Patient Presentation & History
We present a typical, yet diagnostically challenging, case of Osteoid Osteoma in an active young patient, highlighting the characteristic clinical features and the diagnostic journey.
Patient Demographics:
A 16-year-old male, otherwise fit and healthy, presented with a 9-month history of progressive right ankle pain. He is an avid soccer player, active in school sports.
Onset and Characterization of Pain:
The patient initially reported vague, intermittent ache in his right ankle, predominantly over the lateral malleolus. Over several months, the pain progressively worsened in intensity and frequency. Key characteristics noted were:
*
Nocturnal Exacerbation:
The pain was consistently worse at night, frequently waking him from sleep.
*
Relief with NSAIDs:
He reported near-complete relief with over-the-counter non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen, which he was taking almost daily by the time of presentation. This response was a crucial diagnostic clue.
*
Activity-Related Aggravation:
While present at rest, the pain was exacerbated by physical activity, particularly running and soccer.
*
Localization:
The pain was consistently localized to the lateral aspect of the right ankle, specifically around the distal fibula. There was no history of acute trauma or injury.
Associated Symptoms:
No constitutional symptoms such as fever, chills, weight loss, or night sweats were reported. There was no neurological compromise, paresthesia, or weakness. He denied any skin changes, swelling, or warmth initially, though subtle localized swelling became apparent later. The chronic pain was impacting his sleep quality, academic performance, and ability to participate in sports.
Past Medical History:
Unremarkable. No prior surgeries, significant illnesses, or allergies.
Social History:
Non-smoker, occasional alcohol use. Lives at home with parents. Attends high school.
Clinical Examination
General Inspection:
The patient appeared to be in no acute distress at rest. Gait analysis revealed a subtle antalgic limp on the right side, with decreased stance phase on the affected limb. There was no obvious gross deformity or atrophy of the musculature.
Local Inspection (Right Ankle):
*
Swelling:
Mild, localized soft tissue swelling was noted over the distal lateral malleolus.
*
Erythema/Skin Changes:
No significant erythema, warmth, or trophic changes were observed.
*
Muscle Atrophy:
No significant disuse atrophy of the surrounding musculature.
Palpation:
*
Tenderness:
Exquisite, point tenderness was elicited directly over the distal fibula, approximately 3 cm proximal to the tip of the lateral malleolus. This tenderness was highly reproducible.
*
Warmth:
Mild increase in local skin temperature compared to the contralateral ankle.
*
Crepitus:
No crepitus noted on palpation or range of motion.
Range of Motion (ROM):
*
Right Ankle:
* Dorsiflexion: 10 degrees (normal contralateral: 20 degrees) - Painful at end range.
* Plantarflexion: 35 degrees (normal contralateral: 45 degrees) - Painful at end range.
* Inversion: 10 degrees (normal contralateral: 20 degrees) - Painful.
* Eversion: 5 degrees (normal contralateral: 10 degrees) - Painful.
* Subtalar and midfoot motions were also mildly restricted and painful, likely due to guarding.
*
Contralateral Ankle:
Full and painless range of motion in all planes.
Neurological Assessment:
*
Motor Strength:
5/5 in all major muscle groups of the right lower extremity (foot dorsiflexion, plantarflexion, inversion, eversion, great toe extension, knee flexion/extension). No signs of motor weakness.
*
Sensory:
Intact light touch and pinprick sensation in all dermatomes of the right foot and ankle (L4, L5, S1, S2 distribution). No paresthesia or dysesthesia.
*
Reflexes:
Deep tendon reflexes (Patellar L4, Achilles S1) were 2+ bilaterally and symmetrical.
*
Pathological Reflexes:
Negative Babinski sign.
Vascular Assessment:
*
Pulses:
Dorsalis pedis and posterior tibial pulses were 2+ and symmetrical bilaterally. Capillary refill was brisk (<2 seconds) in all toes.
* No signs of arterial or venous insufficiency.
Special Tests:
No specific ligamentous instability was noted on anterior drawer or talar tilt tests, indicating the primary pathology was bony rather than ligamentous. Pain with a "hop test" was present, indicating weight-bearing bony pain.
Imaging & Diagnostics
Initial Radiographs (X-rays):
Anterior-posterior, lateral, and oblique views of the right ankle were obtained.
*
Findings:
The initial radiographs were subtle but revealed focal cortical thickening (sclerosis) along the lateral cortex of the distal fibula. A small, central radiolucent area, suggestive of a nidus, was faintly discernible within the sclerotic bone. There was no overt fracture line, periosteal reaction inconsistent with benign activity, or significant soft tissue calcification.
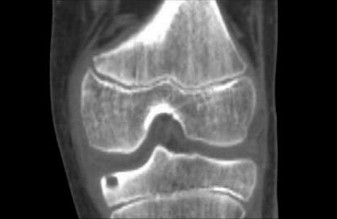
(Figure 1: Axial CT image demonstrating the characteristic radiolucent nidus surrounded by dense cortical sclerosis in the distal fibula.)
Computed Tomography (CT Scan):
A CT scan of the right ankle was performed to further characterize the lesion. CT is considered the gold standard for delineating the nidus of an osteoid osteoma.
*
Findings:
The CT scan unequivocally demonstrated a well-circumscribed, approximately 8 mm in diameter, central radiolucent nidus located within the thickened cortex of the distal fibula. The nidus exhibited a small, central focus of mineralization (sclerotic dot), characteristic of osteoid osteoma. There was extensive surrounding reactive cortical sclerosis, consistent with the chronic nature of the lesion. Crucially, the CT allowed for precise three-dimensional localization of the nidus, essential for planning any intervention.
Magnetic Resonance Imaging (MRI Scan):
An MRI of the right ankle was performed to assess the extent of marrow edema, soft tissue involvement, and to exclude other pathologies.
*
Findings:
MRI showed marked bone marrow edema surrounding the nidus in the distal fibula, reflecting the inflammatory response to the lesion. There was also a reactive periosteal edema and mild synovitis in the adjacent ankle joint, likely secondary to the inflammatory mediators released by the osteoid osteoma. While the nidus itself was less clearly visualized than on CT, the extensive edema pattern often helps narrow the differential diagnosis in conjunction with other imaging. No large soft tissue mass or clear signs of aggressive malignancy (e.g., cortical destruction with large extra-osseous extension) were noted.
Bone Scintigraphy (Technetium-99m Methylene Diphosphonate Bone Scan):
A bone scan was performed due to the chronic pain and equivocal initial X-ray.
*
Findings:
The bone scan demonstrated intense, focal uptake of the radiotracer specifically localized to the distal fibula, often described as a "double-density sign" on delayed images, indicating high metabolic activity in the region of the lesion. This high sensitivity makes bone scan useful for localizing the lesion, especially when conventional radiographs are inconclusive or the lesion is small.
Laboratory Investigations:
* Complete Blood Count (CBC): Within normal limits.
* Erythrocyte Sedimentation Rate (ESR): Within normal limits.
* C-Reactive Protein (CRP): Within normal limits.
* Alkaline Phosphatase: Within normal limits.
* All inflammatory markers were unremarkable, helping to rule out an infectious or aggressive neoplastic process.
Differential Diagnosis
The clinical presentation and initial imaging findings necessitated a thorough differential diagnosis, particularly given the chronic pain in a young, active individual.
| Feature | Osteoid Osteoma | Osteoblastoma | Brodie's Abscess (Chronic Osteomyelitis) | Stress Fracture | Enostosis (Bone Island) |
|---|---|---|---|---|---|
| Clinical Presentation | Young patient, nocturnal pain, relieved by NSAIDs, localized tenderness. | Young patient, less severe pain, less nocturnal, less NSAID relief, larger lesion. | Localized pain, possibly warmth/swelling, intermittent low-grade fever, malaise. | Active patient, pain with activity, relieved by rest, less severe nocturnal pain. | Asymptomatic, incidental finding. |
| Age Group | 10-20 years (most common) | 10-30 years (slightly older than OO) | Any age, often children/adolescents with predisposing factors (e.g., trauma). | Active individuals, athletes, military recruits (any age). | Any age. |
| Size of Lesion (Nidus) | Typically <1.5-2 cm | >2 cm, often larger than OO | Variable size, often larger than OO, with surrounding sclerosis. | Hairline fracture or periosteal reaction. | Variable, often small, dense focus. |
| Pain Characteristics | Severe, localized, worse at night , dramatically relieved by NSAIDs . | Aching, duller pain, less nocturnal, less responsive to NSAIDs. | Chronic dull ache, intermittent acute flares, less NSAID specific relief. | Activity-related, "start-up" pain, improves with rest. NSAIDs less dramatic relief. | None. |
| Radiographic Features | Small radiolucent nidus (<1.5 cm) with variable central sclerosis, surrounded by dense cortical sclerosis. | Larger radiolucent nidus (>2 cm), less reactive sclerosis, often expansile. | Radiolucent focus (abscess cavity) often with central sequestrum, thick reactive sclerosis. | Periosteal reaction, cortical thickening, endosteal callus, subtle fracture line. | Homogeneously dense, well-defined sclerotic lesion, often spiculated margins blending with trabeculae. |
| CT Findings | Diagnostic: Well-defined radiolucent nidus with central sclerotic dot, dense reactive sclerosis. | Larger nidus, often with less reactive sclerosis. | Clearly defined lucency with a rim of sclerosis, possibly sequestrum/cloaca. | Cortical thickening, medullary edema, sometimes occult fracture line. | Classic "brush border" appearance, very dense, no lucency. |
| MRI Findings | Nidus may be difficult to see directly, extensive marrow edema, periosteal reaction. | Heterogeneous signal, often less extensive marrow edema than OO. | Marrow edema, periosteal reaction, rim-enhancing fluid collection (abscess). | Linear low signal on T1/T2 (fracture line), extensive marrow edema, periosteal edema. | Hypointense on all sequences, no edema. |
| Bone Scan | Intense focal uptake ("double-density sign"). | Intense focal uptake. | Intense focal uptake. | Intense focal uptake (linear or diffuse). | May show mild uptake, but usually less intense than active lesions. |
| Histopathology | Trabeculae of osteoid and woven bone, loose vascular connective tissue, osteoblasts, osteoclasts. | Similar to OO but larger, less reactive bone, often more irregular trabeculae. | Necrotic bone, inflammatory cells (neutrophils, lymphocytes), fibrous tissue. | Reactive new bone formation, fibrous tissue. | Mature lamellar bone. |
| Treatment | NSAIDs, RFA, surgical excision. | Surgical excision (often preferred due to size/location). | Antibiotics, surgical debridement. | Rest, activity modification, bracing. | None (benign, self-limiting). |
Surgical Decision Making & Classification
The decision to intervene surgically for an osteoid osteoma is primarily driven by the persistence and severity of symptoms despite conservative management, as osteoid osteomas are benign and self-limiting, eventually regressing over several years.
Indications for Intervention in this case:
1.
Intractable Pain:
The patient's pain was severe, nocturnal, significantly impacted his quality of life, and required daily high-dose NSAIDs for symptomatic relief. This dependence on NSAIDs, with associated potential gastrointestinal and renal side effects in the long term, was a major concern.
2.
Impairment of Function:
His ability to participate in sports and daily activities was severely limited.
3.
Risk of Skeletal Deformity:
While less common in the distal fibula, osteoid osteomas in certain locations (e.g., spine, near growth plates) can lead to scoliosis or growth disturbances. For this patient, the chronic pain and altered gait could lead to compensatory musculoskeletal issues.
Classification of Osteoid Osteoma:
Osteoid osteomas are classified based on their anatomical location within the bone:
*
Cortical (80%):
Most common, typically found in the diaphysis of long bones (femur, tibia, fibula). Characterized by dense surrounding sclerosis. Our patient's lesion in the distal fibular cortex falls into this category.
*
Medullary/Cancellous (10-15%):
Found in the metaphysis or epiphysis of long bones, or in small bones. Less surrounding sclerosis, often leading to more extensive marrow edema on MRI.
*
Subperiosteal (5-10%):
Located beneath the periosteum, often without significant intraosseous sclerosis. Can be harder to detect on plain radiographs and may cause significant soft tissue reaction.
Treatment Options Considered:
- Conservative Management (NSAIDs): Attempted for 9 months. While providing temporary relief, the long-term reliance on NSAIDs was deemed unsustainable and undesirable.
- Percutaneous Radiofrequency Ablation (RFA): This is the current gold standard for the treatment of accessible osteoid osteomas due to its minimally invasive nature, high success rate, and low complication profile. It involves thermal destruction of the nidus.
- Percutaneous CT-Guided Excision/Drilling: Involves using a specialized burr or drill under CT guidance to remove the nidus. Less commonly used than RFA but can be effective.
- Open Surgical Excision: Historically the primary treatment. Involves open surgical exposure and en bloc removal of the nidus and surrounding reactive bone. Reserved for lesions that are difficult to access percutaneously (e.g., near critical neurovascular structures, joint surfaces), very large lesions, or when pathological confirmation is critical.
- Cryoablation/Laser Ablation: Other thermal ablation methods, less commonly employed for osteoid osteoma than RFA.
Decision for this case:
Given the classic clinical presentation, definitive CT findings of a solitary cortical nidus in the distal fibula, and the patient's desire for definitive treatment and return to sport,
CT-guided Radiofrequency Ablation (RFA)
was chosen as the primary intervention. This approach offered the best balance of efficacy, minimal invasiveness, and rapid recovery. Open excision was considered if RFA proved unsuccessful or if the nidus was deemed too challenging for safe percutaneous access.
Surgical Technique / Intervention
Procedure: CT-Guided Radiofrequency Ablation (RFA) of Osteoid Osteoma, Right Distal Fibula.
Pre-operative Planning:
Detailed review of the CT scan was performed to precisely localize the nidus, measure its depth from the skin, and assess its proximity to critical neurovascular structures (superficial peroneal nerve, peroneal artery/vein) and the ankle joint capsule. The optimal trajectory for probe insertion was mapped.
Anesthesia:
The procedure was performed under general anesthesia with fluoroscopic guidance in a hybrid operating room setting, allowing for patient comfort and motionless field during critical probe placement. Local anesthetic infiltration (e.g., lidocaine with epinephrine) was also used for supplementary pain control and vasoconstriction.
Patient Positioning:
The patient was positioned supine on the CT table with the right lower extremity externally rotated and draped in a sterile fashion, ensuring access to the lateral aspect of the distal fibula. A radiolucent positioning device was used to stabilize the ankle.
Surgical Approach and Nidus Localization:
1.
Initial CT Scan:
A pre-procedural diagnostic CT scan was performed to confirm the nidus location and serve as a baseline.
2.
Skin Marking:
The skin entry point and trajectory were marked based on the CT findings.
3.
Incision:
A small (5 mm) skin incision was made at the planned entry point.
4.
Trocar Insertion:
Under continuous CT fluoroscopic guidance, a bone biopsy trocar (e.g., 11-gauge coaxial needle system) was carefully advanced through the soft tissues and cortical bone, aiming directly for the center of the nidus. Sequential CT scans were performed after each advancement to verify correct trajectory and depth.
5.
Biopsy (Optional but Recommended):
Once the tip of the trocar was within the nidus, a small core biopsy specimen was obtained for histopathological confirmation, though not strictly necessary for diagnosis given the classic imaging. This step can serve as a valuable quality control and to confirm the benign nature of the lesion.
6.
Radiofrequency Probe Placement:
After biopsy, the inner stylet was removed, and the radiofrequency ablation probe (typically an 11-gauge electrode with a 5-10 mm active tip) was inserted through the outer cannula, ensuring the active tip was centered within the nidus. Final CT confirmation of probe tip position within the nidus was crucial. Probes are typically insulated except for the active tip to protect surrounding tissues.
Ablation Protocol:
1.
Temperature Monitoring:
Some RFA systems allow for real-time temperature monitoring at the probe tip, aiming for temperatures exceeding 90°C to achieve tissue necrosis.
2.
Ablation Cycle:
The RFA generator was activated, and the ablation cycle commenced. This typically involves heating the nidus for 4-6 minutes, depending on the system and nidus size. The energy delivery causes coagulative necrosis of the nidus and the pain-producing nerve fibers within it.
3.
Repeat Cycles (if necessary):
For larger nidi, or if the initial ablation appears incomplete on post-ablation imaging, additional ablation cycles or repositioning of the probe might be necessary.
4.
Cooling:
After the ablation cycle, the probe was allowed to cool before withdrawal to prevent adherence to surrounding tissues.
Post-Ablation Assessment:
A final CT scan was performed to confirm the probe's removal and to assess for any immediate complications (e.g., hematoma, fracture) and to document the immediate post-ablation changes (e.g., gas within the ablated zone).
Wound Closure:
The small skin incision was closed with a single suture or sterile adhesive strips. A sterile dressing was applied.
Potential Considerations/Challenges:
*
Proximity to Neurovascular Structures:
Careful planning and meticulous CT guidance are paramount to avoid thermal injury to nerves (e.g., superficial peroneal nerve) or vessels.
*
Proximity to Joint Surfaces:
Ablation near articular cartilage could potentially cause damage to the joint. In such cases, surgical excision or alternative strategies might be preferred.
*
Incomplete Ablation:
The primary cause of recurrence is incomplete destruction of the nidus, necessitating re-ablation.
*
Anatomical Variants:
Atypical locations or complex bone anatomy can make probe placement more challenging.
Post-Operative Protocol & Rehabilitation
The post-operative protocol following CT-guided Radiofrequency Ablation (RFA) for osteoid osteoma is typically straightforward due to the minimally invasive nature of the procedure.
Immediate Post-Operative Period (Day 0-7):
*
Pain Management:
Patients usually experience immediate and significant pain relief. Mild post-procedural discomfort at the access site and localized inflammatory pain may occur for a few days, managed with simple analgesics (e.g., acetaminophen, mild opioids as needed initially). NSAIDs are typically avoided for 24-48 hours to minimize any theoretical anti-inflammatory effect on the ablation zone, though this is not universally practiced.
*
Weight-Bearing:
Immediate weight-bearing as tolerated is permitted. Crutches may be used for comfort for 1-2 days, but full weight-bearing is generally encouraged.
*
Wound Care:
The small incision site should be kept clean and dry. Dressing changes as per standard post-operative care. Sutures or steri-strips are typically removed at 7-10 days.
*
Monitoring:
Monitor for signs of infection (redness, swelling, purulent discharge, fever), hematoma, or neurological deficits (paresthesia, weakness).
Early Rehabilitation (Week 1-4):
*
Activity:
Progressive increase in activity levels. Patients are encouraged to resume normal daily activities as comfort allows.
*
Range of Motion:
Gentle active and passive range of motion exercises for the ankle and foot are initiated to prevent stiffness.
*
Strengthening:
Light isometric exercises for surrounding musculature can begin once pain-free.
*
Return to Sport/Activity:
Low-impact activities (e.g., cycling, swimming) can be resumed within 1-2 weeks. Return to full, unrestricted sporting activities (e.g., soccer) is typically permitted at 4-6 weeks, provided the patient is pain-free and has full range of motion and strength. This is significantly faster than recovery from open surgical excision.
Long-Term Follow-up:
*
Clinical Review:
Follow-up appointments are typically scheduled at 6 weeks, 3 months, and 6 months to assess pain relief, functional recovery, and ensure no recurrence.
*
Imaging:
Repeat imaging (e.g., X-ray or CT scan) is usually not required unless symptoms recur. If pain returns, a repeat CT scan or MRI may be performed to rule out incomplete ablation or a new lesion. The reactive sclerosis around the ablated nidus can take many months to years to fully remodel and resolve.
Complications of RFA (Rare):
*
Skin Burn:
Rare, due to careful probe insulation and trajectory planning.
*
Infection:
Extremely rare due to minimal invasiveness and sterile technique.
*
Nerve Damage:
Rare, but possible if the probe is placed too close to a nerve, leading to transient or permanent neuropathy. This emphasizes the importance of precise CT guidance.
*
Vascular Injury:
Extremely rare.
*
Incomplete Ablation/Recurrence:
The most common "complication," occurring in 5-10% of cases, usually managed with repeat RFA.
*
Pathological Fracture:
Extremely rare post-ablation for OO due to the small size of the lesion and usually strong surrounding bone.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls for Diagnosis and Management:
- Clinical Triad: Always think of Osteoid Osteoma in a young patient presenting with chronic, localized bone pain, especially if it is worse at night and dramatically relieved by NSAIDs. This is the cardinal presentation.
- "NSAID Test": Administering a strong dose of NSAIDs (e.g., naproxen or ibuprofen) and observing significant relief is a powerful diagnostic indicator.
- CT is King: While X-rays can be suggestive, a CT scan is the gold standard for definitively identifying the radiolucent nidus and its characteristic central sclerotic dot. It provides precise anatomical localization crucial for intervention. MRI shows extensive edema but may not clearly visualize the nidus.
- Bone Scan Sensitivity: Bone scintigraphy is highly sensitive, showing intense focal uptake. It's excellent for localizing the approximate area of pathology when radiographs are vague, but non-specific for OO.
- RFA as First-Line: For accessible lesions, CT-guided Radiofrequency Ablation (RFA) is the preferred minimally invasive treatment due to its high success rate (80-95%), rapid recovery, and low complication profile.
- Spinal Location: Osteoid osteomas in the spine can cause painful scoliosis. Early diagnosis and treatment can correct the scoliosis in many cases.
- Subperiosteal Location: These can be harder to detect on plain films due to less reactive sclerosis. High index of suspicion with CT/MRI.
- Growth Plate Involvement: Lesions near open growth plates need careful consideration, as they can potentially cause growth disturbances if not treated appropriately or if ablation causes collateral damage.
- Pathology: If open excision is performed, histopathology shows osteoid and woven bone trabeculae lined by prominent osteoblasts, within a vascular connective tissue stroma.
Pitfalls to Avoid:
-
Misdiagnosis:
- Stress Fracture: Common in athletes, can mimic OO. However, stress fractures typically have activity-related pain, less dramatic nocturnal pain, and less profound NSAID relief. Imaging (X-ray, CT) will differentiate the pathology.
- Infection (Brodie's Abscess): Chronic osteomyelitis can cause similar pain and localized sclerosis. However, systemic inflammatory markers (ESR, CRP) are usually elevated in infection, and there may be a history of trauma or prior infection. CT findings differ.
- Osteoblastoma: Often larger than OO (>2cm), less reactive sclerosis, less nocturnal pain, and less response to NSAIDs. Management is typically surgical excision.
- Malignancy (Ewing Sarcoma, Osteosarcoma): Though rare, aggressive tumors must be ruled out, especially with atypical features, larger size, or constitutional symptoms. Careful review of imaging and potentially biopsy is critical.
- Incomplete Ablation/Recurrence: This is the most common reason for treatment failure (5-10%). It usually necessitates a repeat RFA or, less commonly, open excision. Meticulous CT guidance and ensuring the RFA probe adequately covers the entire nidus are crucial.
- Neurovascular Injury: During RFA, improper probe placement can lead to thermal injury to adjacent nerves or vessels. This is a significant risk, especially in anatomically challenging areas (e.g., posterior tibia, humeral shaft). Precise pre-operative planning and intra-operative CT guidance are paramount.
- Articular Cartilage Damage: When the nidus is subchondral, thermal injury to the joint surface is a risk, potentially leading to premature osteoarthritis. In these cases, open surgical excision or very careful controlled ablation might be considered.
- Unnecessary Open Surgery: In the past, open excision was common. Failing to recognize the suitability for RFA leads to unnecessary morbidity, longer recovery, and increased costs for the patient.
- Failure to Obtain Biopsy (if indicated): While classical imaging can be diagnostic, in atypical cases or when there is any doubt about the benign nature of the lesion, a biopsy should be obtained during the percutaneous intervention. This provides definitive histopathological diagnosis.
