Unraveling Chondromyxoid Fibroma: An Oncology Case Study Guide
Patient Presentation & History
A 28-year-old male, otherwise fit and healthy with no significant medical comorbidities, presented to the orthopedic clinic with a 6-month history of insidious onset, progressively worsening right knee pain. The pain was dull, aching, localized to the proximal tibia, and exacerbated by weight-bearing activities and relieved by rest. He reported occasional nocturnal pain that disrupted sleep. There was no history of acute trauma, but he did recall a minor fall onto the knee approximately 8 months prior, which caused transient discomfort but resolved within a few days. He denied any fevers, chills, night sweats, or significant constitutional symptoms. His occupation involves light manual labor. He is a non-smoker and consumes alcohol socially. Review of systems was unremarkable.
Clinical Examination
Right Lower Extremity
- Inspection: Mild fullness noted over the anteromedial aspect of the proximal right tibia, approximately 5 cm distal to the tibial tubercle. No overt skin discoloration, erythema, or visible deformity. Quadriceps musculature appeared symmetrical bilaterally, with no significant atrophy.
- Palpation: Localized tenderness was elicited directly over the anteromedial proximal tibia. A firm, fixed, non-pulsatile mass measuring approximately 4x3 cm was palpable within the bone, deep to the extensor retinaculum. The overlying skin was not warm. No associated lymphadenopathy was palpable in the groin.
-
Range of Motion (ROM):
- Knee: Full active and passive ROM (0-140 degrees flexion) with terminal flexion pain. No crepitus or mechanical symptoms. Patellar tracking was normal.
- Ankle/Foot: Full, pain-free ROM.
- Neurological Assessment: Sensation intact to light touch in all dermatomes (L2-S1) of the right lower extremity. Motor strength 5/5 in all muscle groups (hip flexion, knee extension, ankle dorsiflexion, plantarflexion, great toe extension). Deep tendon reflexes (patellar, Achilles) were 2+ and symmetrical. No pathological reflexes.
- Vascular Assessment: Distal pulses (dorsalis pedis, posterior tibial) were 2+, symmetrical, and easily palpable. Capillary refill was brisk (<2 seconds). No signs of venous insufficiency.
- Gait: Antalgic gait, favoring the right lower extremity, with a shortened stance phase on the right. Able to ambulate independently.
Imaging & Diagnostics
Initial Radiographs
Anteroposterior (AP) and lateral radiographs of the right tibia were obtained. These revealed an eccentric, geographic lytic lesion in the metaphysis of the proximal right tibia, approximately 4 cm distal to the articular surface. The lesion measured approximately 4.5 x 3.0 cm and exhibited a well-defined, lobulated, sclerotic rim, suggesting a benign or low-grade process. Internal trabeculations giving a "soap bubble" appearance were noted. There was no clear periosteal reaction or cortical breach. No pathological fracture was evident. The joint space appeared well-maintained.
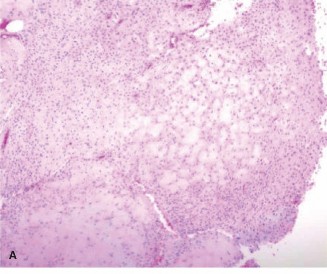
Figure 1: Anteroposterior radiograph of the proximal tibia demonstrating an eccentric, geographic lytic lesion with a sclerotic rim and internal septations.
Computed Tomography (CT)
A CT scan of the right tibia was performed to further characterize the bony architecture and cortical integrity. This confirmed the eccentric, lytic lesion within the proximal tibial metaphysis, immediately adjacent to the medial cortex. The CT provided excellent detail of the lesion's internal septations and confirmed the thick, sclerotic rim. It demonstrated a thin, intact cortical shell over the medial aspect of the lesion with no overt breach, but highlighted significant thinning of the cortex. No aggressive periosteal reaction or soft tissue mass was identified. The articular surface appeared uninvolved. CT was critical for pre-operative templating to assess bone stock and guide the extent of curettage and potential reconstruction.
Magnetic Resonance Imaging (MRI)
An MRI of the right knee and proximal tibia was subsequently performed to assess soft tissue involvement, marrow extension, and relationship to neurovascular structures. The MRI revealed a well-circumscribed intraosseous lesion demonstrating heterogeneous signal intensity. On T1-weighted images, the lesion was largely hypointense, while on T2-weighted images, it showed high signal intensity with internal septations, consistent with its chondromyxoid matrix. Areas of lower signal on T2 were also present, corresponding to the fibrous and collagenous components. Peripheral and septal enhancement was noted after gadolinium administration. There was no evidence of extraosseous extension or significant soft tissue edema beyond the immediate periosteal reaction, which was minimal. The lesion was separated from the articular cartilage by a thin rim of normal bone, and crucial neurovascular structures (popliteal artery, tibial nerve) were safely distant. The MRI further confirmed the absence of a pathological fracture.
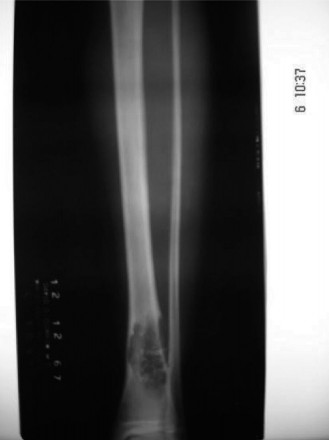
Figure 2: Coronal T2-weighted MRI image showing a well-demarcated, lobulated lesion with high signal intensity and internal septations, confirming its intraosseous nature and absence of significant soft tissue extension.
Biopsy
Given the radiographic characteristics and the patient's age, a percutaneous core needle biopsy was performed under fluoroscopic guidance. This was crucial for definitive diagnosis and differentiation from other lytic lesions, particularly low-grade chondrosarcoma. Histopathological examination revealed characteristic features of chondromyxoid fibroma: lobules of chondroid and myxoid tissue with stellate or spindle-shaped cells in a myxoid background, intermixed with more cellular areas containing uniform ovoid or spindle cells with scattered multinucleated giant cells, typically localized at the periphery of the lobules. Focal areas of nuclear atypia were identified, which is known to occur in CMF but requires careful differentiation from chondrosarcoma. The biopsy confirmed the diagnosis of Chondromyxoid Fibroma.
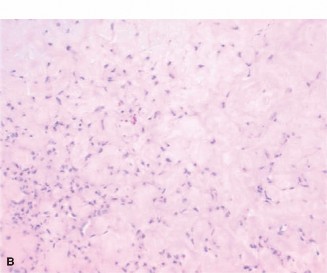
Figure 3: Histopathological image (H&E stain) showing characteristic lobules of chondroid and myxoid tissue, with stellate cells and scattered multinucleated giant cells at the periphery, consistent with chondromyxoid fibroma.
Differential Diagnosis
The radiographic appearance of chondromyxoid fibroma can overlap with several other benign and low-grade malignant bone tumors. A thorough differential diagnosis is crucial for appropriate management.
| Feature | Chondromyxoid Fibroma (CMF) | Enchondroma | Low-Grade Chondrosarcoma | Aneurysmal Bone Cyst (ABC) |
|---|---|---|---|---|
| Age Group | Typically 2nd-3rd decade (10-30 years) | Any age, most common 20-50 years | Typically >40 years | Typically <20 years |
| Location | Metaphysis of long bones (proximal tibia, distal femur); small bones of foot | Medullary cavity of small bones of hands/feet; long bone metaphysis/diaphysis | Pelvis, proximal femur, shoulder, long bones (secondary to enchondroma) | Metaphysis of long bones, vertebrae |
| Radiographic Appearance | Eccentric, geographic lytic lesion with sclerotic rim; lobulated; internal septations ("soap bubble"); no matrix calcification | Well-circumscribed lytic lesion, often with chondroid matrix calcifications (rings and arcs); no sclerotic rim typically | Lytic lesion with endosteal scalloping, cortical destruction/thickening; permeative changes; amorphous calcifications | Expansile, eccentric, lytic lesion; multiple fluid-fluid levels on MRI; thin sclerotic rim; blowout appearance |
| MRI Characteristics | T1 hypointense, T2 hyperintense; heterogeneous; peripheral/septal enhancement; lobulated contour; no fluid-fluid levels | T1 hypointense, T2 hyperintense; lobulated; often "frond-like" enhancement if active | T1 hypointense, T2 hyperintense; aggressive features (cortical breach, soft tissue mass); prominent enhancement; often fluid-fluid levels if secondary | T1/T2 variable, classic fluid-fluid levels (blood products of different ages); septations; T2 hyperintense due to fluid |
| Histopathology | Lobules of myxoid and chondroid tissue; stellate/spindle cells; prominent peripheral multinucleated giant cells; hypercellular areas; mild atypia is common | Mature hyaline cartilage; small, uniform chondrocytes; absence of significant atypia or mitosis | Atypical chondrocytes (enlarged nuclei, binucleation); hypercellularity; myxoid change; permeation of bone; mitosis rare | Blood-filled cystic spaces separated by fibrous septa containing spindle cells, giant cells, and osteoid formation; no epithelial lining |
| Growth Behavior | Slow-growing, benign | Slow-growing, benign | Slow-growing, malignant (low metastatic potential for Grade 1) | Rapidly expansile, benign but locally aggressive |
Surgical Decision Making & Classification
The primary goals of managing a chondromyxoid fibroma are to achieve complete tumor eradication, prevent recurrence, preserve joint function, and maintain skeletal integrity. Given the symptomatic nature of the lesion (pain, nocturnal pain, antalgic gait), its size, and the significant cortical thinning observed on CT, surgical intervention was deemed necessary to prevent a pathological fracture and alleviate symptoms. Non-operative management, such as watchful waiting, is generally reserved for asymptomatic, very small lesions in non-weight-bearing bones, which was not applicable in this case.
The Enneking staging system is routinely applied for benign bone tumors to guide surgical planning and predict recurrence risk. Based on the radiographic and MRI findings, demonstrating an intact but thinned cortical shell and absence of significant soft tissue extension, this lesion was classified as:
*
Enneking Stage 2 (Active):
This stage indicates a benign lesion that is locally aggressive, well-defined radiographically, but may have thin or breached cortical margins and may extend beyond the bone into soft tissues. In this specific case, while the cortex was thinned, it remained intact, and there was no gross soft tissue extension, making it an active benign lesion with potential for local recurrence if not adequately treated.
Surgical intervention was planned as intralesional curettage with adjuvant therapy and bone grafting . The choice of intralesional curettage over wide en bloc excision is justified by the benign nature of CMF and its well-defined borders, which minimize the risk of local recurrence if an adequate intralesional margin is achieved with adjuvant treatment. Wide excision, with its associated morbidity (e.g., bone defect, need for major reconstruction), is typically reserved for malignant tumors or highly aggressive benign lesions with significant extraosseous extension or recurrence after intralesional treatment.
The pre-operative biopsy was crucial not only for definitive diagnosis but also to guide the surgical approach. A carefully planned biopsy tract was created, which was subsequently incorporated into the definitive surgical incision to prevent tumor cell seeding, a critical principle in orthopedic oncology.
Surgical Technique / Intervention
Pre-operative Planning
Detailed pre-operative planning involved reviewing all imaging studies (X-rays, CT, MRI) to precisely delineate the tumor's extent, its relationship to the growth plate, articular cartilage, and neurovascular structures. The plan included:
1.
Patient Positioning:
Supine position on a radiolucent table with a bump under the ipsilateral hip for slight external rotation, allowing optimal access to the anteromedial proximal tibia.
2.
Tourniquet:
A pneumatic tourniquet was applied high on the thigh to achieve a bloodless field, crucial for clear visualization during curettage and identification of viable bone.
3.
Surgical Approach:
A straight longitudinal incision was planned over the palpable mass on the anteromedial aspect of the proximal tibia, ensuring the biopsy tract was excised en bloc. The skin and subcutaneous tissues were incised, and the deep fascia was opened. The periosteum was carefully incised and elevated to expose the underlying bone.
Tumor Excision and Adjuvant Therapy
- Cortical Window Creation: Utilizing power burrs and osteotomes, a generous cortical window was created over the most accessible aspect of the tumor, usually where the cortex was thinnest, taking care to avoid compromising the joint surface or the medial collateral ligament attachment. The window was large enough to allow thorough visualization and instrumentation of the entire lesion.
- Intralesional Curettage: The tumor mass, which had a gelatinous, myxoid consistency, was meticulously curetted using various sizes of angled curettes. The goal was complete removal of all visible tumor tissue from the osseous cavity. Special attention was paid to the periphery of the lesion and any septations, ensuring thorough debridement down to healthy bleeding bone. The cavity was repeatedly irrigated with copious amounts of saline to remove microscopic tumor fragments.
-
Adjuvant Therapy:
Following mechanical curettage, adjuvant therapy was employed to further decrease the risk of local recurrence.
- High-speed burring: The entire tumor cavity wall was aggressively burred to remove any microscopic tumor remnants embedded in the surrounding bone. This creates a larger margin of resection at the microscopic level.
- Phenol Application: A chemical adjuvant, 95% ethyl alcohol (phenol), was applied to the cavity for 3 minutes, then neutralized with copious amounts of normal saline. Phenol acts as a chemical cauterant, inducing osteonecrosis of remaining tumor cells in the cavity walls. Care was taken to protect soft tissues, especially neurovascular structures, during application.
- Cryotherapy: While not performed in this specific case due to equipment availability, liquid nitrogen cryotherapy is another effective adjuvant, inducing cellular necrosis through rapid freezing and thawing cycles.
- Assessment of Cavity: After adjuvant treatment, the cavity was meticulously inspected to ensure no residual tumor was visible and that the cavity walls were firm and sclerotic, indicating effective treatment.
Reconstruction and Fixation Construct
-
Defect Management:
The resulting bone defect was significant, approximately 4.5 cm in its largest dimension, leading to a structural compromise of the proximal tibia. To prevent pathological fracture and restore structural integrity:
- Bone Grafting: The defect was packed with a combination of autologous cancellous bone graft harvested from the ipsilateral iliac crest and allograft cancellous chips. The autograft provided osteoinductive and osteoconductive properties, promoting bone healing, while the allograft augmented volume. The graft was tightly packed into the cavity to provide immediate structural support.
- Bone Cement (PMMA): In some cases, polymethylmethacrylate (PMMA) bone cement can be used as an alternative or adjunct to bone grafting. PMMA offers immediate structural stability and its exothermic reaction can act as a local adjuvant by inducing thermal necrosis of residual tumor cells. However, in a young patient, promoting biological healing with bone graft was prioritized.
-
Internal Fixation:
Given the size of the defect and the load-bearing nature of the proximal tibia, internal fixation was deemed necessary to protect the bone graft and provide immediate stability.
- A proximal tibia locking plate was applied to the medial aspect of the tibia. This plate provided rigid fixation, bridging the defect and protecting the bone graft from excessive stress during early weight-bearing. Screws were carefully placed to ensure maximal purchase in healthy host bone, both proximal and distal to the lesion, avoiding the joint space and the tumor cavity as much as possible, while providing compression across the cortical window.
- Wound Closure: The cortical window was closed with a small bone graft plug or a resorbable mesh to contain the bone graft. The periosteum was repaired, followed by meticulous layered closure of the deep fascia, subcutaneous tissue, and skin. A sterile dressing was applied.
Post-Operative Protocol & Rehabilitation
The post-operative management focused on protecting the surgical site, promoting bone healing, minimizing complications, and restoring function.
Immediate Post-Operative (Day 0-7)
- Pain Management: Multimodal analgesia regimen including opioids, NSAIDs (if not contraindicated for bone healing concerns), and acetaminophen.
- Wound Care: Daily dressing changes, vigilant monitoring for signs of infection (erythema, swelling, discharge). Staples/sutures removed at 10-14 days.
- Deep Vein Thrombosis (DVT) Prophylaxis: Routine DVT prophylaxis (e.g., low molecular weight heparin or aspirin) was initiated and continued for 4-6 weeks.
- Mobilization: Non-weight-bearing (NWB) on the right lower extremity with crutches or a walker. Strict adherence to NWB was emphasized to protect the bone graft and fixation construct.
- Range of Motion: Immediate, gentle, active-assisted range of motion (AAROM) of the knee within a comfortable, pain-free arc was initiated, progressing as tolerated. Continuous passive motion (CPM) machine use for 2-4 hours daily to prevent arthrofibrosis.
Early Rehabilitation (Week 2-6)
- Weight-bearing: Progressive weight-bearing initiated gradually after radiographic evidence of early bone graft incorporation and healing of the cortical window (typically 4-6 weeks post-op). This progression included toe-touch weight-bearing (TTWB), followed by 25% partial weight-bearing (PWB) in a hinged knee brace, advancing to 50% PWB.
-
Physical Therapy:
- ROM: Continue to work on achieving full knee flexion and extension.
- Strengthening: Isometric quadriceps and hamstring exercises. Gentle strengthening of hip and ankle musculature.
- Gait Training: Progression with crutches to single crutch, then independent ambulation as tolerated and strength improves.
- Imaging: Repeat radiographs at 6 weeks and 3 months to assess bone graft incorporation and integrity of the fixation.
Intermediate Rehabilitation (Month 3-6)
- Weight-bearing: Full weight-bearing (FWB) as tolerated, provided there is radiographic evidence of substantial bone graft maturation and stability of the fixation.
-
Physical Therapy:
- Strengthening: Advanced strengthening exercises for the entire lower extremity, including closed kinetic chain exercises (e.g., squats, lunges) and proprioceptive training.
- Functional Training: Focus on gait normalization, balance, and activities of daily living.
- Activity Modification: Avoid high-impact activities or twisting motions on the knee until complete bone healing is confirmed, typically 6-9 months post-op.
Long-Term Follow-up (Beyond 6 Months)
- Imaging Surveillance: Annual radiographs for the first 3-5 years to monitor for local recurrence, which is reported in 10-25% of cases following curettage. MRI may be considered if recurrence is suspected based on symptoms or plain film changes.
- Clinical Assessment: Regular clinical follow-ups to assess pain, function, and stability.
- Return to Activity: Gradual return to sports and higher-level activities after clearance by the surgeon, typically 9-12 months post-operatively, once full strength, range of motion, and radiographic healing are achieved.
- Hardware Removal: Hardware removal is not routinely performed unless symptomatic.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- CMF is a Benign Lesion: Despite occasional worrisome histological features (e.g., cellularity, atypia, giant cells), CMF is fundamentally benign. However, it can be locally aggressive and recur, necessitating meticulous surgical technique.
- Radiographic Features are Key: Eccentric, geographic lytic lesion with a sclerotic rim, often with internal septations, is highly suggestive. Location (metaphysis of long bones) and age (2nd-3rd decade) are important clues.
- Biopsy is Mandatory: Definitive diagnosis hinges on histopathology. Percutaneous core needle biopsy is preferred to minimize tissue contamination, but must be planned carefully so the tract can be excised en bloc with the definitive surgery.
- Thorough Curettage + Adjuvant Therapy: Mechanical curettage alone has a higher recurrence rate. The combination with local adjuvants (high-speed burring, phenol, or cryotherapy) significantly reduces recurrence by treating microscopic tumor cells in the cavity walls.
- Structural Reconstruction: For large defects, structural support with bone graft (auto/allo) and internal fixation (e.g., locking plate) is crucial to prevent pathological fracture and allow early rehabilitation.
- Enneking Staging: Apply the Enneking classification for benign tumors (Stage 2 is typical for symptomatic CMF) to guide surgical aggressiveness and predict outcomes.
- Distinguishing from Chondrosarcoma: This is the most critical differential. CMF can have areas of hypercellularity and atypia, mimicking low-grade chondrosarcoma. Pathologists experienced in bone tumors are essential for accurate diagnosis. Lack of cortical destruction, periosteal reaction, and typical age group favor CMF.
Pitfalls
- Misdiagnosis as Chondrosarcoma: Over-diagnosis of chondrosarcoma leads to overly aggressive surgery (wide excision) with unnecessary morbidity. Conversely, under-diagnosis of chondrosarcoma as CMF can lead to inadequate treatment and potential progression. Rely heavily on expert histopathological review.
- Inadequate Curettage: Incomplete removal of tumor tissue is the leading cause of local recurrence. This highlights the importance of creating a sufficiently large cortical window, using various curette sizes, and meticulous adjuvant therapy.
- Biopsy Tract Contamination: Improper biopsy technique (e.g., multiple passes through different tissue planes, inadequate planning for definitive surgery) can seed tumor cells, making subsequent surgery more complex and increasing recurrence risk. The biopsy tract must be excised with the tumor.
- Pathological Fracture: The lesion can predispose to pathological fracture. If present, it complicates management, requiring careful stabilization and often influencing the choice of internal fixation and reconstruction.
- Neurovascular Injury: Aggressive curettage or adjuvant application (especially phenol) in close proximity to neurovascular bundles (e.g., around the knee or elbow) poses a risk of iatrogenic injury. Meticulous dissection and protection of these structures are paramount.
- Growth Plate Involvement in Pediatric Patients: If the lesion is close to or involves the physis in skeletally immature patients, aggressive curettage or adjuvant therapy can lead to growth arrest or deformity. Balancing tumor eradication with physis preservation is critical.
- Recurrence Management: Recurrent CMF often requires more aggressive re-curettage with robust adjuvant therapy or, in some cases, marginal en bloc excision if the recurrence is particularly aggressive or located in a technically challenging area.
