Introduction and Epidemiology
Osteoid osteoma is a benign, bone-forming neoplasm characterized by a highly vascularized core of osteoblastic tissue, termed the nidus, which is typically less than 1.5 centimeters in diameter. The nidus is classically surrounded by a distinct zone of sclerotic, reactive cortical bone. While osteoid osteomas account for approximately 10% to 12% of all benign bone tumors, their presentation in the proximal femur—particularly within the intra-articular confines of the hip joint—presents unique diagnostic and therapeutic challenges for the orthopedic surgeon.
Epidemiologically, this lesion predominantly affects young adults and adolescents, with a peak incidence occurring in the second and third decades of life. There is a marked male predilection, with a male-to-female ratio approaching 2:1 or 3:1 in most epidemiological cohorts. The proximal femur is the most frequent anatomical site of involvement, accounting for nearly 30% of all cases, with the femoral neck and intertrochanteric region being the most common specific locations.
The classic clinical presentation of an extra-articular osteoid osteoma involves severe, unrelenting nocturnal pain that is characteristically relieved by nonsteroidal anti-inflammatory drugs (NSAIDs) or salicylates. This pharmacological responsiveness is mediated by the high local concentrations of prostaglandins, specifically prostaglandin E2 (PGE2) and prostacyclin, elaborated by the osteoblasts within the nidus.
However, intra-articular osteoid osteomas of the hip frequently deviate from this classic clinical and radiographic paradigm. Because the femoral neck lacks a robust periosteal layer within the joint capsule, the intense periosteal reaction and dense sclerosis seen in diaphyseal lesions are often absent or significantly blunted. Furthermore, the localized release of prostaglandins directly into the synovial fluid provokes a profound, reactive synovitis. This lymphoplasmacytic synovial infiltrate can mimic monoarticular inflammatory arthritis, septic arthritis, or transient synovitis.
Patients frequently present with referred pain to the groin, anterior thigh, or knee, accompanied by an antalgic gait, decreased range of motion, and a joint effusion. The resulting capsular distension and synovial hypertrophy can lead to secondary joint contractures and, in chronic cases, premature degenerative changes or regional osteopenia. Due to the overlapping symptomatology with femoroacetabular impingement (FAI), labral pathology, and inflammatory arthropathies, the mean delay in diagnosing an intra-articular osteoid osteoma of the hip is often reported to be between 8 and 24 months.
Historically, open surgical excision via an arthrotomy or surgical dislocation was the gold standard for definitive management. While effective, open procedures carry significant morbidity, including prolonged rehabilitation, risk of avascular necrosis (AVN), and the potential need for internal fixation. Currently, percutaneous radiofrequency ablation (RFA) is considered the first-line treatment for most extra-articular lesions. However, RFA is relatively contraindicated for intra-articular lesions located within 10 to 15 millimeters of critical neurovascular structures or articular cartilage due to the risk of irreversible thermal necrosis. Consequently, arthroscopic excision has emerged as an elegant, minimally invasive, and highly effective surgical modality for the management of intra-articular osteoid osteomas of the hip, allowing for precise nidus resection, concurrent management of reactive synovitis or secondary intra-articular pathology, and minimal disruption of the capsuloligamentous stabilizers.
Surgical Anatomy and Biomechanics
A profound understanding of the complex osseous, capsuloligamentous, and vascular anatomy of the hip joint is paramount when undertaking arthroscopic excision of a proximal femoral osteoid osteoma. The surgeon must navigate tight anatomical constraints to achieve complete tumor resection while strictly preserving the biomechanical integrity of the proximal femur and safeguarding the delicate blood supply to the femoral head.
Osseous and Capsular Anatomy
The hip is a highly constrained, multiaxial ball-and-socket joint. The proximal femur consists of the femoral head, which is covered by hyaline cartilage except at the fovea capitis, and the femoral neck, which projects superomedially and anteriorly from the femoral shaft. The normal neck-shaft angle averages 130 degrees, with approximately 10 to 15 degrees of physiological anteversion.

The joint capsule is a robust, fibrous sleeve that attaches proximally to the margins of the acetabulum, blending with the labrum, and distally to the intertrochanteric line anteriorly and the femoral neck posteriorly (approximately 1 to 1.5 cm proximal to the intertrochanteric crest). This anatomical arrangement dictates that the anterior femoral neck is entirely intra-articular, whereas the posterior femoral neck is only partially intra-articular. The capsule is reinforced by three primary ligaments: the iliofemoral ligament (Y ligament of Bigelow) anteriorly, the pubofemoral ligament inferiorly, and the ischiofemoral ligament posteriorly. During arthroscopic access to the peripheral compartment, a capsulotomy is routinely performed, typically through the iliofemoral ligament. Meticulous capsular closure is critical post-resection to restore the anterior restraint and prevent iatrogenic microinstability, particularly in young, active patients.
Vascular Anatomy of the Proximal Femur
The most critical anatomical consideration during arthroscopic resection of a femoral neck osteoid osteoma is the preservation of the extraosseous arterial ring and the ascending retinacular vessels. The primary blood supply to the adult femoral head is derived from the medial femoral circumflex artery (MFCA).

The deep branch of the MFCA courses posteriorly between the pectineus and iliopsoas, crossing the obturator externus to reach the trochanteric fossa. From the extracapsular arterial ring, the ascending cervical branches (retinacular vessels) pierce the joint capsule near its distal attachment. These vessels travel proximally along the femoral neck within the synovial retinacular folds (Weitbrecht's retinacula). The posterosuperior group (lateral epiphyseal arteries) is the most clinically significant, supplying the majority of the femoral head.
When an osteoid osteoma is located on the posterosuperior or lateral aspect of the femoral neck, aggressive burring or thermal ablation carries a high risk of damaging these terminal vessels, leading to catastrophic avascular necrosis. The arthroscopic approach provides superior illumination and magnification, allowing the surgeon to visualize the retinacular folds and meticulously dissect the overlying synovium to protect the vascular supply during nidus excavation.
Biomechanical Considerations
The proximal femur is subjected to massive compressive and tensile forces during the normal gait cycle. The medial cortex of the femoral neck (calcar femorale) primarily resists compressive loads, while the lateral cortex is subjected to significant tensile forces.
The excision of an osteoid osteoma inherently creates a cortical defect, which acts as a biomechanical stress riser. The risk of a subsequent iatrogenic femoral neck fracture is directly proportional to the size and location of the cortical defect. Biomechanical studies indicate that defects exceeding 30% of the cross-sectional diameter of the femoral neck significantly reduce the load-to-failure threshold, particularly under torsional or bending stress. Lesions located on the tension side (superior/lateral neck) are at a higher risk of propagating into a complete fracture than those on the compression side (inferior/medial neck). Preoperative planning must carefully assess the anticipated size of the defect; if extensive resection is required to eradicate the nidus, prophylactic internal fixation with cannulated screws should be performed concurrently.
Indications and Contraindications
The decision algorithm for treating an osteoid osteoma of the hip requires a multidisciplinary approach involving orthopedic surgery, musculoskeletal radiology, and pathology. While non-operative management with chronic NSAID therapy is a viable option for patients willing to tolerate long-term medication use, the natural history of spontaneous resolution can take several years. Most patients elect for surgical or interventional management due to the profound impact of nocturnal pain and functional limitation on their quality of life.
Radiofrequency ablation (RFA) under CT guidance is highly successful for diaphyseal and extra-articular lesions. However, the unique anatomy of the hip joint frequently renders RFA suboptimal or contraindicated. Arthroscopic excision is favored when the lesion's proximity to sensitive structures precludes safe thermal ablation, or when the intra-articular inflammatory milieu has generated secondary pathology requiring mechanical debridement.
Operative vs Non Operative Indications
| Modality | Primary Indications | Relative/Absolute Contraindications |
|---|---|---|
| Conservative Management | - Mild, tolerable symptoms - Patient preference to avoid surgery - Excellent response to low-dose NSAIDs - Lesions in non-weight-bearing, low-risk areas |
- Intractable pain failing medical management - Gastrointestinal/renal intolerance to NSAIDs - Progressive joint contracture or muscle atrophy - Severe reactive synovitis mimicking septic joint |
| CT-Guided RFA | - Extra-articular lesions (e.g., lesser trochanter, subtrochanteric) - Lesions >1.5 cm from articular cartilage - Lesions >1.5 cm from major neurovascular bundles - Clear radiographic nidus without secondary joint pathology |
- Intra-articular lesions <1.0 cm from hyaline cartilage (risk of chondrolysis) - Lesions adjacent to the MFCA or sciatic nerve - Presence of symptomatic cam/pincer morphology requiring concurrent treatment - Extensive reactive synovitis requiring synovectomy |
| Arthroscopic Excision | - Intra-articular lesions of the femoral neck/head or acetabulum - Lesions <1.0 cm from articular cartilage or retinacular vessels - Concomitant intra-articular pathology (labral tear, FAI, chondral defect) - Severe hypertrophic synovitis requiring debridement - Need for histological confirmation of the diagnosis |
- Extra-articular diaphyseal lesions inaccessible via hip arthroscopy - Advanced secondary osteoarthritis (Tonnis Grade 3) - Active joint infection - Medical comorbidities precluding general anesthesia or traction |
| Open Surgical Dislocation | - Massive lesions requiring extensive structural grafting - Lesions completely inaccessible arthroscopically (e.g., deep posterior neck near the lesser trochanter) - Revision cases with severe capsular scarring/distortion |
- Standard, accessible intra-articular lesions (due to unnecessary morbidity) - High risk of AVN if posterior soft tissues are extensively stripped |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is the cornerstone of a successful arthroscopic osteoid osteoma excision. The primary objective is to precisely localize the nidus, which can be challenging intraoperatively due to overlying reactive bone, thickened synovium, and the limited field of view provided by the arthroscope.
Advanced Imaging Modalities
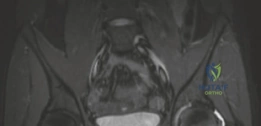
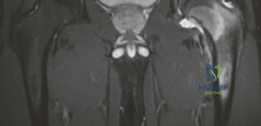
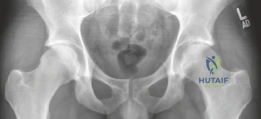
Standard anteroposterior (AP) pelvis and lateral hip radiographs are often the initial diagnostic step, but they may only show non-specific regional osteopenia or subtle cortical thickening in intra-articular cases.
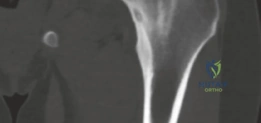
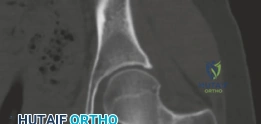
High-resolution, thin-slice computed tomography (CT) with multiplanar reconstruction is the gold standard for defining the osseous anatomy. The CT scan accurately delineates the nidus as a well-circumscribed, radiolucent focus, often containing a central fleck of mineralization, surrounded by varying degrees of sclerosis. The surgeon must meticulously map the location of the nidus relative to reliable arthroscopic landmarks, such as the medial synovial fold, the zona orbicularis, and the articular margin of the femoral head.
Magnetic resonance imaging (MRI) is highly sensitive for detecting the profound bone marrow edema and adjacent soft tissue inflammation characteristic of osteoid osteomas. While MRI may obscure the nidus itself due to the overwhelming edema signal, it is invaluable for assessing the extent of reactive synovitis, evaluating the integrity of the articular cartilage, and identifying concurrent labral pathology.
Patient Positioning and Setup
Hip arthroscopy can be performed in either the supine or the lateral decubitus position, largely dependent on surgeon preference. The supine position on a specialized fracture or traction table is the most widely utilized configuration, as it facilitates straightforward anatomical orientation and simplifies the use of intraoperative fluoroscopy.
The patient is placed supine with the perineum resting snugly against an oversized, well-padded perineal post to provide counter-traction. The operative leg is secured in a traction boot, and the contralateral leg is placed in abduction and slight extension to allow unimpeded access for the C-arm fluoroscope.

Before prepping and draping, a trial of traction is performed under fluoroscopic guidance to ensure adequate joint distraction (typically 10 to 15 millimeters) can be achieved. This distraction is essential to break the suction seal of the hip joint and allow safe entry of instruments into the central compartment without causing iatrogenic scuffing of the femoral or acetabular cartilage. The C-arm must be positioned to easily obtain true AP and cross-table lateral views of the proximal femur, as real-time fluoroscopic localization of the nidus is a critical step in the procedure.
Detailed Surgical Approach and Technique
The arthroscopic excision of an osteoid osteoma is a highly technical procedure that requires seamless integration of arthroscopic visualization, fluoroscopic targeting, and precise osseous resection.
Portal Placement and Central Compartment Access
The procedure begins with the establishment of standard arthroscopic portals under fluoroscopic guidance. The anterolateral (AL) portal is typically established first, located approximately 1 centimeter anterior and 1 centimeter superior to the tip of the greater trochanter. A spinal needle is advanced into the central compartment, and the joint capsule is punctured. The vacuum seal is broken by injecting sterile saline, and a guidewire is introduced, followed by a cannulated dilator and the arthroscope.
The mid-anterior portal (MAP) is then established under direct intra-articular visualization. This portal is located approximately 5 to 7 centimeters distal and slightly medial to the AL portal, in line with the anterior superior iliac spine (ASIS).
Diagnostic Arthroscopy and Capsulotomy
Once access to the central compartment is achieved, a systematic diagnostic arthroscopy is performed. The surgeon evaluates the acetabular labrum, the ligamentum teres, and the chondral surfaces of the acetabulum and femoral head. In cases of intra-articular osteoid osteoma, the central compartment often exhibits profound, hyperemic synovitis.

To gain access to the peripheral compartment and the femoral neck, an interportal capsulotomy is performed connecting the AL and MAP portals. Using a radiofrequency wand or arthroscopic scalpel, the capsule is incised parallel to the acetabular rim, typically extending from the 12 o'clock to the 3 o'clock position. In cases where the lesion is located very distally on the femoral neck, a T-capsulotomy may be required, extending vertically down the axis of the femoral neck.

Peripheral Compartment Access and Synovectomy
Following the capsulotomy, traction is released, and the hip is flexed to approximately 30 to 45 degrees to relax the anterior structures and expand the peripheral compartment. The arthroscope is moved to the peripheral compartment.
The surgeon will immediately encounter the reactive synovitis, which can be thick, villous, and highly vascular. A thorough synovectomy is performed using an arthroscopic shaver and radiofrequency wand. This step is critical not only for alleviating the patient's inflammatory symptoms but also for exposing the underlying bony architecture of the femoral neck. Meticulous care must be taken during the synovectomy along the posterosuperior and posteroinferior aspects of the neck to avoid injuring the ascending retinacular vessels.
Nidus Localization and Excision
Locating the nidus is the most challenging aspect of the procedure. The overlying cortex may appear entirely normal, or it may exhibit subtle neovascularization, a localized hyperemic blush, or a slight cortical prominence.
The surgeon uses a combination of preoperative CT mapping, direct arthroscopic visualization, and intraoperative fluoroscopy to pinpoint the lesion. A spinal needle or a Kirschner wire is introduced percutaneously and advanced to the suspected location on the femoral neck. AP and lateral fluoroscopic views are obtained to confirm the trajectory and depth relative to the radiolucent nidus.

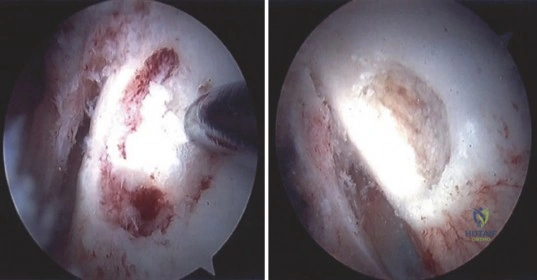
Once the location is confirmed, a high-speed arthroscopic burr is used to carefully unroof the overlying cortex. As the cortex is breached, the nidus typically appears as a distinct, hyperemic, cherry-red, granular tissue mass that contrasts sharply with the surrounding white, sclerotic bone.
Curettes and pituitary rongeurs are utilized to meticulously excavate the nidus. It is imperative to obtain adequate tissue samples for histopathological analysis to definitively confirm the diagnosis of osteoid osteoma and rule out malignancy or infection. Following the removal of the gross nidus, the high-speed burr is reintroduced to extend the resection margin by 1 to 2 millimeters into the surrounding reactive bone, ensuring complete eradication of the tumor and minimizing the risk of recurrence.

Fluoroscopic Confirmation and Closure
Following excision, the cavity is thoroughly irrigated to remove all osseous debris. Intraoperative fluoroscopy is repeated to confirm complete removal of the radiolucent nidus and to assess the size of the resulting cortical defect.
If the defect is small and located on the compressive side of the neck, no further intervention is required. However, if the resection violates more than 30% of the neck diameter, or if it is located on the superior tension side, the surgeon must strongly consider prophylactic internal fixation. This is typically achieved percutaneously using two or three parallel 6.5 mm or 7.3 mm cannulated screws placed over guidewires under fluoroscopic control.
Finally, the joint is thoroughly lavaged. The capsulotomy is meticulously closed using heavy, non-absorbable sutures passed through the capsular leaflets. Capsular closure is essential to restore the normal biomechanical restraints of the hip, prevent iatrogenic microinstability, and optimize the healing environment.
Complications and Management
While arthroscopic excision of an osteoid osteoma is generally safe and highly effective, the complex anatomy of the hip joint and the necessary osseous resection inherently carry significant risks. The surgeon must be acutely aware of potential complications and possess the technical acumen to manage them effectively.
Iatrogenic Femoral Neck Fracture
The most devastating complication following excision of a proximal femoral lesion is an iatrogenic fracture of the femoral neck. The cortical defect created by the burr acts as a stress riser. If the patient bears full weight prematurely, or if the initial resection was overly aggressive, the defect can propagate into a complete, displaced fracture.
Management of a postoperative femoral neck fracture in a young patient is an orthopedic emergency. Anatomic reduction and rigid internal fixation (typically with a dynamic hip screw or multiple cannulated screws) must be performed urgently to preserve the viability of the femoral head. To mitigate this risk, meticulous preoperative planning is essential to estimate the required resection volume, and prophylactic fixation should be employed liberally for large or tension-sided defects.
Avascular Necrosis (AVN)
Avascular necrosis of the femoral head can occur secondary to direct mechanical injury to the ascending retinacular vessels during synovectomy or burring, or due to thermal necrosis if radiofrequency ablation is used inappropriately close to the vascular supply.
AVN presents with progressive groin pain and mechanical symptoms weeks to months after the index procedure. Early-stage AVN may be managed with core decompression or bisphosphonate therapy, but advanced collapse inevitably requires total hip arthroplasty. Prevention is paramount; the surgeon must maintain strict anatomical orientation, utilize meticulous dissection techniques, and avoid indiscriminate use of thermal devices near the posterosuperior femoral neck.
Recurrence and Incomplete Excision
Failure to completely excise the entire nidus will result in the recurrence of the classic nocturnal pain, often within weeks of the surgery. Incomplete excision is usually secondary to inaccurate intraoperative localization or timidity during the burring phase.
If recurrence is suspected, a repeat thin-slice CT scan is indicated to identify the residual nidus. Revision surgery is technically demanding due to altered landmarks and postoperative scarring. CT-guided RFA may be considered as a salvage option for recurrences if the anatomical constraints allow, otherwise, a revision arthroscopic or open procedure is necessary.
Summary of Complications
| Complication | Estimated Incidence | Etiology/Risk Factors | Prevention and Salvage Strategies |
|---|---|---|---|
| Femoral Neck Fracture | 1% - 3% | Excessive cortical resection (>30% diameter); Tension-sided defects; Premature weight-bearing | Prevention: Prophylactic cannulated screw fixation for large defects; Strict adherence to post-op weight-bearing restrictions. Salvage: Urgent open reduction and internal fixation (ORIF). |
| Avascular Necrosis (AVN) | < 1% | Injury to MFCA retinacular branches; Thermal necrosis from RFA/wand | Prevention: Meticulous dissection of posterosuperior neck; Avoid thermal devices near retinacula. Salvage: Core decompression (early stage); Total Hip Arthroplasty (late stage collapse). |
| Incomplete Excision / Recurrence | 4% - 7% | Inaccurate localization; Failure to burr 1-2mm beyond the nidus margin | Prevention: Rigorous pre-op CT mapping; Real-time intra-op fluoroscopic targeting; Pathological confirmation of nidus. Salvage: Repeat CT to define residual nidus; Revision arthroscopic excision or CT-guided RFA. |
| Iatrogenic Chondral Injury | 1% - 2% | Inadequate joint distraction during portal placement; Instrument crowding | Prevention: Ensure >10mm distraction before establishing central portals; Avoid levering instruments against cartilage. Salvage: Chondroplasty; Microfracture for full-thickness defects. |
| Pudendal/Sciatic Neurapraxia | 2% - 5% | Prolonged traction time; Excessive traction force; Inadequate perineal padding | Prevention: Limit traction time to < 2 hours; Use well-padded, oversized perineal post; Release traction during peripheral compartment work. Salvage: Usually transient, resolving with observation and supportive care. |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation protocol following arthroscopic excision of an osteoid osteoma is highly individualized, dictated primarily by the size and location of the osseous defect, the integrity of the capsular closure, and whether prophylactic internal fixation was utilized. The overarching goal is to protect the structural integrity of the proximal femur while restoring normal joint kinematics and preventing capsular adhesions.
Phase 1: Protection and Early Mobility (Weeks 0-4)
The immediate postoperative phase prioritizes the protection of the healing cortical bone and the repaired joint capsule.
* Weight-Bearing: If the cortical defect is small (<10-15% of neck diameter) and located on the compressive side, patients may be allowed touch-down weight-bearing (TDWB) or partial weight-bearing (PWB) with crutches immediately. If a larger defect is present without prophylactic fixation, strict non-weight-bearing (NWB) status is maintained for 4 to 6 weeks.
* Range of Motion (ROM): Early, passive ROM is initiated to prevent intra-articular adhesions. A continuous passive motion (CPM) machine or stationary bicycle (with zero resistance) is often utilized. Extreme external rotation and extension are avoided to protect the anterior capsular closure.
* Strengthening: Isometric exercises for the quadriceps, hamstrings, and gluteal musculature are initiated immediately.
Phase 2: Restoration of Kinematics and Progressive Loading (Weeks 4-8)
As radiographic evidence of early bone healing emerges and soft tissue healing progresses, the rehabilitation focus shifts to restoring normal gait and progressive strengthening.
* Weight-Bearing: Patients are gradually transitioned to full weight-bearing (FWB) over a 1- to 2-week period, weaning off crutches as pain and gait mechanics allow.
* Range of Motion: Active and active-assisted ROM exercises are progressed to achieve full, symmetric mobility.
* Strengthening: Closed kinetic chain exercises (e.g., mini-squats, leg presses) are introduced. Core stabilization and abductor strengthening are emphasized to normalize pelvic mechanics and prevent Trendelenburg gait.

Phase 3: Advanced Strengthening and Return to Sport (Weeks 8-16+)
The final phase aims to return the patient to their pre-injury level of activity, including high-impact sports or heavy manual labor.
* Clearance: Progression to Phase 3 requires a pain-free normal gait, full ROM, and symmetric muscle strength. Radiographic confirmation of progressive consolidation of the cortical defect is highly recommended before initiating impact activities.
* Activities: Plyometrics, agility drills, and sport-specific training are gradually integrated.
* Outcomes: The vast majority of patients experience dramatic and immediate relief of their preoperative nocturnal pain
