Patient Presentation & History
A 62-year-old female presented to the emergency department via ambulance after a low-energy fall at home, reporting acute onset of severe left thigh pain and inability to bear weight. The mechanism of injury was described as a simple trip over a rug, resulting in a rotational force on the left lower extremity followed by a collapse to the ground. She denied any direct impact or significant height from which she fell.
History of Present Illness:
Prior to the fall, the patient reported a 3-4 month history of insidious, dull aching pain in her left proximal thigh, which had been progressively worsening. Initially, the pain was intermittent and activity-related, partially relieved by rest and over-the-counter analgesics. Over the last month, the pain had become more constant, interfering with sleep and requiring stronger analgesics. She attributed this progressive discomfort to "arthritis" or "old age." The acute episode was characterized by a sudden, sharp, excruciating pain that immediately rendered her non-ambulatory.
Past Medical History:
The patient has a significant oncological history. She was diagnosed with Stage IV Estrogen Receptor (ER) positive, Progesterone Receptor (PR) positive, HER2-negative Invasive Ductal Carcinoma of the breast three years prior. Her initial presentation involved axillary lymph node involvement, and subsequent staging revealed metastatic disease to the lumbar spine and pelvic bones. She has been on systemic therapy, including hormonal therapy (anastrozole) and a CDK4/6 inhibitor (palbociclib), along with a bone-modifying agent (denosumab) for the past two years. She underwent palliative radiation therapy to the L3 vertebra one year ago for symptomatic spinal metastases. She denies any new focal neurological deficits prior to her fall.
Current Medications:
* Anastrozole 1mg daily
* Palbociclib 125mg daily (21 days on, 7 days off cycle)
* Denosumab 120mg subcutaneous every 4 weeks
* Tramadol 50mg TID PRN for pain
* Acetaminophen 1000mg TID PRN for pain
* Pantoprazole 40mg daily
Allergies: No known drug allergies (NKDA).
Social History:
She is a former smoker (quit 15 years ago, 20 pack-year history) and an occasional social alcohol consumer. She lives alone in a single-story home and was previously fully independent with all activities of daily living (ADLs). Her reported Karnofsky Performance Status prior to the acute injury was estimated at 70% (able to care for self, unable to carry on normal activity or do active work).
Review of Systems (relevant findings):
*
Constitutional:
Generalized fatigue, mild unintentional weight loss of approximately 3 kg over the last 6 months.
*
Musculoskeletal:
Chronic generalized body aches.
*
Oncologic:
No new breast lumps or skin changes.
*
Other:
Otherwise negative for acute cardiopulmonary, gastrointestinal, or genitourinary symptoms. No fever or chills.
Clinical Examination
Upon presentation, the patient was alert and oriented, appearing acutely distressed due to severe pain in her left thigh.
General Inspection:
The patient was lying supine on the stretcher. Her left lower extremity was noticeably shortened and held in an externally rotated posture compared to the contralateral limb. There was visible swelling and ecchymosis extending from the proximal thigh to the mid-femoral region. No open wounds, skin tenting, or signs of impending compartment syndrome were observed.
Palpation:
Exquisite tenderness was elicited upon gentle palpation over the entire proximal and mid-femur, particularly prominent around the subtrochanteric region. Palpation also revealed gross instability and crepitus with even minimal attempts at passive movement of the thigh, consistent with a femoral shaft fracture. The overlying skin was warm but not erythematous.
Range of Motion:
Active hip and knee range of motion of the left leg was severely limited and provoked unbearable pain, rendering objective measurement impossible. Passive range of motion was similarly restricted and elicited significant guarding and discomfort. The contralateral hip and knee had full, pain-free range of motion.
Neurological Assessment:
Detailed neurological assessment of the left lower extremity was performed distal to the suspected fracture site.
*
Motor Strength:
Intact motor strength (5/5 MRC scale) in ankle dorsiflexion (L4), ankle plantarflexion (S1), great toe extension (L5), and knee extension/flexion (L3/L4, S1/S2). No foot drop or other gross motor deficits were detected.
*
Sensation:
Intact sensation to light touch and pinprick in all dermatomes distal to the knee (L2-S1), specifically over the patellar region (L3), medial malleolus (L4), dorsum of foot (L5), and lateral foot/plantar aspect (S1).
*
Reflexes:
Patellar reflex (L3-L4) and Achilles reflex (S1) were bilaterally present and symmetrical (2+).
*
Peripheral Nerves:
No signs of femoral, sciatic, or peroneal nerve impingement or injury were identified.
Vascular Assessment:
* Bilateral dorsalis pedis and posterior tibial pulses were strong and symmetrical (2+).
* Capillary refill in the toes of the left foot was brisk, less than 2 seconds.
* The limb was warm and well perfused. No signs of acute arterial compromise were noted.
Systemic Examination:
*
Cardiac:
Regular rate and rhythm, no murmurs.
*
Pulmonary:
Clear to auscultation bilaterally, no respiratory distress.
*
Abdominal:
Soft, non-tender, non-distended, no hepatosplenomegaly.
*
Skin:
No new suspicious lesions.
Overall, the clinical picture was highly suggestive of a displaced, unstable pathological fracture of the proximal femur, necessitating urgent orthopedic stabilization.
Imaging & Diagnostics
Following the initial clinical assessment, a comprehensive imaging and diagnostic workup was initiated to confirm the diagnosis, characterize the extent of the lesion, assess for other systemic involvement, and facilitate surgical planning.
1. Plain Radiographs (X-rays):
Anterior-posterior (AP) and lateral views of the entire left femur, including the hip and knee joints, were obtained immediately.
*
Findings:
The radiographs clearly demonstrated a highly comminuted and displaced subtrochanteric fracture of the left femur. The fracture involved the lesser trochanter and extended distally into the proximal diaphysis. Crucially, an underlying, expansile lytic lesion was evident at the fracture site, characterized by significant cortical thinning and destruction, with a permeative appearance consistent with aggressive bone erosion. The lesion appeared to involve more than 50% of the cortical circumference. There was evidence of extensive endosteal scalloping and disruption of the trabecular architecture. Multiple scattered, less defined lytic and mixed (lytic/sclerotic) lesions were also noted in the ipsilateral femoral shaft, pelvic brim, and sacrum, indicative of widespread metastatic disease.
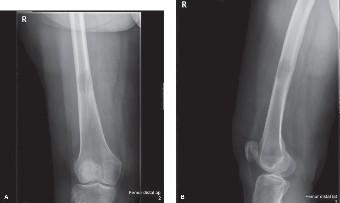
*
Significance:
Plain films confirmed the diagnosis of a pathological fracture and provided initial insight into the tumor's destructive nature and extent of bone involvement. They are the initial cornerstone of diagnosis.
2. Computed Tomography (CT) Scan:
A CT scan of the left femur with coronal, sagittal, and 3D reconstructions was performed. This was followed by a CT of the chest, abdomen, and pelvis to complete the staging and rule out new visceral metastases.
*
Findings (Femur):
The CT scan provided exquisite detail of the fracture morphology, confirming the comminution and displacement. It precisely delineated the extent of cortical bone destruction (over 70% circumferentially at the subtrochanteric region) and the intramedullary component of the lytic lesion. Soft tissue involvement was limited to hematoma surrounding the fracture, without evidence of a large extraosseous tumor mass. The exact dimensions of the intramedullary canal were measured, crucial for intramedullary nail templating. The scan also confirmed other smaller, non-fractured lytic lesions within the proximal femur and acetabulum.
*
Significance:
CT is invaluable for pre-operative planning, allowing precise measurement for hardware selection (nail diameter, length, screw trajectory), assessing the integrity of surrounding bone, and identifying areas requiring cement augmentation. It helps in evaluating the quality of bone for distal locking screw purchase.
3. Magnetic Resonance Imaging (MRI):
An MRI of the left femur was considered but deferred in this acute setting, primarily due to the clear radiographic and CT findings confirming a pathological fracture in a patient with a known metastatic primary.
*
Indications (if performed):
MRI would be indicated if the primary diagnosis was unclear, if extensive soft tissue tumor involvement was suspected, or if precise intramedullary and skip lesion assessment was needed for a potential en bloc resection (less likely in this palliative setting). It excels in delineating soft tissue extent, marrow involvement, and neurovascular bundle relationship.
4. Biopsy:
Given the patient's known history of metastatic breast cancer and the classic radiographic appearance, an open intraoperative biopsy was planned at the time of surgical fixation. This approach minimizes additional procedures and provides definitive histological confirmation.
*
Procedure:
During surgery, a curette sample of the tumor tissue from the fracture site was obtained and sent for immediate frozen section and subsequent permanent histopathological analysis, along with immunohistochemistry.
*
Pathology Report (post-op):
Confirmed metastatic adenocarcinoma, consistent with the known breast primary (positive for ER, PR, negative for HER2, GATA3 positive). This provided definitive proof of the metastatic nature of the lesion.
5. Systemic Staging / Surveillance:
*
Positron Emission Tomography-Computed Tomography (PET-CT):
A whole-body PET-CT was performed post-stabilization to assess the overall metastatic disease burden.
*
Findings:
Revealed multiple metabolically active lesions in the axial skeleton (vertebrae, ribs, pelvis), the previously identified femoral lesion, and several new small pulmonary nodules, indicating progressive systemic disease. No new brain metastases were identified.
*
Significance:
This comprehensive staging guides systemic oncologic treatment decisions and helps identify other impending fracture sites.
*
Bone Scintigraphy (Technetium-99m):
While a PET-CT was chosen for its comprehensive nature, a bone scan could also have been used to screen for widespread skeletal metastases, particularly blastic lesions.
6. Laboratory Investigations:
Preoperative blood tests were crucial for assessing systemic health, identifying complications of malignancy, and preparing for surgery.
*
Complete Blood Count (CBC):
Mild anemia (Hb 10.2 g/dL), mild leukocytosis (WBC 11.5 x 10^9/L) likely related to stress/inflammation, normal platelet count.
*
Basic Metabolic Panel (BMP):
Serum calcium was elevated at 11.8 mg/dL (normal 8.5-10.2 mg/dL), consistent with hypercalcemia of malignancy. Renal function was within normal limits.
*
Liver Function Tests (LFTs):
Mildly elevated alkaline phosphatase (ALP 180 U/L) suggesting increased bone turnover, otherwise normal.
*
Coagulation Profile (PT/INR, PTT):
Within normal limits, indicating no significant coagulopathy.
*
Tumor Markers:
CA 15-3 was elevated, reflecting known disease progression.
*
Significance:
Hypercalcemia required immediate medical management (IV fluids, bisphosphonates). Normal coagulation profile allowed for safe surgical intervention. These tests provide a snapshot of the patient's physiological status and guide perioperative management.
Differential Diagnosis
When encountering a pathological fracture in a long bone, particularly in an adult, a broad differential diagnosis must be considered. While the patient's known history of metastatic breast cancer heavily weighted the diagnosis, a systematic approach is essential for board examinations and for situations where the primary malignancy is unknown or when unusual features are present.
Here, we compare three key differential diagnoses for an aggressive, lytic bone lesion leading to a pathological fracture of the femur:
| Feature | Pathologic Fracture (Metastatic Carcinoma) | Primary Malignant Bone Tumor (e.g., High-Grade Sarcoma) | Multiple Myeloma / Solitary Plasmacytoma |
|---|---|---|---|
| Patient Demographics | Typically older adults (>40-50 years), strong association with known primary cancer (breast, lung, prostate, kidney, thyroid). | Can occur at any age, but certain types are more common in younger patients (osteosarcoma, Ewing's sarcoma) or older adults (chondrosarcoma, fibrosarcoma). | Predominantly older adults (>60 years). |
| Clinical Presentation | Insidious onset of dull, aching bone pain, gradually worsening over weeks to months, often nocturnal. Minor trauma precipitates acute fracture. Systemic symptoms related to primary cancer (fatigue, weight loss). | Localized pain (often severe), palpable soft tissue mass, rapid growth, tenderness, warmth, acute fracture. Less commonly systemic symptoms initially. | Diffuse bone pain, fatigue, anemia, recurrent infections, renal insufficiency, hypercalcemia. Pathologic fractures are a hallmark. |
| Radiographic Appearance | Classic: Predominantly lytic (e.g., kidney, thyroid, lung, melanoma), blastic (prostate, breast sometimes), or mixed. Cortical destruction, permeative pattern, endosteal scalloping. Often multifocal. Minimal or non-specific periosteal reaction. | Aggressive: Ill-defined, wide zone of transition. Cortical destruction. Associated soft tissue mass. Characteristic periosteal reactions (sunburst, Codman's triangle, onion skin) depending on type. Usually solitary lesion. | "Punched out" purely lytic lesions without sclerotic borders or periosteal reaction. Often multifocal (skull, spine, pelvis). Diffuse osteopenia. |
| CT Scan Findings | Precise cortical destruction and intramedullary extension. Evaluation of soft tissue component and relationship to neurovascular structures. Multi-focal lesions. | Detailed bone destruction, internal matrix (osteoid/chondroid), large soft tissue component, periosteal reaction. | Bone marrow infiltration, diffuse osteopenia, punched-out lytic lesions. Absence of osteoblastic activity. |
| MRI Findings | Marrow replacement, soft tissue extension (variable). T1 hypointense, T2 hyperintense. May show skip lesions. | Extensive marrow infiltration, large soft tissue mass, precise tumor margins (often ill-defined on T1/T2), neurovascular involvement. | Diffuse marrow signal changes (low T1, high T2), focal plasmacytomas, involvement of multiple sites. |
| Systemic Symptoms | Common: hypercalcemia, fatigue, weight loss, anorexia. Symptoms related to primary site (e.g., dyspnea in lung mets). | Less common until advanced disease: fever, weight loss, malaise. | Very common: anemia (normocytic), renal failure, hypercalcemia, amyloidosis, recurrent infections. |
| Laboratory Markers | Elevated Alkaline Phosphatase (ALP), hypercalcemia (especially with lytic lesions), anemia of chronic disease. Specific tumor markers (e.g., CA 15-3 for breast, PSA for prostate, CEA for colorectal). | Elevated ALP (osteosarcoma), ESR, CRP. May have normal calcium. Lactic dehydrogenase (LDH) sometimes elevated. | M-spike (monoclonal protein) on serum/urine protein electrophoresis, elevated total protein, abnormal serum free light chain ratio, hypercalcemia, elevated beta-2 microglobulin, renal insufficiency. |
| Biopsy Findings | Carcinoma cells, often glandular or pleomorphic. Immunohistochemistry (IHC) panel guides primary site identification (e.g., ER/PR/GATA3 for breast, TTF-1/Napsin A for lung, PSA for prostate). | Malignant cells with specific features depending on type: osteoid production (osteosarcoma), chondroid matrix (chondrosarcoma), small round blue cells (Ewing's). High mitotic activity. | Monoclonal proliferation of plasma cells (>10% marrow plasma cells or presence of plasmacytoma), specific kappa/lambda light chain restriction. |
| Treatment Goal & Prognosis | Palliation, pain control, functional preservation, prevention of further SREs. Prognosis dependent on primary tumor biology and extent of metastases. | Curative intent with neoadjuvant/adjuvant chemotherapy, wide surgical resection, radiation. Prognosis varies with grade and stage. | Systemic therapy (chemotherapy, novel agents, stem cell transplant), supportive care for bone disease (bisphosphonates). Prognosis variable. |
In our patient's case, the strong history of Stage IV breast cancer with known bone metastases, coupled with the characteristic lytic radiographic appearance, significantly narrowed the differential diagnosis, making metastatic carcinoma the most probable diagnosis. However, a biopsy is always essential for definitive confirmation, especially if any atypical features are present or if the primary source is not previously known. Considering a new primary or transformation of the existing cancer is also prudent.
Surgical Decision Making & Classification
The decision to proceed with surgical intervention for a pathological femoral fracture, particularly in the setting of metastatic bone disease, is a multifaceted one, balancing the patient's functional needs, quality of life, estimated life expectancy, and the inherent risks of surgery.
Rationale for Operative Intervention:
For our patient, the operative approach was strongly indicated for several reasons:
1.
Unstable Fracture:
The comminuted subtrochanteric fracture rendered the left femur mechanically unstable, precluding weight-bearing and causing severe, unmanageable pain.
2.
Pain Relief:
Surgical stabilization provides immediate and significant pain relief by immobilizing the fracture fragments, improving the patient's quality of life.
3.
Functional Restoration:
Operative fixation allows for early mobilization and weight-bearing, facilitating rehabilitation, preventing complications of immobility (e.g., pneumonia, DVT, pressure ulcers), and maintaining independence.
4.
Local Tumor Control:
While not curative, surgical debridement and cement augmentation contribute to local tumor control and provide a stable construct for subsequent adjuvant therapies.
5.
Quality of Life:
The overarching goal in metastatic disease management is to maximize the patient's quality of life. An immobile, painful fracture severely impacts this.
Contraindications for Operative Intervention:
While strongly indicated, surgery may be contraindicated in certain scenarios:
*
Extremely Short Life Expectancy:
Patients with a predicted life expectancy of less than 1 month, where the morbidity of surgery outweighs potential benefits.
*
Prohibitive Medical Comorbidities:
Uncontrolled sepsis, severe coagulopathy that cannot be corrected, unstable cardiac or pulmonary status precluding safe anesthesia.
*
Poor Performance Status:
Karnofsky Performance Status < 40% (bed-ridden, requires extensive care) may suggest that the patient cannot tolerate surgery or benefit from rehabilitation.
*
Non-displaced, Stable Lesions:
In very select cases, non-displaced fractures or impending fractures in non-weight-bearing bones may be managed with radiation therapy alone. This was not the case here.
Surgical Goal:
The primary surgical goal was
palliation
– to provide immediate pain relief, restore mechanical stability, facilitate early mobilization, and improve the patient's overall quality of life. The aim was not curative resection, but durable fixation.
Classification Systems Used in Decision Making:
-
Mirels' Scoring System for Impending Pathological Fractures:
Although our patient presented with an actual fracture, the Mirels' score is retrospectively useful to understand the fracture risk.- Site: Peritrochanteric (score 3)
- Pain: Functional (score 3) – prior to acute fracture, pain limited activity.
- Lesion Type: Lytic (score 3)
- Size: >2/3 cortical destruction (score 3)
-
Total Mirels' Score:
3 + 3 + 3 + 3 =
12
.
A score of 7 or higher indicates a high risk of impending fracture and prophylactic fixation is often recommended. A score of 12 clearly demonstrates the high probability of fracture.
-
Tokuhashi Score (Modified):
This scoring system helps estimate a patient's postoperative survival, which in turn guides the aggressiveness and type of surgical reconstruction.- Karnofsky Performance Status: 70% (score 1)
- Number of Bone Metastases: Multiple (score 1) – based on skeletal survey
- Visceral Metastases: Present (score 0) – new pulmonary nodules on PET-CT
- Primary Tumor Site: Breast (score 2) – generally better prognosis than lung/stomach.
- Paralysis: None (score 2)
-
Score Calculation:
1 (KPS) + 1 (Bone Mets) + 0 (Visceral Mets) + 2 (Primary Breast) + 2 (No Paralysis) =
6
.
A modified Tokuhashi score of 6 suggests a relatively poorer prognosis (<6 months). This score guided the choice towards a robust, yet less extensive and less morbid, stabilization technique (intramedullary nailing) rather than a complex endoprosthetic reconstruction, which might be considered for a longer life expectancy (e.g., >1 year).
-
ECOG (Eastern Cooperative Oncology Group) Performance Status:
This system categorizes functional impairment more broadly. Our patient's pre-fracture status was ECOG 1 (ambulatory, restricted in physically strenuous activity). Post-fracture, she would temporarily be ECOG 3-4. Surgical intervention aims to restore her to ECOG 1-2.
Choice of Implant and Strategy:
Given the subtrochanteric location, the extent of lytic destruction, and the Tokuhashi score suggesting a shorter life expectancy, a
long cephalomedullary intramedullary nail
was selected.
*
Advantages of IMN:
*
Load Sharing:
IMN provides excellent axial load sharing, which is crucial in weakened bone, reducing stress shielding and improving durability.
*
Minimally Invasive:
Smaller incisions compared to plating, potentially less blood loss, and preserved periosteal blood supply.
*
Biomechanical Stability:
Effectively neutralizes bending, rotation, and shear forces across the fracture.
*
Early Mobilization:
Allows for immediate or early weight-bearing, facilitating aggressive rehabilitation.
*
Cement Augmentation:
The intramedullary canal provides a contained space for polymethylmethacrylate (PMMA) cement augmentation, which significantly enhances stability, fills voids, and provides local tumor control via exothermic reaction.
*
Bypassing Lesions:
A long nail can bypass the entire tumor-involved segment, providing durable fixation through healthy bone proximal and distal to the lesion, and proactively reinforcing other at-risk lesions in the same bone.
This decision-making process emphasizes a holistic, patient-centered approach, integrating oncological prognosis with orthopedic principles to achieve the best possible functional outcome and quality of life for a patient with advanced metastatic bone disease.
Surgical Technique / Intervention
The surgical intervention for this patient involved an antegrade long cephalomedullary intramedullary nailing of the left femur, augmented with polymethylmethacrylate (PMMA) cement, following thorough pre-operative planning.
Pre-operative Planning:
1.
Imaging Review:
Detailed review of AP/Lateral X-rays and CT scans of the left femur. The CT scans were critical for assessing the extent of cortical breach, the size of the intramedullary lesion, the diameter of the femoral canal at the isthmus and distal to the lesion, and the exact fracture morphology.
2.
Templating:
Digital templating was performed to select the appropriate nail length (bypassing the entire lytic lesion and extending into healthy distal femur), nail diameter, and determine the optimal entry point (piriformis fossa vs. greater trochanter). We aimed for a nail that would span the entire femur distally, reaching within 2-3 cm of the femoral condyles, to protect against potential future distal femoral lesions.
3.
Blood Management:
Given the potential for increased blood loss due to the hypervascular nature of some metastatic lesions and the bone destruction, appropriate blood cross-matching and availability were ensured.
4.
Team Communication:
A multidisciplinary meeting involved orthopedic trauma, orthopedic oncology, and anesthesia to review the case and optimize patient care.
Patient Positioning & Anesthesia:
The patient was placed supine on a fracture table. The left leg was placed in gentle traction, facilitating reduction and access, with the hip flexed to approximately 15 degrees and adducted to allow for a straight-line approach to the piriformis fossa entry point. The right leg was abducted and flexed to provide access for image intensifier C-arm placement. General anesthesia was administered, and a single dose of broad-spectrum prophylactic antibiotics (e.g., Cefazolin) was given intravenously pre-incision.
Surgical Approach:
1.
Preparation and Draping:
The left lower extremity was sterilely prepped from the mid-abdomen to the foot and draped to allow full visualization of the hip and entire femur.
2.
Incision and Entry Point:
A curvilinear incision approximately 5-7 cm long was made over the tip of the greater trochanter, extending proximally along the gluteal ridge. The gluteus medius and minimus were split longitudinally in line with their fibers, or partially released, to expose the piriformis fossa. The piriformis fossa was chosen as the entry point for its biomechanical advantages in subtrochanteric fractures, aligning the nail with the true axis of the femur. A guidewire was then advanced under fluoroscopic guidance to confirm the ideal entry point.
Surgical Technique (Antegrade Long Cephalomedullary Nailing with Cement Augmentation):
1.
Guidewire Placement:
A cannulated awl was used to open the entry portal. A flexible guidewire was then carefully advanced down the femoral canal, across the comminuted subtrochanteric fracture site, and into the distal metaphysis. Fluoroscopy (AP and lateral views) confirmed optimal guidewire position.
2.
Reaming:
Sequential flexible reamers were used to ream the femoral canal over the guidewire. Reaming was performed meticulously through the entire length of the intended nail insertion, including the lytic tumor site. The canal was reamed to 1.5mm-2mm larger than the planned nail diameter to facilitate cement injection and nail passage. Reaming served a dual purpose: preparing the canal for the nail and debriding some intralesional tumor tissue.
3.
Tumor Curettage and Biopsy:
Through the fracture site, which was easily accessible with retraction, extensive curettage of the lytic tumor tissue was performed using specialized curettes. Gross tumor was meticulously removed from the defect. Tissue samples were collected and sent for intraoperative frozen section and definitive histopathological analysis to confirm the diagnosis of metastatic adenocarcinoma.
4.
Cement Augmentation (PMMA):
This was a critical step. With the fracture site reduced and held with cerclage wires (if needed, though often the guidewire and gentle traction suffice for temporary alignment), PMMA bone cement was mixed. Once the cement reached a doughy consistency (allowing for injectability but preventing excessive extravasation), it was injected directly into the lytic defect and surrounding compromised bone using a cement gun and catheter. The goal was to fill the cavity comprehensively and create a robust mantle around the nail path within the tumor zone. Approximately 20-30 mL of cement was used.
5.
Nail Insertion:
While the cement was partially polymerizing, the chosen long cephalomedullary nail (e.g., a Gamma 3 or similar design) was carefully inserted over the guidewire, traversing the cement-filled defect. Gentle rotational and translational movements were used to guide the nail past the fracture and through the cement. The cement provides immediate structural support and enhances the mechanical interlock between the nail and the weakened bone.
6.
Proximal Locking:
Once the nail was seated to the appropriate depth (confirmed by fluoroscopy for proper cephalic screw placement), the proximal lag screw(s) were inserted into the femoral head and neck. This provides critical angular stability to the proximal fragment.
7.
Reduction Confirmation:
Throughout the process, fracture reduction, length, and rotation were continuously monitored using fluoroscopy. The traction and rotational positioning on the fracture table were adjusted as needed.
8.
Distal Locking:
Under fluoroscopic guidance, distal locking screws were placed in two planes to prevent rotation and shortening. It was ensured that these screws were placed in healthy cortical bone distal to the tumor burden.
9.
Wound Closure:
The surgical site was thoroughly irrigated with copious saline. Meticulous hemostasis was achieved. A small suction drain was placed in the deep tissues if significant bleeding was anticipated, though often not required. The fascia, subcutaneous tissue, and skin were closed in layers. A sterile dressing was applied.
Post-operative Radiographs:
Immediately post-operatively, AP and lateral radiographs of the entire left femur were obtained in the recovery room to confirm satisfactory nail position, fracture reduction, correct locking screw placement, and adequate cement fill.
Post-Operative Protocol & Rehabilitation
The post-operative management and rehabilitation protocol are critically tailored to optimize pain control, facilitate early mobilization, and integrate subsequent oncologic care, aiming to maximize the patient's functional recovery and quality of life in the context of advanced metastatic disease.
Immediate Post-Operative Period (Days 0-7):
1.
Pain Management:
*
Multimodal Analgesia:
An aggressive multimodal pain regimen was initiated immediately, including patient-controlled analgesia (PCA) with opioids, regularly scheduled NSAIDs (if no contraindications, considering renal function and bleeding risk), acetaminophen, and potentially neuropathic agents (gabapentin/pregabalin) for any radicular or neuropathic pain components.
*
Regional Anesthesia:
Consideration for an adductor canal block or fascia iliaca block pre- or intra-operatively can significantly reduce immediate post-operative pain and opioid requirements.
2.
Wound Care:
* The surgical site was inspected daily for signs of infection (erythema, swelling, discharge), hematoma, or wound dehiscence.
* Dressing changes were performed as per standard hospital protocol. Sutures or staples were typically removed at 10-14 days post-op.
3.
Deep Vein Thrombosis (DVT) Prophylaxis:
* Pharmacological DVT prophylaxis (e.g., low molecular weight heparin or direct oral anticoagulants) was initiated once bleeding risk was deemed acceptable, usually within 12-24 hours post-surgery.
* Mechanical prophylaxis (intermittent pneumatic compression devices) was used concurrently.
4.
Mobilization and Rehabilitation (Physical Therapy):
*
Day 0-1:
Out-of-bed activity was initiated immediately. Physical therapy (PT) commenced with passive and active-assisted range of motion (ROM) exercises for the hip and knee.
*
Weight-Bearing Status:
Given the robust intramedullary nail fixation spanning the entire femur, and the cement augmentation providing immediate mechanical stability, the patient was allowed immediate full weight-bearing as tolerated (FWBAT) with an assistive device (walker). This aggressive weight-bearing strategy is a key advantage of modern IMN constructs in pathological fractures.
*
Gait Training:
Initial gait training with a walker was commenced. Emphasis was placed on safe transfers, bed mobility, and progressing ambulation distance.
*
Occupational Therapy (OT):
Assessed the patient's ability to perform activities of daily living (ADLs) and provided adaptive equipment as needed to facilitate independence.
5.
Monitoring and Systemic Management:
*
Hypercalcemia:
Close monitoring of serum calcium levels was crucial, with continued intravenous fluids and appropriate medical therapy (e.g., bisphosphonates or calcitonin) to manage hypercalcemia of malignancy.
*
Oncologic Review:
Communication with the patient's medical oncologist was maintained to coordinate the timing of resuming systemic therapy and initiating adjuvant radiation.
Early Post-Operative Period (Weeks 1-6):
1.
Progression of Rehabilitation:
* The patient continued with supervised physical therapy, gradually progressing weight-bearing and ambulation distance. The focus shifted to strengthening exercises for the hip and knee musculature, improving balance, and stair training.
* The goal was to transition from a walker to crutches or a cane, ultimately aiming for independent ambulation without an assistive device if feasible.
2.
Adjuvant Therapy:
*
Radiation Therapy:
As soon as the surgical wound had sufficiently healed (typically 2-4 weeks post-op), the patient was referred to radiation oncology. Post-operative radiation to the stabilized femoral lesion is highly recommended for local tumor control, pain reduction, and to potentially mitigate disease progression at the surgical site, particularly for radiosensitive tumors like breast cancer metastases. A typical regimen might involve 30 Gy in 10 fractions.
*
Systemic Oncology Treatment:
Her medical oncologist resumed her systemic therapy (Anastrozole, Palbociclib, Denosumab) as soon as medically appropriate, usually once she was tolerating oral intake and stable from a surgical standpoint. Denosumab (or bisphosphonates) play a crucial role in reducing skeletal-related events.
3.
Follow-up:
* Regular orthopedic follow-up appointments were scheduled to monitor wound healing, implant integrity, and assess functional progress. Radiographs were typically obtained at 6 weeks and 3 months post-op to check for implant loosening, fracture healing (though true union is often not the primary goal), or progression of lytic disease around the hardware.
* Close coordination with medical and radiation oncology continued for ongoing systemic disease management and surveillance.
Long-Term Post-Operative Period (Beyond 6 weeks):
1.
Functional Optimization:
Continued focus on maximizing functional independence and quality of life. The patient was encouraged to maintain an active lifestyle within her pain tolerance and physical capabilities.
2.
Surveillance for Complications:
*
Implant Failure:
Monitoring for signs of hardware failure (e.g., screw loosening, nail breakage) on serial radiographs, which can occur due to continued tumor lysis or mechanical stress over time.
*
Infection:
Vigilance for late prosthetic infection, though rare, is important.
*
Local Progression:
Monitoring for local recurrence or progression of metastatic disease around the implant.
*
New Skeletal Events:
Continued surveillance for new pathological fractures or symptomatic metastatic lesions elsewhere in the skeleton.
*
Pain Management:
Ongoing comprehensive pain management strategies were employed, often involving chronic pain specialists.
3.
Psychological Support:
Providing psychological support and access to palliative care services was important given the patient's advanced cancer diagnosis and the impact of treatment.
The post-operative protocol prioritizes functional recovery and comfort, integrating seamlessly with comprehensive oncologic care to provide the best possible outcome for patients with metastatic bone disease.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- High Index of Suspicion: Always consider metastatic bone disease in older patients presenting with atraumatic or low-energy fractures, particularly in weight-bearing bones (femur, spine). A known cancer history makes the diagnosis highly probable, but even in its absence, a pathologic fracture mandates a full workup to find the primary.
- Prophylactic Fixation is Superior: Fixation of an impending pathological fracture (Mirels' score ≥ 7) is generally preferred over fixing a frank fracture. Prophylactic surgery is technically less challenging, has lower morbidity, better outcomes, and allows for planned surgery before an acute event.
- Multidisciplinary Approach is Key: Optimal management requires seamless collaboration between orthopedic oncology/trauma, medical oncology, radiation oncology, palliative care, pain management, and physical therapy. This ensures comprehensive care for both the orthopedic issue and the underlying malignancy.
-
Life Expectancy Guides Surgical Strategy:
Use prognostic scoring systems (e.g., Modified Tokuhashi, ECOG/Karnofsky performance status) to estimate life expectancy.
- Short prognosis (<6 months): Focus on durable, simple fixation (e.g., IMN, plating) for rapid mobilization and pain relief, with minimal morbidity.
- Longer prognosis (>1-2 years): Consider more extensive reconstructions (e.g., endoprosthesis, custom implants) for greater longevity and potentially limb salvage.
- Intramedullary Nailing for Long Bone Diaphyseal & Proximal/Distal Metaphyseal Lesions: For femoral and humeral metastases, IMN is the implant of choice due to its load-sharing characteristics, minimal soft tissue stripping, and ability to bypass extensive disease segments. A long nail that prophylactically extends beyond the primary lesion to the entire bone segment is recommended to prevent new fractures or stress risers.
- Cement Augmentation (PMMA) is Crucial: Polymethacrylate (PMMA) bone cement significantly enhances construct stability, especially in large lytic defects, providing immediate mechanical support and filling voids. It also generates an exothermic reaction, contributing to local tumor necrosis. It is particularly valuable for proximal and distal lesions where screw purchase might be compromised.
- Biopsy is Mandatory for Confirmation: A tissue diagnosis is essential. If the primary is unknown, or if there is any suspicion of a different etiology (e.g., new primary, transformation), a core needle or open biopsy is critical. For known primaries, intraoperative biopsy during definitive fixation is typically sufficient.
- Post-operative Radiation Therapy: Highly recommended for local tumor control and pain relief at the fracture site after surgical stabilization, especially for radiosensitive tumors (e.g., breast, prostate, myeloma, lung). It improves local control rates and reduces the risk of hardware failure.
- Bone-Modifying Agents: Continue bisphosphonates (e.g., zoledronic acid) or denosumab perioperatively and long-term, as they reduce skeletal-related events (SREs), alleviate pain, and improve bone quality.
- Early Mobilization: The primary goal of stabilization is early weight-bearing and functional recovery to prevent complications of immobility and improve quality of life.
Pitfalls:
- Delayed Diagnosis or Misdiagnosis: Attributing a pathological fracture solely to trauma without investigating the underlying etiology (especially if primary is unknown) delays critical oncological management.
- Inadequate Surgical Fixation: Using short implants, non-locked constructs, or failing to bypass the entire lesion/bone segment. This leads to early hardware failure, pain, and re-operation, which is particularly devastating in this patient population.
- Underestimation of Tumor Burden: Not performing complete skeletal imaging (e.g., PET-CT, bone scan) can miss other symptomatic or impending metastatic lesions, leading to subsequent fractures or uncontrolled disease.
- Failure to Augment with Cement: For large lytic defects, omitting cement augmentation drastically reduces the immediate stability and durability of the construct, increasing the risk of mechanical failure.
- Poor Communication with Oncology Team: Lack of coordination regarding systemic therapy, radiation, or hypercalcemia management can compromise the overall treatment plan and patient outcomes.
- Overly Aggressive Surgery: Performing extensive resections or complex, highly morbid reconstructions in patients with very limited life expectancy may not offer commensurate functional benefits and can prolong recovery, hindering quality of life.
- Infection Risk: Cancer patients are often immunocompromised, increasing the risk of surgical site infection. Meticulous aseptic technique, perioperative antibiotics, and careful wound management are paramount.
- Ignoring Hypercalcemia of Malignancy: This common and potentially life-threatening complication must be actively sought and managed pre- and post-operatively.
- Neurological Deficits: Failing to assess for or manage existing or new neurological deficits, particularly in spinal metastases, can lead to permanent impairment.
- Refractory Pain: Despite surgical stabilization, patients may experience persistent pain due to ongoing tumor progression, neuropathic pain, or hardware-related issues, requiring continuous and aggressive pain management strategies, often involving a pain specialist.
