Patient Presentation & History
A 17-year-old male presented to the orthopedic clinic with a 4-month history of progressive left knee pain and swelling. The pain was initially insidious, described as a dull ache, worse with activity, and minimally responsive to over-the-counter analgesics. Over the past 6 weeks, the pain intensified, becoming constant and sharp, particularly at night, significantly disturbing his sleep. He reported a noticeable increase in the size of his distal thigh/proximal knee region. There was no specific history of trauma, although he recalled a minor fall during a basketball game approximately 5 months prior, which he initially attributed the knee discomfort to. He denies any systemic symptoms such as fever, chills, night sweats, or unintentional weight loss. His medical history is otherwise unremarkable, with no known genetic predispositions or prior radiation exposure. He is an active high school athlete with no significant comorbidities. Family history is negative for malignancy.
Clinical Examination
Inspection: On inspection, there was a visible, firm, fusiform swelling involving the distal third of the left thigh, extending to the suprapatellar region. The overlying skin appeared taut and slightly erythematous, but without overt signs of inflammation or breaks in integrity. There was no obvious atrophy of the surrounding musculature despite limited activity due to pain. A subtle prominence was noted on the anterolateral aspect of the distal femur.
Palpation: Palpation revealed a large, firm, immobile, and tender mass originating from the distal femoral metaphysis, approximately 15 x 10 cm in size. The mass was warm to touch. There was significant point tenderness over the anterolateral distal femur. The mass appeared to be intimately associated with the bone and did not show fluctuance. There was no crepitus. No regional lymphadenopathy was appreciated in the ipsilateral inguinal region.
Range of Motion (ROM): Active and passive range of motion of the left knee was significantly restricted and painful. Flexion was limited to 70 degrees (normal 140-150 degrees), and extension lacked 15 degrees (normal 0 degrees). Both maneuvers elicited sharp pain, especially at the extremes of motion. Patellar tracking was difficult to assess due to swelling, but patellar ballotment suggested a large intra-articular effusion, or at least significant synovial thickening/periarticular swelling.
Neurological Assessment: Distal neurological examination revealed intact sensation to light touch and pinprick in the L2-S1 dermatomes. Motor strength of ankle dorsiflexion, plantarflexion, great toe extension, and knee extension (quadriceps) was 5/5, though resisted knee extension was painful. No pathological reflexes were elicited.
Vascular Assessment: Distal pulses (dorsalis pedis and posterior tibial) were palpable and symmetrical bilaterally. Capillary refill time was brisk in the toes. There was no evidence of venous stasis or deep vein thrombosis.
Imaging & Diagnostics
Plain Radiographs:
Initial anteroposterior and lateral radiographs of the left knee revealed an aggressive, mixed lytic and blastic lesion centered in the distal femoral metaphysis. Key findings included:
*
Cortical destruction:
Irregular destruction of the cortical bone, particularly on the anterior and lateral aspects.
*
Periosteal reaction:
Prominent sunburst periosteal reaction extending into the adjacent soft tissues, indicative of rapid tumor growth. A Codman's triangle was also appreciated.
*
Soft tissue mass:
A large extraosseous soft tissue component was evident, with irregular calcification and ossification within it, confirming the palpable mass.
*
Metaphyseal involvement:
The lesion largely spared the epiphyseal plate but extended proximally into the diaphysis and distally towards the joint line, though the articular cartilage appeared intact on plain films.
* No definite pathological fracture was observed, but the cortical integrity was severely compromised.
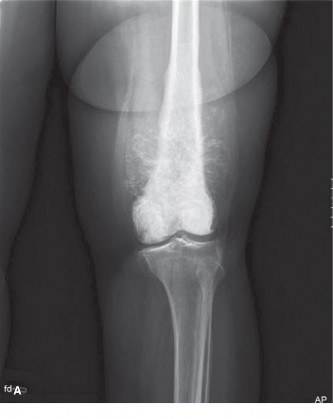
(Figure 1: Anteroposterior radiograph demonstrating a mixed lytic and blastic lesion in the distal femur with significant periosteal reaction and a large soft tissue component.)
Magnetic Resonance Imaging (MRI):
A dedicated MRI of the left femur with gadolinium contrast was performed for local staging.
*
T1-weighted images:
Showed a heterogeneous mass with intermediate to low signal intensity, replacing normal bone marrow.
*
T2-weighted images and STIR sequences:
Demonstrated high signal intensity within the mass and surrounding soft tissue edema, clearly delineating the extent of the intramedullary and extraosseous components.
*
Post-contrast T1-weighted images with fat saturation:
Revealed intense, heterogeneous enhancement of the tumor, indicating its vascular nature.
*
Key findings for local staging:
*
Intramedullary extent:
The tumor extended approximately 12 cm proximally from the distal femoral physis, reaching into the diaphyseal region. No skip lesions were identified.
*
Soft tissue extension:
Extensive extraosseous soft tissue mass measured approximately 10 x 15 x 12 cm, with tumor involvement of vastus medialis and intermedius muscles.
*
Neurovascular bundle involvement:
The popliteal neurovascular bundle (femoral artery, vein, sciatic nerve) was displaced posteriorly by the tumor mass but appeared to be encased by the tumor in a segment of approximately 4 cm, suggesting potential compromise or requiring meticulous dissection.
*
Joint involvement:
The articular cartilage of the distal femur appeared preserved, but the tumor abutted the joint capsule, indicating a potential need for marginal resection of the capsule or wider joint resection if a breach was suspected.
*
Epiphyseal extension:
While primarily metaphyseal, there was evidence of microscopic epiphyseal involvement at the posteromedial aspect.
Computed Tomography (CT) Scans:
*
Local CT:
A CT scan of the left femur provided detailed assessment of cortical destruction, mineralization patterns, and cross-sectional tumor dimensions. It confirmed the extensive cortical breach and allowed for 3D reconstruction crucial for surgical planning and osteotomy guides.
*
Chest CT:
A high-resolution CT of the chest was performed to rule out pulmonary metastases, which are the most common site for osteosarcoma dissemination. This scan revealed several small, non-calcified pulmonary nodules in both lung fields, suspicious for metastatic disease. The largest nodule measured 6 mm in the right upper lobe. This immediately classified the patient as M1 (metastatic disease).
Bone Scan (Technetium-99m): Whole-body bone scintigraphy showed intense radionuclide uptake at the distal left femur, consistent with the primary tumor. No other suspicious foci of increased uptake were identified throughout the axial or appendicular skeleton, suggesting no obvious skeletal metastases elsewhere, which is critical in treatment planning.
Positron Emission Tomography-Computed Tomography (PET-CT): An FDG PET-CT scan was performed to assess metabolic activity of the primary tumor and evaluate for additional distant metastatic sites. It showed intense FDG avidity in the distal left femur (SUVmax 18.2) and confirmed the metabolic activity of the pulmonary nodules identified on chest CT, further supporting their metastatic nature. No other metabolically active lesions were detected.
Biopsy: Following comprehensive imaging, a CT-guided core needle biopsy of the distal femoral lesion was performed by a musculoskeletal radiologist, under strict aseptic conditions. The biopsy trajectory was meticulously planned to avoid contamination of future surgical fields and to ensure an excisable tract with the definitive surgical resection. Histopathological examination revealed highly pleomorphic spindle cells producing osteoid matrix, consistent with high-grade osteosarcoma. Mitotic figures were abundant, and areas of necrosis were present.
(Figure 2: Coronal MRI T1-weighted image demonstrating the extensive intramedullary and extraosseous extent of the osteosarcoma, crucial for templating resection margins.)
Templating: Pre-operative templating was performed based on MRI and CT 3D reconstructions. Resection margins were planned to achieve a wide oncological margin (2 cm proximal and distal intramedullary, 1 cm extraosseous soft tissue margin) while preserving neurovascular structures. The planned reconstruction involved a modular distal femoral endoprosthesis due to the significant bone defect and proximity to the knee joint. The overall length of resection was calculated to be approximately 14 cm, extending into the proximal diaphysis. This templating also guided the choice of endoprosthesis size and articulation.
Differential Diagnosis
Given the patient's age, location, and aggressive radiographic features, the primary differentials for this destructive bone lesion included other primary malignant bone tumors.
| Condition | Typical Age | Radiographic Features | Histopathology | Key Distinguishing Features |
|---|---|---|---|---|
| Osteosarcoma | 10-25 years (bimodal peak: adolescent growth spurt & >60 for secondary) | Mixed lytic and blastic areas, aggressive periosteal reaction (sunburst, Codman's triangle), cortical destruction, soft tissue mass. Often metaphyseal. | Malignant stromal cells producing osteoid matrix (diagnostic feature). Highly pleomorphic cells, abundant mitoses, atypical chondroid or fibrosarcomatous differentiation possible. | Presence of malignant osteoid. Rapid growth, aggressive imaging. Elevated alkaline phosphatase (ALP) in some cases. |
| Ewing Sarcoma | 5-25 years | Permeative lytic lesion, often diaphyseal (can be metaphyseal), lamellated ("onion-skin") periosteal reaction, soft tissue mass. May mimic osteomyelitis. | Sheets of small, round, blue cells with scant cytoplasm. Immunohistochemistry positive for CD99 (MIC2). Characteristic chromosomal translocation t(11;22)(q24;q12) (EWSR1-FLI1) or other EWSR1 translocations. | "Onion-skin" periosteal reaction. Younger age group. Genetic testing for translocation. Bone marrow involvement is common. |
| Chondrosarcoma (High-Grade) | >40 years | Punctate, ring-and-arc calcifications within a lytic lesion, endosteal scalloping, cortical destruction. Primarily affects axial skeleton and proximal long bones but can occur in distal femur. | Malignant cartilage production. Chondrocytes with varying degrees of atypia, hypercellularity, pleomorphism, and mitoses depending on grade. High-grade (G2/G3) lesions show significant pleomorphism, binucleation, and myxoid change. | Presence of malignant cartilage matrix (no osteoid). Older age group. Usually slower growing unless high-grade. May arise from pre-existing enchondroma or osteochondroma. |
| Giant Cell Tumor (GCT) | 20-40 years | Eccentric lytic lesion in metaphysis/epiphysis, often extending to subchondral bone. Non-sclerotic margins, "soap-bubble" appearance. Cortical expansion rather than frank destruction initially. | Spindle cell stromal background with numerous evenly distributed multinucleated giant cells. No osteoid or cartilage matrix production. | Typically purely lytic, epiphyseal extension. Benign but locally aggressive. Absence of malignant osteoid. Histology shows characteristic giant cells. |
The definitive diagnosis of osteosarcoma was established through biopsy, correlating precisely with the aggressive radiographic features and the patient's demographic. The discovery of pulmonary metastases categorized this as Stage III osteosarcoma according to the Enneking Surgical Staging System (G2, T2, M1).
Surgical Decision Making & Classification
The management of high-grade osteosarcoma is inherently multidisciplinary, involving orthopedic oncology, medical oncology, radiation oncology, radiology, and pathology. The presence of pulmonary metastases immediately indicated systemic disease, necessitating a robust neoadjuvant chemotherapy regimen.
Neoadjuvant Chemotherapy:
The patient commenced a standard neoadjuvant chemotherapy protocol, typically comprising a multi-agent regimen such as MAP (Methotrexate, Adriamycin (Doxorubicin), Cisplatin). This approach serves several critical purposes:
1.
Downstaging the tumor:
Potentially shrinking the primary tumor, facilitating a safer and more effective surgical resection with wider margins.
2.
Addressing micrometastases:
Targeting systemic disease (including the known pulmonary nodules) early in the treatment course.
3.
Assessing tumor response:
The degree of tumor necrosis after neoadjuvant chemotherapy (assessed in the resected specimen) is a powerful prognostic indicator. Good responders (≥90% necrosis) generally have a better prognosis.
Surgical Decision:
Despite the presence of metastases, limb salvage surgery was deemed appropriate given the resectability of the primary tumor, the patient's young age, and the potential for long-term survival with aggressive treatment. The goals were:
*
Achieve wide oncological margins:
Essential for local disease control.
*
Preserve a functional limb:
Maximizing the patient's quality of life.
*
Address metastatic disease:
Surgical resection of oligometastatic pulmonary nodules would be considered if they remained stable or responded favorably to chemotherapy.
Enneking Surgical Staging System (Modified for Primary Malignant Bone Tumors):
This patient was classified as:
*
Grade (G):
G2 (High-grade tumor) based on biopsy showing highly malignant features.
*
Site (T):
T2 (Extra-compartmental tumor) due to the extensive extraosseous soft tissue component, involvement of multiple muscle groups, and proximity to the neurovascular bundle and joint capsule.
*
Metastasis (M):
M1 (Regional or distant metastasis) due to the presence of pulmonary nodules.
Therefore, the patient presented with a Stage III (G2, T2, M1) osteosarcoma of the distal femur. The decision for limb salvage surgery was made in conjunction with the multidisciplinary team, balancing oncological safety with functional outcome. The aggressive nature of osteosarcoma, even with metastases, warrants definitive surgical treatment of the primary tumor to prevent local recurrence, manage pain, and potentially improve overall survival in carefully selected cases.
Surgical Technique / Intervention
After completing three cycles of neoadjuvant chemotherapy, follow-up MRI demonstrated a good response, with reduction in tumor volume and decreased edema, though the extensive soft tissue component remained. The patient was cleared for limb salvage surgery.
Patient Positioning and Preparation:
The patient was positioned supine on the operating table. A high thigh tourniquet was applied but not inflated initially, allowing for assessment of vascularity and potential for en bloc resection without tourniquet effect on margins. The entire limb from the iliac crest to the toes was meticulously prepped and draped to allow full sterile access, including the possibility of extending the incision or performing a rotational flap if needed for soft tissue coverage. A sterile stockinette was applied.
Surgical Approach:
A longitudinal anterior incision was made, extending from the mid-thigh proximally, crossing the knee joint, and ending at the mid-tibia. This wide skin incision allowed for adequate exposure and a generous skin ellipse to be included if necessary for the wide margin. Subcutaneous tissues were carefully dissected. The deep fascia was incised longitudinally. The quadriceps mechanism was identified. The rectus femoris and vastus intermedius were carefully mobilized off the tumor's anterior aspect, while aiming to maintain a cuff of normal tissue around the tumor.
Resection of the Tumor:
1.
Identification of Neurovascular Bundle:
The popliteal fossa was carefully dissected to identify and protect the popliteal artery, vein, and tibial and common peroneal nerves. Due to the MRI finding of potential neurovascular encasement, meticulous dissection was performed. Intraoperative assessment revealed the tumor was intimately adherent but not directly invading the adventitia of the vessels, allowing for careful stripping and preservation of the neurovascular bundle by dissecting along a clear fascial plane.
2.
Soft Tissue Dissection:
Wide soft tissue margins were achieved by excising a block of muscle (vastus medialis, intermedius, and part of vastus lateralis) and surrounding fascia that were involved or in close proximity to the tumor. A cuff of normal tissue around the extraosseous component was maintained.
3.
Osteotomies:
Pre-operative templating guided the precise location of the proximal and distal osteotomies.
* The
proximal osteotomy
was performed at 14 cm from the articular surface, ensuring a 2 cm intramedullary clear margin beyond the MRI-demonstrated proximal tumor extent. This was confirmed by intraoperative frozen section analysis of the proximal marrow margin, which was negative for tumor cells.
* The
distal osteotomy
was performed through the proximal tibia, requiring resection of the entire knee joint due to tumor proximity and potential microscopic epiphyseal involvement. The menisci, cruciate ligaments, and collateral ligaments were sacrificed en bloc with the distal femur.
* An oscillating saw was used to perform clean, perpendicular osteotomies, with careful irrigation to prevent thermal necrosis.
4.
En Bloc Resection:
The resected specimen, consisting of the distal femur, knee joint capsule, patella (due to knee joint resection), and surrounding soft tissues (muscles, fascia), was removed en bloc. The specimen was oriented for the pathologist for accurate margin assessment.
Reconstruction with Modular Distal Femoral Endoprosthesis:
1.
Preparation of Femoral Canal:
The intramedullary canal of the remaining proximal femur was reamed sequentially to fit the stem of the modular endoprosthesis.
2.
Prosthesis Assembly:
A modular distal femoral endoprosthesis (e.g., Stryker Global Knee, Zimmer NexGen Rotating Hinge) was selected based on the templated length and patient anatomy. The prosthetic components, including the femoral stem, distal femoral component with a hinge mechanism, and a tibial tray with polyethylene insert, were assembled.
3.
Cementation:
The femoral stem was cemented into the prepared femoral canal using polymethylmethacrylate (PMMA) bone cement in a retrograde fashion. The tibial component was cemented onto the proximal tibia after careful preparation of the tibial plateau.
4.
Articulation:
The hinge mechanism of the endoprosthesis was articulated, creating a stable knee joint replacement. The limb length was restored, and tension in the surrounding soft tissues was assessed.
5.
Soft Tissue Repair:
The remaining vastus lateralis and medialis muscles were meticulously repaired to the components and surrounding soft tissues to provide muscle coverage and aid in stability. The retinaculum was closed where possible. Given the wide resection, specific muscle transfers might be considered to enhance function, though for a standard distal femur resection, direct reattachment is often employed.
Wound Closure:
Hemostasis was meticulously achieved. Two large-bore drains were placed in the deep and superficial layers. The subcutaneous tissue and skin were closed in layers with absorbable sutures and skin staples, respectively. A compressive dressing was applied, and the limb was placed in a knee immobilizer.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Period (Days 0-7):
*
Pain Management:
Multimodal analgesia including epidural catheter, patient-controlled analgesia (PCA) with opioids, and NSAIDs/acetaminophen.
*
Deep Vein Thrombosis (DVT) Prophylaxis:
Low molecular weight heparin (LMWH) or unfractionated heparin, along with sequential compression devices.
*
Wound Care:
Daily dressing changes, close monitoring for signs of infection or hematoma. Drains typically removed when output is minimal (<30-50 mL/24h).
*
Mobilization:
Strict bed rest for the first 24-48 hours. Early passive range of motion (PROM) using a continuous passive motion (CPM) machine, starting with 0-30 degrees and progressing as tolerated.
*
Weight Bearing:
Non-weight bearing (NWB) to the operative limb. Touch-down weight bearing (TDWB) may be initiated for balance with an assistive device (crutches or walker) under strict physical therapy supervision.
*
Adjuvant Chemotherapy:
Patient typically resumes adjuvant chemotherapy within 3-4 weeks post-surgery, once wound healing is satisfactory and systemic recovery permits.
Early Rehabilitation (Weeks 1-6):
*
Physical Therapy (PT):
Daily sessions focusing on:
*
Range of Motion:
Gradual progression of knee flexion and extension within protected limits. Target 0-90 degrees by 6 weeks.
*
Strengthening:
Gentle isometric quadriceps and hamstring exercises.
*
Gait Training:
Progression from NWB to partial weight bearing (PWB) with gradual increase in load, using assistive devices. Focus on proper gait mechanics.
*
Transfer Training:
Independent transfers from bed to chair.
*
Occupational Therapy (OT):
Assessment of activities of daily living (ADLs) and provision of adaptive equipment as needed.
*
Wound Management:
Continue monitoring for infection. Suture/staple removal at 2-3 weeks.
Intermediate Rehabilitation (Weeks 6-12):
*
Progression of Weight Bearing:
Gradual increase to full weight bearing (FWB) as pain allows and strength improves.
*
Strengthening:
Progression to isotonic exercises for quadriceps, hamstrings, and calf muscles. Begin closed-chain kinetic exercises.
*
Proprioception and Balance:
Exercises to improve balance and coordination, essential for functional recovery.
*
Functional Mobility:
Emphasis on improving walking distance, ascending/descending stairs, and navigating uneven surfaces.
Long-Term Rehabilitation (Months 3-12+):
*
Advanced Strengthening:
Progressive resistance training, incorporating functional movements.
*
Activity-Specific Training:
Tailored exercises for return to specific activities or sports, within the limitations of the endoprosthesis. Impact activities are generally discouraged.
*
Regular Follow-up:
*
Oncological:
Clinical examination, laboratory tests (CBC, LFTs, ALP), chest CT (every 3 months for the first 2 years, then less frequently), local MRI (every 6-12 months) for surveillance of recurrence/metastasis.
*
Orthopedic:
Radiographs of the operated limb (AP/Lateral) every 6 months for the first 2 years, then annually, to monitor for implant loosening, osteolysis, or heterotopic ossification.
Patient Education: Patients are counselled on the long-term care of their endoprosthesis, including avoiding high-impact activities, maintaining ideal body weight, and seeking immediate medical attention for any signs of infection (e.g., fever, warmth, redness, drainage). They are also informed about the risk of periprosthetic fracture, aseptic loosening, and late infection.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- Multidisciplinary Team (MDT) Approach: Essential for optimal management of osteosarcoma. This includes orthopedic oncology, medical oncology, radiation oncology, diagnostic radiology, and pathology. All major decisions, from diagnosis to treatment and surveillance, must be made in concert.
- Biopsy is Paramount: A well-planned, image-guided (CT or US) core needle biopsy is the gold standard. The biopsy tract must be within the planned surgical resection field to avoid tumor seeding and subsequent contamination of limb salvage planes. Incisional biopsy is reserved for specific cases but requires the same meticulous planning.
- Accurate Staging: Crucial for treatment planning. Beyond local imaging (X-ray, MRI, local CT), always perform chest CT for pulmonary metastases and a bone scan for skeletal metastases. PET-CT can provide additional systemic staging.
- Neoadjuvant Chemotherapy: Standard for high-grade osteosarcoma. It treats micrometastases, assesses tumor response (prognostic), and may downstage the primary tumor.
- Wide Oncological Margins: The primary goal of surgery. Achieving at least a 2 cm gross margin (intramedullary and extraosseous) is critical for local control. Intraoperative frozen sections of margins (especially intramedullary) are invaluable.
- Neurovascular Preservation: Meticulous dissection to protect vital neurovascular structures, as their sacrifice can lead to significant morbidity or necessitate amputation.
- Reconstructive Options: Understand the indications and contraindications for various limb salvage options (e.g., modular endoprosthesis, allograft, allograft-prosthesis composite, rotationplasty, biological reconstruction). Modular endoprostheses are common for large resections and provide immediate stability.
- Post-Operative Surveillance: Rigorous follow-up protocol with regular imaging (chest CT, local MRI) to detect local recurrence or distant metastases early.
Pitfalls:
-
Inadequate Biopsy:
- Misdiagnosis: Leading to inappropriate treatment.
- Contaminated Biopsy Tract: Placing the biopsy incision inappropriately (e.g., horizontally, or not in the planned surgical field) can seed tumor cells into previously uninvolved tissues, potentially compromising limb salvage or necessitating wider resection.
- Insufficient Tissue: Non-diagnostic biopsy requiring repeat procedure.
- Inadequate Staging: Missing metastases can lead to suboptimal treatment and poorer prognosis. Not performing a chest CT or bone scan is a critical error.
- Positive Surgical Margins: Results in a high risk of local recurrence, often requiring further surgery or radiation. Careful pre-operative templating and intraoperative margin assessment are essential.
- Compromised Neurovascular Structures: Poor planning or execution can lead to nerve injury, vascular compromise requiring bypass, or even necessitate amputation.
- Infection: A devastating complication in endoprosthetic reconstruction, often requiring multiple debridements, implant removal, and sometimes amputation. Meticulous aseptic technique and prophylactic antibiotics are crucial.
- Aseptic Loosening: A long-term complication of endoprostheses, particularly in young, active patients, necessitating revision surgery.
- Periprosthetic Fracture: Fractures around the implant can occur due to stress risers or trauma, often requiring complex revision.
- Poor Patient Compliance: Non-adherence to rehabilitation protocols, weight-bearing restrictions, or follow-up can lead to suboptimal functional outcomes or delayed detection of complications/recurrence.
- Ignoring Metastases: Even with metastatic disease at presentation, aggressive treatment of the primary tumor and resectable metastases (e.g., pulmonary metastasectomy) can offer a chance for long-term survival in osteosarcoma patients. Resectability must be carefully assessed.
