, and the screw is inserted. Typical lateral mass screws are 3.5 mm in diameter and 12-16 mm in length.
Pedicle Screw Fixation and Arthrodesis
Cervical pedicle screws offer superior biomechanical pull-out strength compared to lateral mass screws and are particularly useful at the C2 and C7 levels, or in the subaxial spine when lateral masses are fractured or osteoporotic. C2 pedicle screws require a starting point in the cranial-medial quadrant of the C2 lateral mass, with a trajectory directed 20-30 degrees medially and 20-30 degrees cranially. C7 pedicle screws are technically demanding due to the narrow pedicle width and require precise preoperative CT templating and often intraoperative navigation or fluoroscopy.
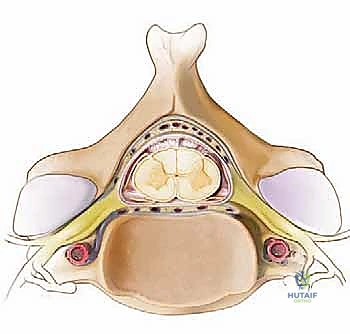
Following successful decompression and instrumentation, the final and arguably most critical step for long-term success is the arthrodesis. The lateral masses, facet joints, and any remaining posterior elements are aggressively decorticated using a high-speed burr until bleeding cancellous bone is exposed. Autologous local bone graft (harvested from the spinous processes and laminae), supplemented with allograft or osteoinductive biologics, is meticulously packed into the decorticated facet joints and along the lateral gutters. Rods are then contoured to match the desired cervical lordosis, seated into the screw heads, and secured with set screws.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, posterior cervical surgery carries a distinct profile of potential complications. A profound understanding of these risks and the immediate salvage protocols is what separates a competent surgeon from a master surgeon.
Intraoperative Complications and Management
Intraoperative complications demand immediate, protocol-driven responses. Vertebral artery injury is the most feared intraoperative complication, occurring in approximately 0.5% to 2% of posterior cervical instrumented cases. It typically occurs during lateral dissection, overly aggressive foraminotomy, or aberrant screw placement. If a pulsatile, bright red hemorrhage is encountered, immediate tamponade with hemostatic agents (e.g., Floseal, Surgicel) and cottonoid patties is required. The surgeon must never blindly coagulate or place clips in the bleeding pool. If the bleeding cannot be controlled, the screw (if it is the source) can sometimes be rapidly inserted to act as a plug. Postoperatively, an emergent angiogram is mandatory to assess for pseudoaneurysm, arteriovenous fistula, or the need for endovascular embolization. Crucially, if one vertebral artery is injured, surgery on the contralateral side must be aborted or modified to prevent bilateral injury, which is often fatal.
Dural tears occur in 1% to 5% of cases, often during the resection of severely hypertrophied or ossified ligamentum flavum. Primary repair with 4-0 or 5-0 non-absorbable suture is the gold standard. If primary repair is impossible, the defect should be covered with an onlay dural substitute and a layer of fibrin sealant. A watertight fascial closure is absolutely critical to prevent a postoperative cerebrospinal fluid (CSF) fistula.
| Complication | Estimated Incidence | Primary Salvage / Management Strategy |
|---|---|---|
| Vertebral Artery Injury | 0.5% - 2.0% | Immediate tamponade, avoid bilateral exposure, emergent postoperative endovascular angiography. |
| C5 Nerve Root Palsy | 5.0% - 12.0% | Rule out hematoma/misplaced screw. Usually managed conservatively with physical therapy; recovery can take 6-12 months. |
| Dural Tear (Incidental Durotomy) | 1.0% - 5.0% | Primary suture repair, dural patch/sealant, mandatory watertight fascial closure, possible lumbar drain. |
| Surgical Site Infection (SSI) | 2.0% - 4.0% | Prompt operative irrigation and debridement (I&D), targeted intravenous antibiotics, retention of hardware if stable. |
| Pseudoarthrosis (Non-union) | 2.0% - 10.0% | Revision surgery with anterior column support, optimization of biology (bone morphogenetic protein, smoking cessation). |
Postoperative Complications and Long-Term Sequelae
C5 nerve root palsy is a well-documented, frustrating complication unique to cervical decompression, occurring in up to 12% of cases. It typically presents as deltoid and biceps weakness 24 to 72 hours postoperatively, often without sensory deficit or pain. The exact pathophysiology is debated but is believed to involve a traction injury to the short C5 nerve root as the spinal cord drifts posteriorly following decompression (the "tethering" effect), or localized reperfusion injury. Management is largely supportive, involving physical therapy to prevent joint contracture; the majority of patients recover over 6 to 12 months.
Postoperative wound infections require aggressive management. Superficial infections may be treated with oral antibiotics, but deep fascial infections demand prompt return to the operating room for thorough irrigation and debridement. Instrumentation should generally be retained unless it is grossly loose, as instability will perpetuate the infection. Long-term sequelae include pseudoarthrosis (failure of fusion) and adjacent segment disease (ASD). Pseudoarthrosis presents with axial neck pain and hardware failure (broken rods or screws) and requires revision surgery. ASD is the biomechanical consequence of a rigid fusion segment placing increased stress on adjacent mobile segments, leading to accelerated degeneration that may require future extension of the fusion construct.
Phased Post-Operative Rehabilitation Protocols
The surgical intervention is only the first phase of the patient's recovery journey. A structured, phased rehabilitation protocol is essential to optimize functional outcomes, ensure osseous integration, and minimize postoperative morbidity.
Acute Postoperative Phase
The acute phase (Weeks 0-4) is focused on wound healing, pain control, and the protection of the surgical construct. Patients are typically mobilized on postoperative day one. The use of a rigid cervical collar (e.g., Aspen or Miami J collar) is surgeon-dependent but is frequently utilized for 4 to 6 weeks to provide external support, limit extremes of motion, and serve as a tactile reminder to the patient.
During this phase, physical therapy is strictly limited to gentle, active range of motion (within the confines of the collar, if used) and isometric neck strengthening. Patients are instructed in proper body mechanics, log-rolling techniques for bed mobility, and lifting restrictions (typically no more than 5-10 pounds). Aggressive stretching or resistance exercises are strictly contraindicated as they can stress the nascent fusion mass and instrumentation.
Subacute and Long-Term Rehabilitation
The subacute phase (Weeks 4-12) begins once early radiographic signs of fusion are noted and acute postoperative pain has subsided. The cervical collar is gradually weaned. Physical therapy transitions to active range of motion, progressive resistance exercises for the cervical and periscapular musculature, and proprioceptive training. Strengthening the trapezius, rhomboids, and levator scapulae is crucial for restoring the dynamic posterior tension band and preventing late post-surgical kyphosis.
Long-term rehabilitation (Months 3-12) focuses on functional restoration and return to pre-morbid activities. Cardiovascular conditioning is emphasized. Return to heavy labor or contact sports is generally delayed until 6 to 12 months postoperatively, contingent upon radiographic evidence of a solid, continuous bony arthrodesis across the decorticated facets and lateral masses. Patients are counseled that while the fusion limits motion at the operated segments, optimal rehabilitation of the surrounding musculature is vital to compensate for this biomechanical alteration.
Summary of Landmark Literature and Clinical Guidelines
The techniques and protocols utilized in posterior cervical surgery today are not arbitrary; they are the culmination of decades of rigorous biomechanical research and extensive clinical trials. Familiarity with this landmark literature is essential for the academic orthopedic surgeon.
Evidence-Based Outcomes and Future Directions
The seminal work by Magerl and Roy-Camille in the 1980s and 1990s revolutionized posterior cervical fixation, moving the field away from wire-based constructs to rigid screw-rod osteosynthesis. Their respective biomechanical and anatomical studies defined the safe trajectories for lateral mass screws that we utilize today. More recently, the AOSpine North America Cervical Spondylotic Myelopathy Study, led by Fehlings et al., provided robust, prospective evidence that both anterior and posterior approaches yield significant functional improvements in patients with CSM, highlighting that the choice of approach should be tailored to the specific pathoanatomy (e.g., sagittal alignment, number of levels) rather than a one-size-fits-all dogma.
Currently, clinical guidelines from the Cervical Spine Research Society (CSRS) and the North American Spine Society (NASS) strongly advocate for the use of intraoperative neuromonitoring during these procedures and emphasize the critical importance of preserving cervical lordosis to optimize clinical outcomes. The future of posterior cervical surgery is rapidly evolving with the integration of enabling technologies. Intraoperative CT navigation and robotic-assisted screw placement are increasingly utilized to enhance the accuracy of pedicle and lateral mass screw insertion, theoretically reducing the incidence of neurovascular complications. Furthermore, advancements in orthobiologics continue to refine our ability to achieve rapid and robust arthrodesis, minimizing the risk of pseudoarthrosis and hardware failure in complex reconstructive cases.
