Multiple Hereditary Exostoses (MHE): Pathophysiology, Clinical Features, & Orthopedic Management
Key Takeaway
Multiple Hereditary Exostoses (MHE) is a genetic disorder forming osteochondromas. Key complications are skeletal deformities (e.g., genu valgum), pain, and neurovascular compression. The most serious risk is malignant transformation into a secondary peripheral chondrosarcoma. Lifelong monitoring and targeted surgical intervention are crucial for managing MHE patients effectively.
Introduction and Epidemiology
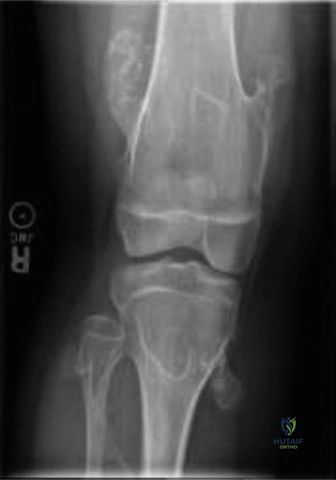
Multiple Hereditary Exostoses (MHE), also referred to as diaphyseal aclasia or familial osteochondromatosis, is an autosomal dominant skeletal dysplasia characterized by the development of multiple osteochondromas. These benign, cartilage-capped bony outgrowths typically arise from the juxta-epiphyseal region of long bones, particularly affecting the metaphyses, and propagate away from the adjacent joint as the bone lengthens. The condition exhibits profound genetic heterogeneity, primarily linked to loss-of-function mutations in either the EXT1 gene located on chromosome 8q24.1 or the EXT2 gene on chromosome 11p11-p12. These genes encode critical glycosyltransferases responsible for the polymerization of heparan sulfate chains. The subsequent deficiency in heparan sulfate proteoglycans disrupts multiple signaling pathways within the growth plate, most notably the Indian Hedgehog (IHH) and Parathyroid Hormone-related Protein (PTHrP) feedback loop, which is essential for normal chondrocyte proliferation and differentiation.
The incidence of MHE is estimated to be approximately 1 in 50,000 live births, establishing it as the most prevalent hereditary skeletal disorder. Penetrance is nearly 100% by adolescence, though expressivity varies dramatically, even among individuals sharing the identical familial mutation. Patients present with a highly variable spectrum of clinical manifestations. Some individuals harbor only a few asymptomatic exostoses, while others suffer from widespread skeletal involvement resulting in severe angular deformities, limb length discrepancies, chronic pain, and profound functional limitations. Exostoses typically initiate development during early childhood and theoretically cease to enlarge upon skeletal maturity and physeal closure.
However, their presence during the critical phases of skeletal growth disrupts normal endochondral ossification, leading to a constellation of secondary structural deformities. Lifelong clinical and radiographic surveillance is imperative for detecting and managing progressive complications. These include mechanical irritation of adjacent soft tissues, neurovascular impingement, progressive joint subluxation, and the most critical risk: malignant transformation into secondary peripheral chondrosarcoma. The lifetime risk of malignant degeneration in MHE patients is estimated between 1% and 5%, significantly higher than in patients with solitary osteochondromas, necessitating a high index of suspicion for any lesion demonstrating interval growth or new-onset pain following skeletal maturity.
Surgical Anatomy and Biomechanics
Exostoses in MHE are true osteocartilaginous lesions. They are composed of a hyaline cartilage cap that undergoes endochondral ossification, forming a trabecular bony stalk that is directly continuous with the medullary cavity and cortical bone of the underlying parent bone. Pathophysiologically, they originate from ectopic growth plate cartilage cells that herniate through a localized defect in the perichondrial ring of LaCroix at the groove of Ranvier. This pathological growth mechanism explains their characteristic orientation pointing away from the adjacent joint and their cessation of growth at skeletal maturity when the ectopic physis fuses.
Morphologically, exostoses present in two primary forms: pedunculated (having a narrow stalk and a bulbous tip) and sessile (having a broad-based attachment to the host bone). The morphological variant heavily influences the biomechanical impact and the surgical approach required for extirpation.

Predilection Sites and Regional Biomechanics
The anatomical distribution of exostoses dictates the specific biomechanical derangements observed in MHE:
- Distal Femur and Proximal Tibia and Fibula: These are the most rapidly growing physes in the body and consequently the most frequent sites for exostosis formation. Asymmetrical physeal tethering here frequently results in progressive genu valgum, rotational malalignment, and clinically significant limb length discrepancies.
- Distal Radius and Ulna: The forearm is disproportionately affected due to the small cross-sectional area of the bones relative to the size of the exostoses. Ulnar growth arrest is the hallmark, leading to relative radial overgrowth, radial bowing, positive ulnar variance, and progressive subluxation or dislocation of the radial head. This cascade is often referred to as a "Madelung-like" deformity.
- Distal Tibia and Fibula: Exostoses arising from the distal fibula can cause premature fusion or tethering of the lateral physis, leading to relative tibial overgrowth and a secondary ankle valgus deformity. This alters tibiotalar contact mechanics, predisposing the joint to early-onset osteoarthritis.
- Proximal Humerus and Scapula: Lesions in the shoulder girdle can cause mechanical block to shoulder range of motion, particularly abduction and external rotation. Scapular exostoses frequently present with painful snapping scapula syndrome.
- Pelvis and Axial Skeleton: Pelvic lesions are often large and sessile, potentially causing impingement on the sciatic nerve or intrapelvic structures. Spinal exostoses, while rarer, carry the severe risk of spinal cord or nerve root compression.
Pathoanatomy of Secondary Deformities
The biomechanical consequences of MHE extend far beyond the space-occupying nature of the lesions. The primary driver of functional impairment is growth plate tethering. According to the Hueter-Volkmann principle, increased compression across a physis retards growth. An exostosis bridging the metaphysis and bridging closely to the physis acts as a mechanical tether.
Furthermore, neurovascular compression is a critical pathoanatomical consideration. The common peroneal nerve is uniquely vulnerable as it courses around the fibular neck, where proximal fibular exostoses frequently arise. Similarly, posterior distal femoral exostoses can displace and compress the popliteal artery and vein, leading to claudication, venous stasis, or pseudoaneurysm formation secondary to chronic mechanical friction.
Indications and Contraindications
The management of MHE requires a nuanced approach, balancing the risks of surgical intervention against the natural history of the disease. Prophylactic excision of asymptomatic lesions is universally contraindicated due to the multiplicity of lesions and the inherent risks of surgery. Intervention is strictly reserved for symptomatic lesions, progressive deformities, or suspicion of malignancy.
Operative Versus Non Operative Management Parameters
| Clinical Scenario | Non Operative Management | Operative Management |
|---|---|---|
| Asymptomatic Exostosis | Routine clinical surveillance; annual radiographs during growth. | Contraindicated (unless high risk of impending joint subluxation). |
| Mechanical Pain or Bursitis | Activity modification, NSAIDs, localized corticosteroid injections (for bursitis). | Excision of the exostosis and overlying bursa if conservative measures fail. |
| Neurovascular Compromise | Not applicable. | Urgent surgical decompression and complete excision of the offending lesion. |
| Angular Deformity (e.g. Genu Valgum) | Observation if mild and non-progressive. | Guided growth (hemiepiphysiodesis) in skeletally immature; Corrective osteotomy in mature patients. |
| Forearm Deformity | Observation if functional ROM is maintained and radial head is stable. | Ulnar lengthening, radial osteotomy, or excision of tethering lesions to prevent radial head dislocation. |
| Limb Length Discrepancy | Shoe lifts for discrepancies < 2 cm. | Epiphysiodesis of the longer limb or distraction osteogenesis of the shorter limb for discrepancies > 2 cm. |
| Suspected Malignant Degeneration | Serial MRI if findings are equivocal and lesion is small. | Wide en bloc resection if MRI confirms cartilage cap > 1.5cm in adults, or biopsy confirms chondrosarcoma. |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is the cornerstone of successful surgical outcomes in MHE, given the complex three-dimensional anatomy of the deformities and the proximity of exostoses to critical neurovascular structures.
Advanced Imaging Protocols
Standard orthogonal radiographs remain the initial imaging modality of choice to assess the size, location, and base (sessile vs. pedunculated) of the exostosis, as well as to quantify angular deformities using standard mechanical axis deviation (MAD), lateral distal femoral angle (LDFA), and medial proximal tibial angle (MPTA) measurements.
For surgical planning, particularly in areas with complex overlapping anatomy (e.g., pelvis, scapula, spine, and complex forearm deformities), computed tomography (CT) with 3D reconstruction is invaluable. CT precisely delineates the bony stalk and its relationship to the host bone cortex, aiding in the determination of osteotomy trajectories.
Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the cartilage cap. In skeletally mature patients, a cartilage cap thickness exceeding 1.5 cm is highly suspicious for secondary peripheral chondrosarcoma and necessitates a fundamental shift in surgical strategy from marginal excision to wide en bloc resection. MRI is also critical for assessing the relationship of the exostosis to adjacent nerves, vessels, and muscle bellies, particularly in the popliteal fossa or neurovascular bundles of the arm.
Patient Positioning and Setup
Positioning is dictated by the anatomical location of the targeted lesions.
* Lower Extremity: The patient is positioned supine on a radiolucent table to facilitate intraoperative fluoroscopy. A sterile tourniquet is utilized for distal femoral, tibial, and foot/ankle procedures to ensure a bloodless field, aiding in the identification of neurovascular structures displaced by the exostosis.
* Forearm and Upper Extremity: The patient is placed supine with the operative arm extended on a radiolucent hand table. Tourniquet control is mandatory.
* Pelvis and Proximal Femur: Lateral decubitus or floppy lateral positioning may be required depending on whether the lesion is situated on the iliac wing, ischium, or proximal femur. Fluoroscopy must be readily available to confirm complete resection of the base.
Detailed Surgical Approach and Technique
Surgical intervention in MHE encompasses three distinct categories of procedures: simple excision of symptomatic exostoses, corrective osteotomies for angular deformities, and complex reconstructive procedures for joint subluxation or limb length discrepancies.
Principles of Exostosis Excision
The primary objective of exostosis excision is the complete removal of the osteochondroma, including its entire cartilage cap and the overlying perichondrium. Failure to remove the perichondrial layer significantly increases the risk of local recurrence, particularly in skeletally immature patients.
- Surgical Approach: Incisions should be meticulously planned to allow for extensile exposure if necessary, taking into account the displaced anatomy. Standard internervous planes are utilized whenever possible. For example, a direct lateral approach to the distal femur or a subvastus approach is commonly employed for lateral or medial distal femoral lesions, respectively.
- Soft Tissue Dissection: The exostosis is often covered by a reactive bursa. This bursa should be excised. The overlying muscles and tendons are frequently stretched over the lesion; these must be carefully mobilized and retracted. The periosteum of the host bone is incised at the base of the exostosis.
- Osteotomy of the Stalk: For pedunculated lesions, the stalk is transected at its base, flush with the normal cortical contour of the host bone, using a sharp osteotome or an oscillating saw. For sessile lesions, a more extensive contouring of the host bone is required. Care must be taken not to violate the structural integrity of the host diaphysis or metaphysis, which could precipitate an iatrogenic fracture.
- Cartilage Cap Verification: The excised specimen must be inspected on the back table to ensure the cartilage cap is intact and has not been fragmented during removal.
- Bone Contouring and Hemostasis: The residual base on the host bone is smoothed using a high-speed burr or rasp. Bone wax is applied to the exposed cancellous bone to minimize postoperative hematoma formation, which can serve as a nidus for heterotopic ossification or recurrence.
Management of Lower Extremity Angular Deformities
Genu valgum and ankle valgus are the most common angular deformities requiring intervention.
- Guided Growth (Hemiepiphysiodesis): In skeletally immature patients with significant growth potential remaining, guided growth utilizing tension band plates (e.g., eight-Plates) is the preferred minimally invasive technique. For genu valgum, a plate is applied across the medial physis of the distal femur or proximal tibia. As the lateral physis continues to grow, the deformity gradually corrects. Close radiographic follow-up is mandatory to prevent overcorrection, and the hardware must be removed once neutral mechanical alignment is achieved.
- Corrective Osteotomy: In skeletally mature patients, or those with severe deformities unresponsive to guided growth, acute correction via osteotomy is indicated. For distal femoral valgus, a medial closing-wedge or lateral opening-wedge osteotomy is performed, stabilized with a rigid locking plate. Tibial and fibular osteotomies may be required for complex multi-planar deformities of the knee or ankle.
Management of Forearm Deformities
Forearm reconstruction in MHE is highly complex and guided by the Masada classification system, which categorizes deformities based on the location of the primary exostosis and the degree of radial head subluxation.
- Ulnar Lengthening: The cornerstone of treating Masada Type I and II deformities (characterized by significant ulnar shortening) is ulnar lengthening. This can be achieved acutely with a structural bone graft and rigid plate fixation if the discrepancy is small. However, for discrepancies exceeding 2-3 cm, gradual distraction osteogenesis using a monolateral or circular external fixator is preferred. This allows for concomitant correction of angular deformity and protects the soft tissues from acute stretch injuries.
- Radial Osteotomy: If severe radial bowing is present and limits forearm supination and pronation, a corrective closing-wedge osteotomy of the radius may be performed simultaneously with ulnar lengthening.
- Excision of Tethering Lesions: Exostoses arising from the distal ulna or the proximal radius that are actively tethering growth or mechanically blocking the distal radioulnar joint (DRUJ) or proximal radioulnar joint (PRUJ) must be excised concurrently.
Oncologic Resection for Malignant Transformation
If preoperative MRI or core needle biopsy suggests malignant transformation to a secondary peripheral chondrosarcoma, the surgical paradigm shifts dramatically. Intracapsular excision is strictly contraindicated due to the risk of tumor seeding. The lesion must be treated with wide en bloc resection, ensuring negative margins. This often requires complex skeletal reconstruction utilizing massive allografts, endoprostheses, or vascularized fibular grafts, depending on the anatomical location and the extent of the resection.
Complications and Management
Surgical intervention in MHE carries a unique set of complications related to the altered local anatomy and the underlying genetic defect. Anticipation and prompt management of these complications are critical.
Common Surgical Complications and Salvage Strategies
| Complication | Incidence and Risk Factors | Prevention and Management Strategies |
|---|---|---|
| Local Recurrence | 5-10% in skeletally immature patients; higher if incomplete excision of the cartilage cap or perichondrium occurs. | Prevention: Ensure meticulous extracapsular excision; remove the entire perichondrial sleeve. Management: Repeat excision if the recurrent lesion becomes symptomatic or causes new deformity. |
| Neurovascular Injury | Variable depending on location. High risk with proximal fibular (peroneal nerve) and distal femoral (popliteal vessels) lesions. | Prevention: Meticulous preoperative MRI planning; wide exposure; identification and protection of nerves prior to osteotomy. Management: Immediate intraoperative repair if transected. Observation and EMG for neuropraxia. |
| Iatrogenic Fracture | Rare, but risk increases with excision of large sessile lesions that compromise cortical integrity. | Prevention: Avoid overly aggressive cortical resection; use high-speed burrs for final contouring rather than heavy osteotomes. Management: Intraoperative prophylactic fixation with plates/screws; postoperative immobilization. |
| Overcorrection / Undercorrection | Common in guided growth procedures if follow-up is inadequate. | Prevention: Strict 3-4 month radiographic follow-up intervals during guided growth. Management: Prompt hardware removal upon achieving neutral alignment; reverse guided growth or acute osteotomy for severe overcorrection. |
| Nonunion / Delayed Union | 5-15% following corrective osteotomies or ulnar lengthening procedures. | Prevention: Rigid internal fixation; preservation of periosteal blood supply; optimization of bone graft in acute lengthening. Management: Revision internal fixation with autologous bone grafting; optimization of metabolic factors. |
| Joint Stiffness | Common following complex forearm reconstructions or intra-articular resections. | Prevention: Meticulous hemostasis to prevent hematoma; early implementation of structured physical therapy. Management: Aggressive rehabilitation; manipulation under anesthesia or arthroscopic lysis of adhesions for refractory cases. |
Post Operative Rehabilitation Protocols
Rehabilitation protocols must be highly individualized, dictated by the specific anatomical site and the nature of the surgical intervention. The overarching goals are to protect the surgical repair, restore full range of motion, and optimize functional strength.
Protocol for Simple Exostosis Excision
Following straightforward excision of a pedunculated or sessile exostosis without structural compromise of the host bone, rehabilitation focuses on early mobilization.
* Phase I (0-2 weeks): The extremity is placed in a soft compressive dressing. Weight-bearing is typically allowed as tolerated for lower extremity procedures, utilizing crutches for balance and pain control. Active and active-assisted range of motion (ROM) exercises of adjacent joints are initiated immediately to prevent stiffness and minimize the risk of deep vein thrombosis (DVT).
* Phase II (2-6 weeks): Suture removal occurs at 10-14 days. Patients progress to full weight-bearing. Strengthening exercises targeting the musculature mobilized during the surgical approach are instituted. Return to normal activities and light sports is generally permitted by 4-6 weeks, provided the patient is pain-free and demonstrates symmetric strength.
Protocol for Corrective Osteotomies and Lengthening
Rehabilitation following osteotomies (e.g., distal femoral varus producing osteotomy) or distraction osteogenesis (e.g., ulnar lengthening) is significantly more protracted and requires strict adherence to biomechanical constraints.
* Phase I (0-6 weeks): For lower extremity osteotomies stabilized with plates, patients are typically restricted to touch-down weight-bearing (TDWB) or partial weight-bearing (PWB) to protect the fixation construct. Early ROM is encouraged to prevent arthrofibrosis. For external fixators used in lengthening, meticulous daily pin site care using chlorhexidine or saline is mandatory to prevent superficial pin tract infections. Distraction typically commences 7-10 days postoperatively at a rate of 1 mm per day, divided into 4 increments.
* Phase II (6-12 weeks): Radiographic assessment is critical to evaluate callus formation and progression of union. Weight-bearing is gradually advanced based on radiographic evidence of healing. For lengthening procedures, the consolidation phase begins once the desired length is achieved; the fixator remains in place to act as a neutralization device while the regenerate bone mineralizes.
* Phase III (3-6 months): Full weight-bearing is established. Intensive physical therapy focuses on restoring terminal ROM, proprioception, and advanced strengthening. External fixators are removed only when corticalization of the regenerate bone is visible on at least three out of four cortices on orthogonal radiographs.
Summary of Key Literature and Guidelines
The orthopedic management of Multiple Hereditary Exostoses is guided by a robust body of literature focusing on deformity classification, surgical timing, and oncologic surveillance.
- Forearm Deformity Classification (Masada et al., 1989): The seminal paper by Masada established the widely utilized classification system for forearm deformities in MHE. This system categorizes deformities based on the primary site of exostosis formation (distal ulna, proximal radius, or both) and the presence of radial head dislocation, providing a critical framework for surgical decision-making regarding ulnar lengthening and radial osteotomy.
- Guided Growth Efficacy (Stevens, 2007): Literature surrounding the use of tension band plates for hemiepiphysiodesis has revolutionized the management of angular deformities in the growing child with MHE. The transition from invasive osteotomies to minimally invasive guided growth has significantly reduced morbidity and complication rates for genu valgum and ankle valgus.
- Malignant Transformation and Surveillance Guidelines (Bovée et al., World Health Organization): The WHO classification of soft tissue and bone tumors provides the definitive criteria for differentiating benign osteochondromas from secondary peripheral chondrosarcomas. Current international consensus guidelines strongly advocate for standardized clinical surveillance. While routine whole-body MRI for asymptomatic pediatric patients remains controversial due to cost and logistical challenges, there is universal agreement on the necessity of immediate advanced imaging (MRI) for any lesion demonstrating interval growth, new-onset pain, or neurovascular symptoms in a skeletally mature patient. A cartilage cap thickness exceeding 1.5 cm on T2-weighted MRI in an adult is the most reliable imaging biomarker for malignant transformation, dictating an immediate shift to oncologic resection principles.
- Quality of Life and Functional Outcomes: Recent literature emphasizes that while MHE is a benign skeletal dysplasia, the cumulative burden of multiple surgeries, chronic pain, and cosmetic deformity significantly impacts the psychosocial well-being of patients. Multidisciplinary care, incorporating orthopedic surgery, pain management, and psychological support, is now recognized as the gold standard for comprehensive patient management.
