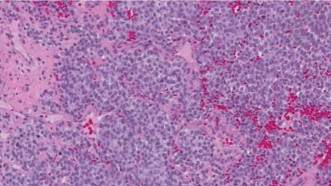
Introduction & Epidemiology
Ewing sarcoma (ES) is a highly aggressive, malignant small round blue cell tumor of bone and soft tissue, belonging to the Ewing sarcoma family of tumors (ESFT). It is characterized by specific chromosomal translocations, most commonly t(11;22)(q24;q12), resulting in the EWS-FLI1 fusion gene, which serves as a molecular hallmark and drives its oncogenesis.
Epidemiologically, ES is the second most common primary malignant bone tumor in children and adolescents, after osteosarcoma.
*
Incidence
: Approximately 2.9 cases per million per year in the United States.
*
Age
: Peak incidence occurs in the second decade of life, typically between 10 and 20 years, with 80% of cases diagnosed before the age of 30. It is rare in very young children (<5 years) and adults (>30 years), though it can occur.
*
Gender
: Slight male predominance (M:F ratio ~1.5:1).
*
Location
: While classically described as a diaphyseal tumor of long bones, ES can occur in any bone or even in soft tissues.
*
Axial skeleton
: Approximately 40-50% of cases, primarily pelvis, spine, ribs, and scapula.
*
Appendicular skeleton
: Approximately 40-50% of cases, commonly in the femur, tibia, humerus, and fibula.
*
Soft tissues
: 10-20% of cases.
*
Clinical Presentation
: Patients typically present with localized pain and swelling, which may be insidious and progressive. Systemic symptoms like fever, weight loss, and fatigue can also occur, mimicking infection. A palpable mass may be present. Pathologic fractures can occur in 5-10% of cases.
*
Prognosis
: Prognosis is highly dependent on the presence of metastatic disease at diagnosis, tumor size, and response to neoadjuvant chemotherapy. Approximately 25% of patients have detectable metastases at presentation, most commonly to the lungs, bone, and bone marrow.
Surgical Anatomy & Biomechanics
Surgical anatomy and biomechanics are critically important in the management of Ewing sarcoma, as successful oncologic resection necessitates wide margins while preserving maximal function and skeletal integrity. The diverse anatomical locations of ES present unique challenges:
Long Bones (Femur, Tibia, Humerus)
- Diaphyseal Involvement : ES frequently involves the diaphysis or metadiaphysis, often leading to significant cortical destruction and soft tissue extension. The peritumoral edema and reactive zone are extensive, requiring wide margins that may necessitate resection of a substantial segment of the bone.
- Neurovascular Bundles : Close proximity to major neurovascular structures (e.g., femoral artery/vein, sciatic nerve in the thigh; brachial artery/median nerve in the arm; popliteal vessels/tibial nerve in the leg) mandates meticulous dissection.
-
Reconstruction
: Large segmental defects require robust reconstruction to restore mechanical stability and function.
- Endoprostheses : Provide immediate stability and allow early mobilization. Biomechanically, they replace the resected bone segment and often the adjacent joint, allowing for weight-bearing or upper extremity function. Load transfer and implant-bone interface longevity are key biomechanical considerations.
- Allografts : Segmental allografts restore bone stock but are susceptible to nonunion, infection, and fracture, impacting long-term biomechanical integrity.
- Vascularized Autografts (e.g., fibula) : Biomechanically, they provide living bone but require significant hypertrophy to withstand physiological loads, a process that takes years.
- Rotationplasty (e.g., distal femur) : A specialized reconstruction for distal femoral tumors, where the distal limb is rotated 180 degrees and reimplanted, creating an ankle joint that functions as a knee for a prosthesis. This provides excellent long-term functional outcomes in selected pediatric patients by preserving the growth potential of the knee joint.
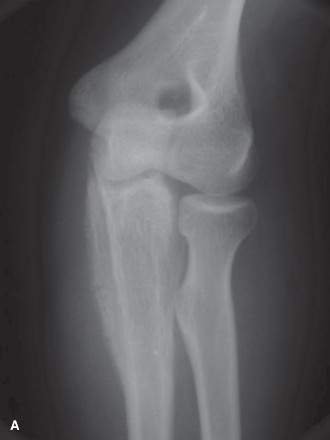
This image potentially depicts a radiographic representation of a Ewing sarcoma in a long bone, highlighting the typical diaphyseal involvement and periosteal reaction.
Pelvis
- Complex Anatomy : The pelvis is an anatomically intricate region with numerous critical structures: major vessels (iliac arteries/veins), nerves (sciatic, femoral, lumbosacral plexus), viscera (bladder, rectum), and muscular attachments for gait and trunk stability.
- Margins : Achieving wide surgical margins in the pelvis is inherently challenging due to its irregular shape and the proximity of vital structures. Marginal or intralesional resections carry a higher risk of local recurrence.
-
Biomechanical Impact
: Resection of parts of the innominate bone (ilium, ischium, pubis) can severely compromise weight-bearing mechanics, hip joint stability, and the attachment sites for major muscle groups, leading to significant gait disturbance and pelvic instability.
- Type I (Iliac) : Resections generally less impactful on weight-bearing.
- Type II (Periacetabular) : Directly affects hip joint and weight-bearing, requiring complex reconstruction to preserve function.
- Type III (Ischial/Pubic) : Can affect sitting balance and muscle attachments.
- Type IV (Sacral) : High risk of neurological deficit.
- Reconstruction : Often involves custom implants, allograft-prosthesis composites, or sometimes no reconstruction (e.g., for certain Type I resections) depending on the extent of bone loss and biomechanical demands.

This image could represent a pelvic Ewing sarcoma on imaging (e.g., MRI) showing the extent of the tumor and its relationship to vital structures.
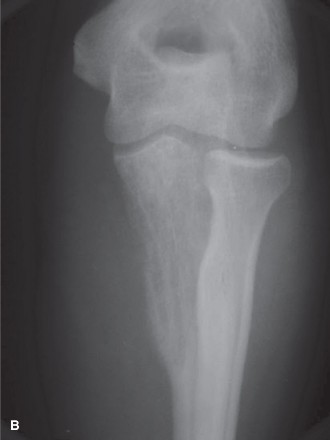
This image might show a different view of a pelvic lesion or a post-resection defect prior to reconstruction.
Spine
- Neurospinal Axis : Spinal ES poses a direct threat to neurological function. Resection requires precise knowledge of the spinal cord, nerve roots, and vertebral artery anatomy.
- Stability : Vertebrectomy leads to segmental instability, necessitating robust spinal reconstruction with cages, allografts, or custom implants, coupled with extensive internal fixation (pedicle screws, rods) to restore biomechanical stability and protect the neural elements.
- Approaches : Often requires combined anterior and posterior approaches to achieve safe en bloc resection and circumferential reconstruction.
This image likely depicts a spinal Ewing sarcoma, potentially on an axial or sagittal MRI, demonstrating spinal cord compression or vertebral body involvement.
Ribs and Scapula
- Ribs : Resection requires consideration of chest wall stability and pulmonary mechanics. Large defects may require reconstruction with mesh or prosthetic materials to prevent flail chest and paradoxical breathing.
- Scapula : Resection impacts shoulder girdle function and stability. Reconstructions range from simple resection (e.g., partial scapulectomy) to complex procedures involving arthrodesis or custom prostheses, depending on the extent and location of the tumor (e.g., glenoid involvement).
In all locations, understanding the tumor's relationship to surrounding muscle compartments, fascia, and periosteum is crucial for planning oncologically sound margins. The extent of soft tissue involvement often dictates the necessity for musculocutaneous flaps or free tissue transfers for wound closure and functional restoration.
Indications & Contraindications
The management of Ewing sarcoma is multimodal, integrating neoadjuvant chemotherapy, local control (surgery and/or radiation therapy), and adjuvant chemotherapy. Surgical resection, when feasible with wide margins, is often the preferred method for local disease control.
Indications for Operative Intervention
-
Resectable Primary Tumor
:
- Achievable wide or marginal surgical margins without unacceptable morbidity.
- Good response to neoadjuvant chemotherapy, indicating tumor shrinkage and potential for clear margins.
- Absence of widespread metastatic disease that would preclude aggressive local treatment.
- This is the cornerstone of curative intent treatment for localized disease.
-
Pathologic Fracture Management
:
- Stabilization of an unstable fracture, especially in weight-bearing bones, to improve patient comfort and facilitate chemotherapy administration.
- Definitive tumor resection and reconstruction are often performed concurrently or sequentially after initial stabilization and completion of neoadjuvant chemotherapy. Initial fracture may lead to tumor contamination, requiring careful planning.
-
Local Recurrence
:
- If surgically resectable with adequate margins, particularly for isolated recurrences.
- Often combined with further chemotherapy and/or radiation.
-
Oligometastatic Disease
:
- Resection of solitary or limited (e.g., <5 lesions) pulmonary metastases, or solitary bone metastases, in conjunction with systemic therapy, can improve survival outcomes in select patients.
-
Biopsy
:
- Though not a definitive treatment, an open biopsy may be indicated if core needle biopsy is inconclusive, or if the tumor location mandates a specific surgical approach for diagnostic purposes, with careful planning for subsequent definitive resection.
Contraindications for Operative Intervention
-
Unresectable Tumor
:
- Extensive local invasion into critical neurovascular structures, major viscera, or joint capsules that precludes achieving adequate surgical margins without unacceptable functional loss or life-threatening complications.
- Tumors with diffuse involvement of an entire bone or multiple bones without clear planes for resection.
-
Widespread Metastatic Disease
:
- Multifocal metastatic disease that is not amenable to local therapy, indicating that systemic control is paramount and local surgical intervention would not alter prognosis.
-
Poor Performance Status or Severe Comorbidities
:
- Patients with significant medical comorbidities or poor overall health status that would make them unfit for major surgical procedures.
-
Inadequate Response to Neoadjuvant Chemotherapy
:
- While not an absolute contraindication, a poor response may indicate a highly aggressive tumor or chemotherapy resistance, necessitating reconsideration of surgical approach, extent of resection, or primary reliance on radiation therapy for local control.
-
Risk of Excessive Morbidity
:
- If the planned resection would result in a devastating functional deficit (e.g., sacrifice of a limb without possibility of functional prosthetic use, severe neurological deficit) that significantly outweighs potential oncologic benefit, alternative local control methods (e.g., definitive radiation) may be favored.
Operative vs. Non-Operative Indications
| Indication Type | Operative Management | Non-Operative Management (or primarily non-surgical) |
|---|---|---|
| Primary Tumor | Resectable tumor (axial or appendicular) with potential for wide/marginal margins. | Unresectable tumor (extensive neurovascular/visceral invasion, diffuse involvement). |
| Metastatic Disease | Oligometastatic disease (e.g., solitary lung or bone metastases). | Widespread, disseminated metastatic disease; poor response to systemic therapy. |
| Pathologic Fracture | Stabilization, and/or definitive resection/reconstruction. | Palliative management for pain if surgical risks outweigh benefits; widespread metastases. |
| Recurrence | Localized, resectable recurrence. | Diffuse or unresectable local/distant recurrence. |
| Patient Status | Good performance status, manageable comorbidities. | Poor performance status, severe comorbidities precluding major surgery. |
| Local Control | Preference for definitive surgical excision for optimal local control. | Primary reliance on definitive radiation therapy (e.g., in pelvis, spine where surgery is difficult). |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is paramount for successful oncologic resection of Ewing sarcoma, given its aggressive nature and the need for complex reconstructions. A multidisciplinary approach involving orthopedic oncology, medical oncology, radiation oncology, diagnostic radiology, pathology, and reconstructive plastic surgery is essential.
Pre-Operative Planning
-
Multidisciplinary Tumor Board Review
:
- Case presentation, review of all imaging, biopsy results, and treatment response to neoadjuvant chemotherapy.
- Discussion and consensus on the optimal local control strategy (surgery vs. radiation vs. combined).
- Definition of surgical goals: en bloc resection with wide margins, functional preservation, and reconstruction strategy.
-
Imaging Review
:
- MRI with Contrast : Gold standard for local staging, evaluating tumor extent, soft tissue involvement, relationship to neurovascular structures, and intramedullary extension. Crucial for planning osteotomies.
-
CT Scan
:
- Local : Provides detailed bony anatomy, cortical destruction, and calcification pattern. Useful for 3D reconstruction and custom implant planning.
- Chest (HRCT) : Essential for detecting pulmonary metastases.
- PET/CT : For systemic staging and evaluation of metabolic activity, particularly in cases of suspected multifocal disease or uncertain chemotherapy response.
- Bone Scan : Technetium-99m bone scintigraphy to detect skeletal metastases.
-
Biopsy
:
- The biopsy tract must be planned carefully to ensure its excisability en bloc with the definitive tumor resection. This often means a direct longitudinal approach without undermining skin flaps.
- Usually performed via core needle biopsy under image guidance (CT or ultrasound). Open biopsy is reserved for specific indications (e.g., inconclusive core biopsy).
-
Neoadjuvant Chemotherapy
:
- Universally administered prior to surgery to reduce tumor burden, treat micrometastases, and evaluate chemosensitivity. This allows for safer and more effective local control.
- Surgical timing is critical, usually several weeks after the last cycle of neoadjuvant chemotherapy to allow for marrow recovery and minimization of chemotherapy-related surgical risks.
-
Reconstruction Planning
:
- Detailed plan for the anticipated bone defect and soft tissue coverage.
- Consideration of options: modular endoprostheses, allografts, autografts (vascularized fibula), allograft-prosthesis composites, or rotationplasty.
- Pre-operative templating with radiographs and CT reconstructions to determine implant size and position.
- In complex cases (e.g., pelvis, spine), 3D printing of models or custom implants may be required.
- Plastic surgery consultation for potential flap coverage (e.g., gastrocnemius, latissimus dorsi flaps) for large soft tissue defects.
-
Blood Management
:
- Pre-operative anemia correction.
- Type and cross-match for multiple units of packed red blood cells.
- Consideration of cell saver technology.
- Nutritional Assessment : Optimization of nutritional status, especially in patients who have undergone extensive chemotherapy.
Patient Positioning
Patient positioning is dictated by the tumor's anatomical location and the chosen surgical approach. General principles include:
*
Access and Exposure
: Position to allow wide exposure for tumor resection and reconstruction, considering potential need for extensile approaches.
*
Sterile Field
: Prep and drape a wide sterile field, extending beyond the immediate surgical site, to allow for potential additional incisions, graft harvest sites, or intraoperative adjustments.
*
Neurovascular Protection
: Careful padding of pressure points and protection of peripheral nerves.
*
Fluoroscopy/Imaging Access
: Ensure intraoperative fluoroscopy or other imaging modalities can be easily utilized.
*
Pathologic Fracture
: Handle the affected limb with extreme care to prevent further displacement or tumor spillage.
Detailed Surgical Approach / Technique
The surgical technique for Ewing sarcoma aims for en bloc resection with wide oncologic margins while preserving maximum limb function. The approach is highly individualized based on tumor location, size, and relationship to neurovascular structures.
Principles of Oncologic Resection
- En Bloc Resection : The entire tumor, along with a cuff of normal tissue (wide margin), must be removed as a single unit without violation of the tumor pseudocapsule or gross contamination of the wound.
- Biopsy Tract Excision : The entire biopsy tract, from skin to tumor, must be included in the en bloc resection specimen to prevent local recurrence from seeded cells.
- Adequate Margins : The goal is a wide margin (typically >2-3 cm of uninvolved tissue) but marginal or even intralesional margins may be accepted in critical areas (e.g., spine, pelvis) if wide margins are not achievable without unacceptable morbidity, with adjuvant radiation playing a compensatory role.
- Neurovascular Preservation : Meticulous dissection to identify, protect, and preserve vital nerves and vessels unless they are demonstrably involved by tumor and must be sacrificed for oncologic clear margins. If sacrificed, immediate reconstruction (e.g., vascular bypass, nerve graft) should be considered.
- Reconstruction Planning : A pre-determined plan for skeletal and soft tissue reconstruction is crucial.
Approach Considerations by Location
Long Bones (e.g., Distal Femur, Proximal Tibia, Humerus)
-
Exposure
:
- Standard extensile approaches are utilized (e.g., anterior approach for distal femur, medial or lateral approach for proximal tibia, deltopectoral or posterior approach for humerus).
- The skin incision is carefully planned to encompass the biopsy scar.
- Internervous Planes : Dissection proceeds through known internervous planes to minimize muscle damage and protect nerves and vessels. For instance, in the distal femur, the anterior approach involves splitting the rectus femoris, retracting the vastus medialis/lateralis, and carefully identifying the femoral artery/vein and sciatic nerve posteriorly.
- Tumor Delineation : The reactive zone around the tumor, often enhanced on MRI, guides the soft tissue dissection. Perfusion of soft tissues should be assessed after dissection, especially prior to flap closure.
- Osteotomy : Osteotomies are performed proximally and distally to the tumor, typically with oscillating saws, ensuring a pre-planned length of normal bone as a wide margin. Intraoperative frozen sections may be used to confirm bone margins if there is concern.
-
Reconstruction
:
- Modular Endoprosthesis : After tumor resection, the resected bone segment is replaced with a custom-fit or modular endoprosthesis. Components are secured with bone cement or press-fit fixation. Joint reconstruction typically involves a hinged knee or elbow prosthesis for stability.
- Allograft : An intercalary allograft is fixed to the host bone with plates and screws, often requiring additional autograft or bone morphogenetic protein (BMP) at the host-allograft junction to promote healing. Osteoarticular allografts replace both bone and joint surface.
- Vascularized Fibula Autograft : The fibula, often harvested from the contralateral leg, is transferred with its pedicle to bridge a bone defect. Requires microvascular anastomosis.
- Rotationplasty : For distal femoral tumors, the knee is disarticulated, and the distal limb is rotated 180 degrees and reimplanted, allowing the ankle to function as a knee for a specialized prosthesis.
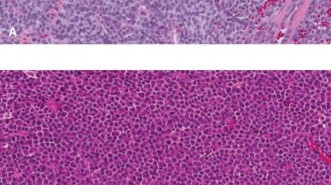
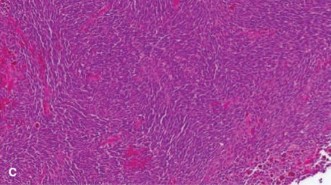
This image could illustrate a resected long bone specimen containing the Ewing sarcoma, demonstrating the gross tumor and achieved margins.
Pelvis
- Complexity : Pelvic resections are among the most challenging due to the intricate anatomy and deep location.
-
Approaches
:
- Type I (Ilium) : Anterior (modified ilioinguinal) or posterior (gluteal) approaches depending on location.
- Type II (Periacetabular) : Often requires combined approaches (e.g., anterior iliofemoral and posterior gluteal) for complete exposure.
- Type III (Ischium/Pubis) : Perineal or obturator approaches.
- Resection : Internal hemipelvectomy, classified by Enneking and Dunham (Type I-IV). Requires careful ligation of vessels and identification of nerve roots (lumbosacral plexus, sciatic nerve).
-
Reconstruction
:
- Often involves custom 3D-printed prostheses or allograft-prosthesis composites for periacetabular defects to restore hip articulation.
- Simple resections (e.g., iliac wing) may require no reconstruction.
- Pelvic fixation can be achieved with reconstruction plates and screws, sometimes with fibular strut grafts.
- Soft Tissue Coverage : Given the large dead space and potential for skin tension, regional flaps (e.g., gluteus maximus, rectus abdominis) or free flaps may be necessary for robust closure.
Spine
- Goals : En bloc resection of the involved vertebral body/bodies while decompressing the spinal cord and stabilizing the spine.
-
Approaches
: Frequently requires a combined anterior and posterior approach.
- Anterior : Thoracotomy or retroperitoneal approach for access to vertebral bodies.
- Posterior : Laminectomy and pedicle screw fixation for stabilization.
- Resection : Vertebrectomy is performed, often with meticulous separation of the dura and nerve roots.
-
Reconstruction
:
- The vertebral body defect is reconstructed using expandable cages (titanium or carbon fiber), allografts, or custom implants.
- Rigid posterior instrumentation (pedicle screws and rods) provides immediate stability.
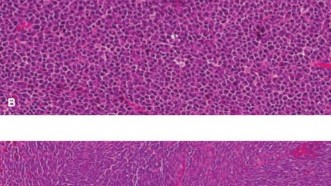
This image could be an intraoperative view during a spinal resection, showing instrumentation or the exposed vertebral column after tumor removal.
Soft Tissue Coverage
- Following large resections, particularly in the lower extremity or pelvis, significant soft tissue defects can result.
- Local Flaps : Myocutaneous or fasciocutaneous flaps are harvested from adjacent tissues (e.g., gastrocnemius flap for knee, latissimus dorsi for shoulder).
- Free Flaps : Microvascular transfer of distant tissue (e.g., anterolateral thigh flap, latissimus dorsi free flap) may be required for complex, large defects or in radiated fields.
Pathological Examination
- The resected specimen is meticulously oriented and marked for the pathologist.
- Ink is applied to surgical margins to allow for precise assessment of tumor-free margins.
- Histopathological examination, including immunohistochemistry (CD99, NKX2.2) and molecular genetics (EWS-FLI1 fusion), confirms diagnosis and assesses tumor response to neoadjuvant chemotherapy (tumor necrosis percentage), which is a key prognostic indicator.
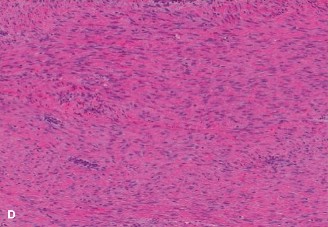
This image might display the final appearance of a reconstruction in a long bone or pelvis, showing the implanted prosthesis or allograft.
Complications & Management
Ewing sarcoma surgery, often complex and extensive, carries a significant risk of both immediate and long-term complications. These can be broadly categorized as local surgical complications and those related to the multimodal treatment regimen.
Local Surgical Complications
-
Local Recurrence
:
- Incidence : Variable, but reported higher with marginal or intralesional resections, especially in challenging anatomical locations like the pelvis and spine. Typically ranges from 10-30%.
- Etiology : Inadequate surgical margins, tumor spillage, or highly aggressive tumor biology.
- Management : Re-resection if technically feasible, often combined with additional chemotherapy and/or definitive radiation therapy. Salvage may involve amputation in extremity cases.
-
Infection
:
- Incidence : Higher in cases involving large reconstructions (endoprostheses, allografts), typically 5-15% for endoprostheses, potentially higher with allografts or in radiated fields.
- Etiology : Contamination during surgery, immunocompromise from chemotherapy, poor wound healing.
- Management : Aggressive debridement, targeted antibiotics (IV and oral), irrigation. For prosthetic infections, may require implant retention with debridement and antibiotics (DAIR), two-stage revision arthroplasty, or definitive implant removal.
-
Hardware Failure / Nonunion
:
-
Incidence
:
- Endoprostheses : Aseptic loosening (5-10%), structural failure (fracture of implant).
- Allografts : Nonunion at the host-allograft junction (20-30%), fracture of the allograft.
- Etiology : Mechanical stress, insufficient bone healing, infection, suboptimal fixation, radiation effects.
- Management : Revision surgery (re-fixation, bone grafting for nonunion, revision arthroplasty for loosening, exchange of broken components).
-
Incidence
:
-
Neurovascular Injury
:
- Incidence : Varies significantly with tumor location and complexity of dissection (e.g., pelvis, spine, proximal humerus). Can range from <1% to >10%.
- Etiology : Direct trauma during dissection, compression, or thermal injury.
- Management : Immediate repair of vessels (primary anastomosis, vein graft) or nerves (primary repair, nerve graft) if feasible. Post-operative neurological deficit management (physiotherapy, bracing).
-
Wound Complications
:
- Incidence : Hematoma, seroma, wound dehiscence, skin necrosis (5-10%). Higher in radiated fields or with large soft tissue resections.
- Etiology : Extensive dissection, dead space, poor tissue perfusion, radiation effects, chemotherapy-induced myelosuppression leading to impaired healing.
- Management : Drainage, debridement, negative pressure wound therapy, primary or rotational flap closure, or free tissue transfer for severe defects.
-
Pathologic Fracture
:
- Incidence : Pre-operative (5-10%), post-operative (e.g., allograft fracture).
- Etiology : Tumor-induced bone destruction, stress shielding from implants.
- Management : Surgical stabilization (internal fixation), revision of reconstruction.
-
Functional Deficits
:
- Incidence : Nearly universal to some degree, highly variable based on resection and reconstruction.
- Etiology : Muscle sacrifice, nerve injury, joint stiffness, limb length discrepancy, gait disturbance.
- Management : Intensive physical and occupational therapy, orthotics, adaptive equipment, gait training, limb lengthening procedures, psychological support.
Multimodal Therapy-Related Complications
- Chemotherapy Toxicity : Myelosuppression, nausea, vomiting, mucositis, cardiotoxicity (doxorubicin), neurotoxicity (vincristine), nephrotoxicity (ifosfamide, cisplatin), secondary malignancies (e.g., AML).
- Radiation Therapy Complications : Radiation dermatitis, osteonecrosis, growth plate arrest, secondary malignancies, pneumonitis (for chest wall irradiation), fibrosis.
Common Complications, Incidence, and Salvage Strategies
| Complication | Typical Incidence (Approx.) | Salvage Strategies |
|---|---|---|
| Local Recurrence | 10-30% | Re-resection (if feasible), salvage amputation, additional chemo/radiation. |
| Infection (Implant) | 5-15% | Debridement & Abx (DAIR), implant retention vs. 2-stage revision, implant removal. |
| Allograft Nonunion | 20-30% | Bone grafting, revision internal fixation, adjunctive BMPs. |
| Endoprosthetic Loosening | 5-10% | Revision arthroplasty, cementation, component exchange. |
| Neurovascular Injury | <1-10% (site-dependent) | Primary repair, nerve grafting, vascular bypass/graft. |
| Wound Dehiscence/Necrosis | 5-10% | Debridement, negative pressure wound therapy, local/free flap coverage. |
| Allograft Fracture | 5-15% | Revision internal fixation, revision to endoprosthesis. |
| Pathologic Fracture | Pre-op: 5-10% | Surgical stabilization (e.g., IM nail, plate fixation), subsequent definitive resection. |
| Limb Length Discrepancy | Variable | Orthotic lifts, epiphysiodesis (contralateral limb), limb lengthening procedures. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is crucial for optimizing functional outcomes, preventing complications, and facilitating a return to activities of daily living for patients treated for Ewing sarcoma. Protocols are highly individualized based on the surgical site, type of reconstruction, patient age, and overall health status. A multidisciplinary team approach involving physical therapists, occupational therapists, and the orthopedic oncology team is essential.
General Principles
- Pain Management : Aggressive pain control to facilitate early mobilization and participation in therapy.
- Wound Care : Meticulous wound care to prevent infection and promote healing.
- Protection of Reconstruction : Strict adherence to weight-bearing restrictions and range-of-motion (ROM) limitations to protect the surgical construct from undue stress.
- Early Mobilization : As soon as safely possible, to prevent stiffness, atrophy, and deep vein thrombosis.
- Gradual Progression : Rehabilitation progresses in a structured, stepwise manner, gradually increasing load and activity.
- Patient Education : Comprehensive instruction for patients and caregivers on exercises, precautions, and signs of complications.
Phase-Specific Rehabilitation
Phase 1: Immediate Post-Operative (Days 0-14)
- Goals : Pain control, wound healing, prevention of complications, initial protection of reconstruction.
-
Activity
:
-
Extremity Surgery (Endoprosthesis/Allograft)
:
- Non-weight-bearing (NWB) or touch-down weight-bearing (TDWB) for lower extremity.
- Upper extremity: Sling immobilization for comfort and protection, passive/active-assisted ROM within protected arcs.
- Isometric exercises of surrounding muscles (e.g., quadriceps sets, gluteal sets).
- Deep breathing exercises, ankle pumps.
- Pelvic Surgery : Bed mobility, gentle trunk rotation, protected transfers to chair (NWB if hip joint involved).
- Spine Surgery : Log-rolling technique for bed mobility, bracing as indicated, gentle core activation without spinal rotation/flexion.
-
Extremity Surgery (Endoprosthesis/Allograft)
:
- Therapy : Education on precautions, basic transfers, assistive device training (crutches, walker).
Phase 2: Early Rehabilitation (Weeks 2-6)
- Goals : Restore basic ROM, initial strengthening, improve endurance, progress weight-bearing as tolerated.
-
Activity
:
-
Extremity
:
- Endoprosthesis : Gradual progression of weight-bearing (e.g., 25-50% PWB) as tolerated, typically starting around 3-4 weeks. Active and passive ROM exercises, progressing to gentle resistance.
- Allograft : Often slower progression, NWB/TDWB for longer periods (6-12 weeks) due to nonunion risk. Gentle active ROM, focus on maintaining joint mobility without stressing healing junctions.
- Rotationplasty : Specific training for ankle dorsiflexion/plantarflexion to simulate knee flexion/extension for prosthetic use.
- Pelvic : Continued protected weight-bearing, gentle hip ROM within pain-free limits, core strengthening.
- Spine : Continued bracing, progressive core strengthening exercises, gentle ambulation.
-
Extremity
:
- Therapy : Gait training with assistive devices, scar massage, proprioceptive exercises, aquatic therapy (once wounds healed).
Phase 3: Intermediate Rehabilitation (Months 2-6)
- Goals : Restore functional strength, improve balance and coordination, normalize gait, advance activities.
-
Activity
:
- Extremity : Progressive increase in weight-bearing (PWB to FWB), resistance exercises, balance training. Introduction of light functional activities (e.g., stationary cycling, swimming).
- Pelvic : Advanced strengthening of hip abductors/adductors and core musculature. Gait re-education, balance training.
- Spine : Discontinuation of bracing (if appropriate), progression to more aggressive core and back strengthening, functional movement patterns.
- Therapy : Sport-specific or activity-specific training for older patients, endurance training, evaluation for orthotics or specialized shoes.
Phase 4: Advanced Rehabilitation / Long-Term Follow-up (Months 6+)
- Goals : Maximize functional independence, return to desired activities, long-term monitoring for complications.
- Activity : Progression to high-level activities as permitted by the reconstruction. Regular exercise for strength, endurance, and flexibility.
- Therapy : Regular follow-up with orthopedic oncology and physical therapy to monitor for implant complications, pain, functional limitations, and recurrence. Lifestyle modifications, psychosocial support.
- Special Considerations : Pediatric patients require ongoing monitoring for limb length discrepancies and potential growth disturbances.
Summary of Key Literature / Guidelines
The management of Ewing sarcoma is a rapidly evolving field driven by ongoing research and collaborative efforts. Key guidelines and landmark literature define current best practices:
- NCCN Guidelines for Bone Cancers : The National Comprehensive Cancer Network (NCCN) provides regularly updated, evidence-based guidelines for the multidisciplinary management of Ewing sarcoma. These are critical for standardized diagnosis, staging, and treatment planning, emphasizing the role of chemotherapy, surgery, and radiation.
- Children's Oncology Group (COG) Protocols : COG is instrumental in developing and conducting large-scale clinical trials for pediatric cancers, including Ewing sarcoma. Their protocols (e.g., AEWS0031, AEWS1031) have established the current standard of care, including specific chemotherapy regimens (e.g., VDC/IE – Vincristine, Doxorubicin, Cyclophosphamide alternating with Ifosfamide, Etoposide) and the importance of timely local control.
- European Society for Medical Oncology (ESMO) Guidelines : ESMO provides similar comprehensive guidelines for adult and pediatric Ewing sarcoma, often aligning with NCCN and COG in core principles while sometimes offering nuanced recommendations based on European data.
- Importance of Multidisciplinary Approach : Consensus across all major guidelines highlights that optimal outcomes are achieved through a multidisciplinary tumor board, ensuring coordinated care from orthopedic oncology, medical oncology, radiation oncology, pathology, and radiology.
- Neoadjuvant Chemotherapy : The literature consistently supports the use of intensive neoadjuvant chemotherapy as standard of care. This approach aims to reduce tumor size, treat micrometastatic disease, and provide prognostic information based on tumor necrosis percentage in the resected specimen (good response: >90% necrosis is associated with improved survival).
-
Local Control (Surgery vs. Radiation)
:
- Surgery with Wide Margins : Considered the preferred method for local control when achievable with acceptable morbidity, particularly for extremity tumors. Studies show superior local control and disease-free survival with wide surgical margins compared to marginal resections or radiation alone.
- Radiation Therapy : An essential component, either as the primary local control modality for unresectable tumors (e.g., many pelvic/spinal tumors) or as an adjuvant to surgery, especially in cases of marginal resection, poor chemotherapy response, or positive surgical margins. Proton therapy is gaining traction for its ability to spare surrounding healthy tissue, particularly in pediatric patients.
- Combined Modality : For certain sites or difficult margins, surgery followed by adjuvant radiation therapy is often utilized.
-
Prognostic Factors
: Key prognostic factors established in the literature include:
- Presence of Metastasis at Diagnosis : The most significant adverse prognostic factor.
- Tumor Volume/Size : Larger tumors (>8-10 cm or >200 ml) are associated with poorer outcomes.
- Response to Neoadjuvant Chemotherapy : Good histological response (>90% necrosis) is a strong positive prognostic indicator.
- Surgical Margin Status : Positive margins correlate with higher local recurrence rates.
- Tumor Location : Axial skeleton tumors (pelvis, spine) generally have a worse prognosis due to difficulty in achieving wide surgical margins and proximity to vital structures.
-
Evolution of Surgical Techniques and Reconstruction
:
- Limb Salvage : Advances in surgical techniques and reconstructive options (modular endoprostheses, allografts, vascularized autografts, rotationplasty) have significantly reduced amputation rates without compromising oncologic outcomes for extremity tumors.
- Pelvic/Spinal Surgery : Progress in complex resections and reconstruction for historically challenging sites has improved local control, though morbidity remains significant.
- Emerging Therapies : Research continues into novel therapeutic agents, including targeted therapies (e.g., PARP inhibitors, IGF-1R inhibitors) and immunotherapy (e.g., PD-1/PD-L1 inhibitors), though their role in standard ES management is still largely investigational.
- Long-Term Outcomes : Growing literature focuses on long-term functional outcomes, quality of life, and surveillance for treatment-related morbidities (e.g., secondary malignancies, chronic pain, functional deficits) in survivors of Ewing sarcoma.
The ongoing collaborative efforts in clinical trials and the adherence to evidence-based guidelines ensure that patients with Ewing sarcoma receive the most effective and contemporary care, balancing oncologic efficacy with functional preservation.
Clinical & Radiographic Imaging