Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater. Today, we are performing a Posterior Cervical Foraminotomy (PCF), a highly precise, motion-preserving procedure designed for the targeted treatment of cervical radiculopathy. Our primary surgical objective is to thoroughly decompress the exiting cervical nerve root, thereby alleviating the often debilitating motor and sensory deficits precipitated by foraminal stenosis or an intraforaminal soft disc herniation. Since its initial popularization by Scoville and Frykholm in the mid-20th century, the PCF has remained a cornerstone of the spine surgeon’s armamentarium. It is a nuanced procedure demanding meticulous anatomical understanding, superior spatial awareness, and exacting surgical precision.
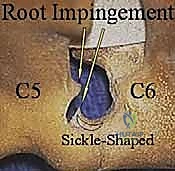
The pathophysiology of cervical radiculopathy is fundamentally driven by mechanical compression and secondary biochemical inflammation of the exiting cervical nerve root. This compression typically arises from degenerative cervical spondylosis, which encompasses osteophyte formation at the uncovertebral joints (joints of Luschka) anteriorly, or hypertrophy of the zygapophyseal (facet) joints posteriorly. Alternatively, acute soft disc herniations—particularly those located laterally within the foramen—can acutely compress the nerve root against the unyielding bony confines of the pedicles and articular processes. The mechanical deformation of the nerve root induces localized ischemia, disrupts axonal transport, and triggers a cascade of inflammatory mediators, resulting in the classic dermatomal pain, paresthesias, and myotomal weakness observed clinically.
Epidemiologically, cervical radiculopathy affects approximately 83 per 100,000 individuals annually, with a peak incidence in the fifth and sixth decades of life. The most frequently affected levels are C5-C6 and C6-C7, corresponding to the C6 and C7 nerve roots, respectively, due to the maximal biomechanical stress and sagittal plane motion occurring at these lower subaxial segments. While the natural history of cervical radiculopathy is often favorable—with many patients experiencing spontaneous resolution of symptoms over several months—a significant cohort will present with progressive neurologic deficits or intractable pain that is entirely refractory to comprehensive conservative management, thereby necessitating surgical intervention.
In the modern era of spine surgery, the Posterior Cervical Foraminotomy represents a critical paradigm of motion preservation. Unlike Anterior Cervical Discectomy and Fusion (ACDF), which permanently eliminates motion at the index segment and alters the biomechanics of the cervical spine, PCF achieves direct neural decompression without the morbidity of arthrodesis. By preserving the intervertebral disc and the majority of the facet joint complex, PCF mitigates the risk of adjacent segment disease (ASD), avoids the complications associated with anterior approaches (such as dysphagia and recurrent laryngeal nerve palsy), and eliminates the need for expensive instrumentation and biologic bone grafts.

Detailed Surgical Anatomy and Biomechanics
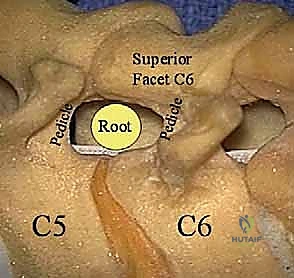
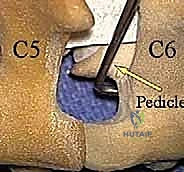
Before initiating any surgical incision, a profound mastery of the three-dimensional surgical anatomy is paramount to ensure both safe decompression and the preservation of spinal stability. The intervertebral foramen, the anatomical aperture through which our target nerve root exits, is a complex, dynamic tunnel. Ventrally, it is bounded by the posterior aspect of the vertebral body, the intervertebral disc, and the uncinate processes. Cranially and caudally, the rigid cortical borders of the adjacent pedicles define the superior and inferior limits of the foramen. Dorsally, the boundary is formed by the superior articular facet of the caudal vertebral segment and the overlying ligamentum flavum.
In a robust, healthy adult, the subaxial cervical foramen averages 9 to 12 mm in height and 4 to 6 mm in width. The exiting cervical nerve root, enveloped in its dural sleeve, typically occupies only about one-third of this available cross-sectional area. However, with advancing age and spondylosis, degenerative changes such as uncovertebral osteophytosis, disc space collapse, and facet hypertrophy can drastically diminish this reserve capacity. Biomechanically, the foramen undergoes dynamic changes during physiological motion; extension and ipsilateral rotation further decrease foraminal volume, exacerbating nerve root impingement, whereas flexion increases foraminal dimensions, which dictates our intraoperative patient positioning strategy.
The neurovascular risks inherent to this procedure are significant and require absolute vigilance. Directly medial to our operative field lies the spinal cord, vulnerable to direct mechanical trauma, thermal injury from electrocautery or high-speed burrs, and ischemic insult. The cervical nerve roots themselves are exquisitely sensitive; excessive traction during microdiscectomy or direct trauma can precipitate iatrogenic motor deficits, most notably the dreaded C5 palsy. Furthermore, the dorsal root ganglion (DRG), typically situated within the medial aspect of the foramen, is highly sensitive to manipulation and can be a source of profound postoperative dysesthesia if traumatized.
Vascular considerations are equally critical. The vertebral artery ascends through the transverse foramina from C6 to C1, lying immediately anterior to the exiting nerve root. While generally protected during a standard posterior approach, anomalous medial deviation of the artery or overly aggressive lateral bony resection can result in catastrophic hemorrhage or ischemic stroke. Additionally, a dense, valveless epidural venous plexus (Batson’s plexus) surrounds the nerve root and thecal sac. These veins are prone to profuse bleeding if not meticulously managed with bipolar electrocautery and hemostatic agents, potentially obscuring the microscopic surgical field and leading to postoperative epidural hematoma.
Exhaustive Indications and Contraindications
| Category | Specific Clinical Scenarios |
|---|---|
| Primary Indications | Unilateral cervical radiculopathy refractory to 6-12 weeks of conservative care; Progressive motor weakness in a specific myotome; Intractable, severe radicular pain; Foraminal soft disc herniation; Unilateral foraminal stenosis due to facet hypertrophy. |
| Relative Indications | Bilateral radiculopathy (requiring bilateral foraminotomies); Previous anterior cervical fusion with adjacent segment foraminal stenosis; Patients at high risk for anterior approach complications (e.g., severe dysphagia, prior neck irradiation). |
| Absolute Contraindications | Cervical myelopathy; Central spinal canal stenosis; Cervical instability or dynamic subluxation (>3.5mm translation or >11 degrees angulation); Severe cervical kyphosis; Predominant axial neck pain without radicular symptoms. |
| Relative Contraindications | Broad-based central disc herniations; Severe anterior uncovertebral osteophytes causing bilateral symptoms; Profound osteoporosis (if facet fracture risk is high). |
Cervical radiculopathy is fundamentally a clinical diagnosis, manifesting as localized motor weakness, sensory aberrations, or diminished reflexes in a strictly dermatomal and myotomal distribution. During the preoperative evaluation, the surgeon must exhaustively assess the patient for the chronicity of symptoms, the exact anatomical distribution of pain, and the presence of objective neurologic deficits. A critical threshold for surgical intervention is the failure of an appropriate, structured course of nonsurgical treatment lasting a minimum of 6 to 12 weeks. This conservative regimen should encompass targeted physical therapy, nonsteroidal anti-inflammatory drugs (NSAIDs), oral corticosteroids, and fluoroscopically guided selective nerve root blocks, which serve an invaluable dual purpose as both diagnostic and therapeutic modalities.
The ideal candidate for a Posterior Cervical Foraminotomy presents with pathology that is distinctly localized to the intervertebral foramen. This includes lateral soft disc herniations that are difficult to access via an anterior approach without extensive uncinate resection, and foraminal stenosis driven primarily by posterior facet joint hypertrophy. The procedure is particularly advantageous for younger patients with soft disc herniations, where preservation of the native motion segment is highly desirable to prevent the long-term biomechanical consequences of arthrodesis. It is also an excellent salvage option for patients who have developed adjacent segment foraminal stenosis following a previous ACDF.
Conversely, the surgeon must maintain a broad differential diagnosis and rigorously rule out conditions that contraindicate a posterior foraminal approach. Cervical myelopathy, characterized by upper motor neuron signs such as hyperreflexia, a positive Hoffman's sign, gait ataxia, and bowel or bladder dysfunction, represents an absolute contraindication to an isolated PCF. These patients require central canal decompression, typically via laminectomy or anterior corpectomy. Furthermore, peripheral nerve entrapment syndromes—such as carpal tunnel syndrome, cubital tunnel syndrome, or thoracic outlet syndrome—can closely mimic cervical radiculopathy ("double crush syndrome") and must be excluded through comprehensive electromyography (EMG) and nerve conduction velocity (NCV) studies.
Finally, the biomechanical stability of the cervical spine must be heavily weighted in the decision-making process. PCF is contraindicated in the presence of gross cervical instability, defined radiographically as greater than 3.5 mm of translation or greater than 11 degrees of angular difference on dynamic lateral flexion-extension radiographs. Performing a foraminotomy, which inherently requires the resection of a portion of the stabilizing facet joint complex, in an already unstable spine will inevitably precipitate progressive deformity, kyphosis, and worsening neurologic compromise. Similarly, patients with a primary complaint of axial neck pain without correlating radicular symptoms will invariably experience poor outcomes with this procedure and should be managed non-operatively or evaluated for alternative pathologies.
Pre-Operative Planning, Templating, and Patient Positioning
Surgical success in posterior cervical decompression is predicated upon exhaustive preoperative planning and meticulous patient positioning. The diagnostic workup must begin with high-quality, weight-bearing plain radiographs, inclusive of anteroposterior, lateral, oblique, and dynamic flexion-extension views. These images are critical for assessing overall cervical alignment, identifying resting or dynamic instability, and evaluating gross osseous degenerative changes. Oblique views are particularly useful for visualizing the osseous borders of the intervertebral foramina and identifying uncinate or facet osteophytes.
Advanced cross-sectional imaging is non-negotiable. Magnetic Resonance Imaging (MRI) without contrast is the gold standard for soft tissue evaluation, providing unparalleled visualization of the spinal cord, nerve roots, intervertebral discs, and ligamentous structures. It is essential for differentiating between soft disc herniations and hard osteophytic ridges. In cases where MRI is contraindicated (e.g., non-compatible pacemakers) or when detailed osseous morphology is required to plan the extent of bony resection, a high-resolution Computed Tomography (CT) scan with sagittal and coronal reconstructions, or a CT myelogram, is invaluable. These modalities allow the surgeon to precisely template the dimensions of the foramen and anticipate the anatomical challenges specific to the patient's pathology.
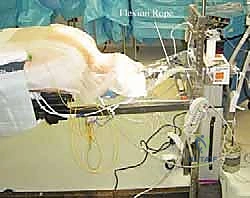
Proper patient positioning on the operating table is a critical phase of the procedure, directly influencing intraoperative blood loss, visualization, and patient safety. We routinely utilize an open Jackson frame (OSI, Orthopaedic Systems, Inc.) with the patient in the prone position. The chest and pelvis are supported on radiolucent bolsters, allowing the abdomen to hang entirely free. This maneuver is vital; it prevents elevated intra-abdominal pressure from transmitting to the epidural venous plexus via the vena cava, thereby drastically reducing epidural bleeding and ensuring a clear, dry surgical field. The table is subsequently tilted into reverse Trendelenburg to further facilitate venous drainage away from the cervical spine.
Cranial fixation is achieved using bivector Gardner-Wells tong traction or a Mayfield skull clamp. The bivector system allows for precise, dynamic control of the cervical spine's sagittal alignment. For a foraminotomy, the neck is positioned in maximal safe flexion. This "military tuck" position effectively "unshingles" the cervical facet joints, opening the posterior elements and increasing the interlaminar space. This reduces the amount of overlying inferior articular process that must be resected to access the nerve root, preserving maximum facet integrity. The shoulders are firmly taped down with broad adhesive tape applying caudal traction, which is essential for obtaining unobstructed intraoperative lateral fluoroscopic images of the lower cervical spine (C6-T1). Throughout this process, continuous communication with the anesthesia team is mandatory to ensure the endotracheal tube remains secure and the spinal cord is not compromised during dynamic positioning.
Step-by-Step Surgical Approach and Fixation Technique
Initial Incision and Superficial Exposure
Following meticulous sterile preparation and draping, intraoperative fluoroscopy is utilized to localize the precise operative level prior to making the incision. This step is critical to avoid wrong-level surgery, a devastating and entirely preventable complication. For a unilateral foraminotomy, a linear vertical incision is made approximately 1.5 to 2.0 cm lateral to the midline on the symptomatic side, centered over the target interspace. If bilateral pathology is being addressed, a standard midline incision is utilized.
Using monopolar electrocautery, the dissection proceeds through the subcutaneous adipose tissue and superficial fascia. Hemostasis must be meticulously maintained from the outset to prevent blood from pooling in the deep, narrow surgical corridor. When utilizing a midline approach, the surgeon must carefully identify the ligamentum nuchae, the avascular midline raphe. Dissecting strictly within this plane minimizes muscle trauma and bleeding. The raphe is split down to the apices of the spinous processes of the cranial and caudal vertebrae defining the target level.
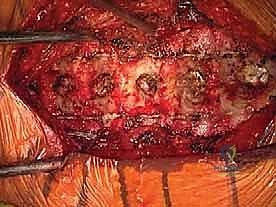
Deep Dissection and Landmark Identification
Once the spinous processes are identified, a Cobb elevator and Bovie electrocautery are used to perform a subperiosteal dissection of the paraspinal musculature (multifidus and semispinalis cervicis) off the lamina. The dissection is carried laterally to expose the junction of the lamina and the lateral mass. It is imperative to maintain the dissection strictly subperiosteally to minimize bleeding and muscle necrosis. At the C2-C3 level, extreme care must be taken to preserve the attachments of the semispinalis cervicis to the C2 spinous process to prevent postoperative kyphosis and intractable axial neck pain.
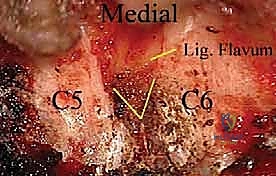
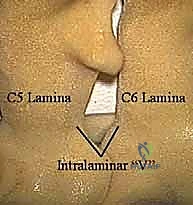
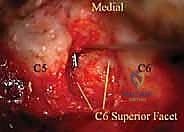
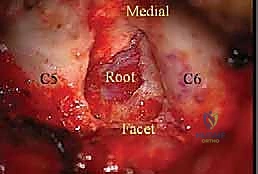
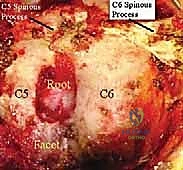
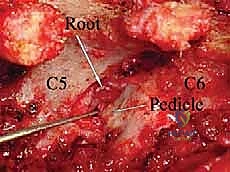
The lateral dissection is complete when the medial aspect of the facet joint is visualized. We must identify the "V-point"—the anatomical junction where the inferior margin of the cranial lamina meets the superior margin of the caudal lamina, directly medial to the facet joint. This V-point overlies the medial aspect of the underlying nerve root and serves as our primary anatomical landmark for the initiation of the bony decompression. The facet capsule should be carefully preserved outside of the planned resection zone to maintain biomechanical stability. A self-retaining retractor, such as a Williams or a tubular retractor system if performing minimally invasive surgery (MIS), is then deployed to maintain the exposure.
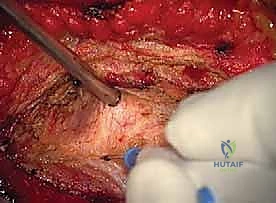
The Bony Foraminotomy and Decompression
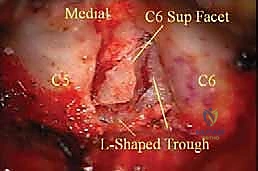
Under the magnification and illumination of the surgical microscope or high-powered loupes, the bony resection commences. A high-speed drill with a 3mm or 4mm matchstick or diamond burr is utilized to thin the inferior margin of the cranial lamina, the superior margin of the caudal lamina, and the medial aspect of the intervening facet joint. The burring is performed in a sweeping motion, gradually egg-shelling the cortical bone until the underlying ligamentum flavum is exposed.
The critical biomechanical rule during this phase is the "50% Rule." Biomechanical studies dictate that resecting more than 50% of the facet joint complex (specifically the pars interarticularis and the articular processes) significantly compromises the structural integrity of the motion segment, leading to iatrogenic instability. Therefore, the lateral extent of the foraminotomy must be strictly limited to the medial half of the facet joint. Once the bone is adequately thinned, 1mm and 2mm Kerrison rongeurs are used to carefully resect the remaining bone and the underlying ligamentum flavum, officially entering the epidural space.
Nerve Root Exposure and Microdiscectomy
With the ligamentum flavum removed, the lateral margin of the thecal sac and the origin of the exiting cervical nerve root are identified. The nerve root is often obscured by a rich plexus of epidural veins. These veins must be managed with precise bipolar electrocautery and the application of hemostatic matrix (e.g., Floseal or Gelfoam) to prevent troublesome bleeding that can obscure the microscopic field. The decompression is carried laterally along the course of the nerve root, utilizing small Kerrison rongeurs to unroof the foramen until the pedicle of the caudal vertebra is palpated inferiorly and the pedicle of the cranial vertebra is palpated superiorly.
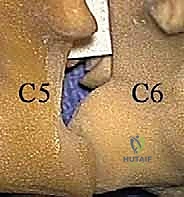
If the indication for surgery is a soft disc herniation, the axilla of the nerve root (the space between the exiting root and the lateral thecal sac) is gently explored using a micro-nerve hook. The nerve root is delicately mobilized superiorly to expose the underlying disc space. It is crucial to avoid excessive medial retraction of the spinal cord or aggressive manipulation of the dorsal root ganglion.
Once the herniated disc fragment is identified, an annulotomy is performed if the fragment is contained, or the extruded fragment is directly grasped. Micro-pituitary rongeurs are utilized to extract the offending disc material. The decompression is considered complete when the nerve root is visually lax, pulsatile, and easily mobilized with a blunt probe, and a micro-hook can be passed freely out the lateral aspect of the foramen without encountering osseous or soft tissue resistance.
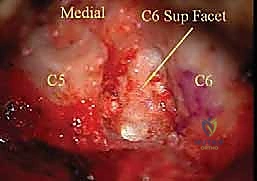
Hemostasis and Layered Closure
Following thorough decompression, the surgical bed is copiously irrigated with sterile saline to remove bone dust and debris. Absolute hemostasis is achieved. The epidural space is inspected, and any residual venous oozing is controlled with absorbable hemostatic agents. We avoid leaving large pieces of hemostatic material in the epidural space, as they can swell and cause secondary compression.
The wound is then closed in multiple anatomical layers to eliminate dead space and prevent the formation of postoperative seromas or hematomas. The deep paraspinal musculature is reapproximated using heavy absorbable sutures (e.g., 0 or 1 Vicryl). The thick fascial layer is closed meticulously in a watertight fashion.
The subcutaneous tissues are approximated with inverted absorbable sutures, and the skin is closed with a running subcuticular suture or surgical staples, depending on surgeon preference and tissue quality. A sterile dressing is applied, and the patient is carefully transitioned from the prone position back to the supine position on the stretcher, with continuous monitoring of neurologic status during the transfer.
Complications, Incidence Rates, and Salvage Management
| Complication | Estimated Incidence | Avoidance Strategy | Salvage/Management Protocol |
|---|---|---|---|
| C5 Nerve Root Palsy | 2% - 5% | Avoid excessive traction on the root; limit posterior cord drift. | High-dose corticosteroids; physical therapy; typically resolves spontaneously over 3-6 months. |
| Incidental Durotomy | 1% - 3% |
