Peripheral Nerve Grafting: Principles, Donor Selection, and Microsurgical Techniques
Key Takeaway
Peripheral nerve grafting is indicated for segmental nerve defects where a tension-free primary neurorrhaphy cannot be achieved. Successful reconstruction relies on meticulous microsurgical technique, appropriate donor nerve selection, and tension-free coaptation. This guide details the biomechanics, indications, and step-by-step surgical execution of perineurial repairs, epineurial-perineurial neurorrhaphy, and the use of modern synthetic conduits and decellularized allografts for optimal sensory and motor recovery.
INTRODUCTION TO PERIPHERAL NERVE RECONSTRUCTION
The reconstruction of peripheral nerve injuries remains one of the most technically demanding challenges in operative orthopaedics and hand surgery. When a nerve is transected, the distal segment undergoes Wallerian degeneration, while the proximal stump attempts to regenerate via axonal sprouting. For functional recovery to occur, these regenerating axons must successfully cross the zone of injury and enter the endoneurial tubes of the distal stump.
While primary end-to-end neurorrhaphy is the gold standard for clean, sharp transections, high-energy trauma, crush injuries, or delayed presentations frequently result in a segmental nerve defect. In these scenarios, attempting to force a primary repair will inevitably introduce tension across the coaptation site.
Surgical Warning: Tension is the absolute enemy of nerve regeneration. Biomechanical studies demonstrate that an elongation of just 8% significantly impairs intraneural venular blood flow, and an elongation of 15% causes complete ischemia of the nerve. Ischemia leads to fibroblast proliferation, intraneural scarring, and an impenetrable barrier to axonal advancement.
Therefore, when a tension-free primary repair is impossible, the surgeon must bridge the gap using autogenous nerve grafts, synthetic conduits, or decellularized nerve allografts.
INDICATIONS FOR NERVE GRAFTING
At times, as a result of extensive tissue destruction, blast injuries, or the necessary resection of a terminal neuroma in delayed presentations, a segmental nerve defect is created. This defect often cannot be overcome through standard gap-management techniques such as nerve mobilization, joint flexion, or rerouting of the nerve.
The principal indication for nerve grafting in the upper extremity and hand is the bridging of defects after segmental nerve injury if a tension-free neurorrhaphy cannot be accomplished.
Less commonly seen, but equally critical, indications include:
* Providing innervation to free vascularized muscle grafts (e.g., functioning free muscle transfers for brachial plexus reconstruction).
* Innervating free neurovascular island flaps to restore protective sensation to critical contact areas.
* Reconstructing the brachial plexus following severe avulsion or rupture injuries.
PRE-GRAFTING CONSIDERATIONS AND GAP MANAGEMENT
Before committing to a nerve graft, the surgeon must exhaust all other techniques for closing small gaps between nerve endings. Autogenous grafts require sacrificing a donor nerve (resulting in donor site morbidity), and they introduce two coaptation sites that regenerating axons must cross, inherently reducing the final functional outcome compared to a single primary repair.
Techniques to manage small gaps include:
* Extensive Mobilization: Mobilizing the nerve ends over a distance of a few centimeters proximally and distally. This must be done carefully to preserve the segmental mesoneurial blood supply.
* Joint Positioning: Positioning the joints near the nerve injury in less-than-awkward, mildly flexed positions.
* Nerve Transposition: Transposing or changing the course of nerve endings (e.g., anterior submuscular transposition of the ulnar nerve at the elbow can yield several centimeters of length).
* Bone Shortening: In cases of combined severe skeletal and neural trauma (e.g., replantation), shortening the bone can simultaneously close the nerve gap.
Clinical Pearl: While joint flexion can assist in closing a gap, extreme flexion must be avoided. If a joint must be flexed beyond 30 degrees to achieve coaptation, the repair will likely fail due to traction injury when the joint is eventually mobilized postoperatively. In such cases, proceed directly to nerve grafting.
SOURCES OF AUTOGENOUS NERVE GRAFTS
When an autograft is required, the ideal donor nerve should be expendable, easily accessible, and anatomically matched to the fascicular topography of the recipient nerve. Donor nerves for nerve grafts in the upper extremities include:
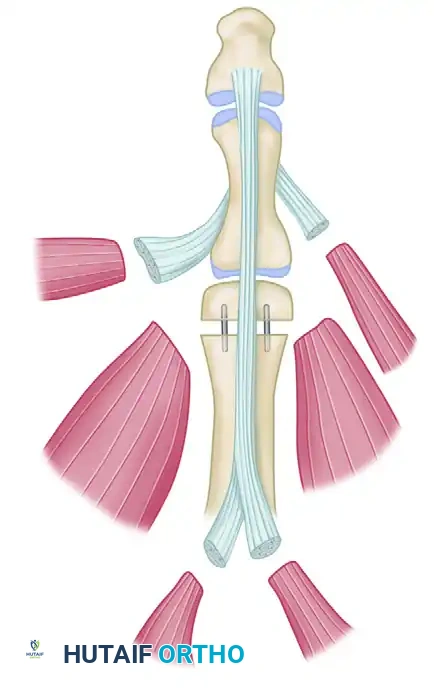
- The Sural Nerve: The workhorse of peripheral nerve grafting. It provides up to 30–40 cm of graft material and leaves a relatively benign sensory deficit on the lateral aspect of the foot. Anatomical studies have determined that the sural nerve best matches the common digital nerve.
- Lateral Antebrachial Cutaneous Nerve (LABC): Best matches the digital nerve proximal to the level of the distal trifurcation.
- Medial Antebrachial Cutaneous Nerve (MABC): Best matches the digital nerve distal to the trifurcation.
- Anterior and Posterior Interosseous Nerves: Useful for small, distal motor or sensory defects. The posterior interosseous nerve (PIN) in the distal forearm is frequently used for distal digital nerve gaps.
- Digital Nerves from Amputated Digits: In the setting of unsalvageable amputations, the digital nerves of the discarded part should be harvested as "spare parts" for grafting adjacent salvageable digits.
- Cross-Digit Harvesting: A segment of a severed nerve from the opposite, but less critical, side of a single digit can be used to repair the digital nerve on the primary contact side. For example, for lacerations of both nerves of the long finger requiring grafting, the ulnar digital nerve (non-contact border) can be sacrificed to graft the radial digital nerve gap (pinch contact border).
ALTERNATIVE NERVE GRAFT MATERIALS: CONDUITS AND ALLOGRAFTS
Alternatives to autogenous nerve grafts are increasingly utilized. Their use avoids sacrificing donor site sensation, eliminates donor site morbidity, and significantly saves surgical time.
Synthetic Conduits
Conduits act by isolating the regenerating nerve ends from surrounding scar tissue while allowing the accumulation of endogenous neurotrophic factors. This creates a localized, highly concentrated fluid microenvironment that guides the formation of a fibrin cable, across which Schwann cells and axons migrate.
Options include:
* Silicone Tubes: Historically used for digital nerve gaps of 5 mm or less, though largely replaced by bioabsorbable options due to the risk of chronic foreign body reaction or nerve compression requiring secondary removal.
* Polyglycolic Acid (PGA) Conduits: Highly effective for gaps less than 3 cm.
* Polycaprolactone Conduits: Absorbable poly(DL-lactide-e-caprolactone) conduits (e.g., Neurolac nerve guide, Polyganics B.V., Groningen, The Netherlands) are transparent, allowing direct visualization of the nerve ends during repair. Bertleff et al. reported that these conduits dissolve more slowly than PGA conduits, and their results indicated that nerve gaps of up to 2 cm could be successfully treated with this material.
* Vein Conduits: Autologous vein grafts, with or without intraluminal skeletal muscle (to provide a laminin-rich scaffold), have also been successfully used for short defects.
The Weber et al. Conduit Repair Technique
In a landmark randomized prospective study, Weber et al. found no significant differences among patients treated with primary repair, standard nerve grafting, or polyglycolic acid conduits for standard gaps. However, in patients with defects smaller than 4 mm, the results were actually better in the conduit repair group than in the primary repair group.
The technique dictates that the minimal distance between nerve stumps inside the conduit should be 5 mm, even in instances in which the ends can be coapted without tension. This 5 mm gap is theorized to optimize the neurotrophic fluid environment and prevent axonal crowding.

FIGURE 68-8 (Part 1): Weber et al. conduit repair technique. The proximal and distal nerve stumps are drawn into the bioabsorbable conduit using horizontal mattress sutures.
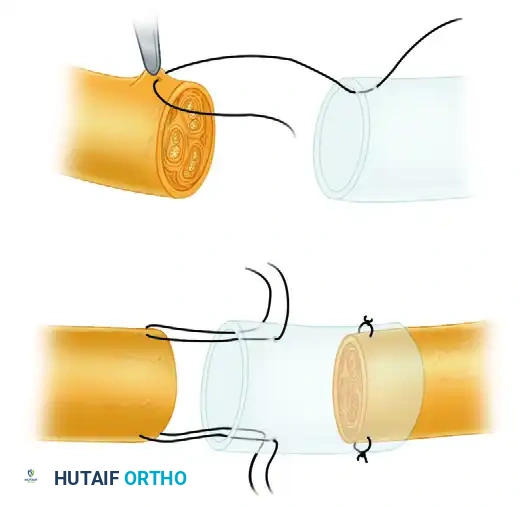
FIGURE 68-8 (Part 2): Minimal distance between nerve stumps is maintained at 5 mm, even in instances in which ends can be coapted without tension, to optimize the regenerative microenvironment.
Decellularized Nerve Allografts
Decellularized nerve allografts (e.g., Avance, AxoGen) are available in lengths up to 5 cm. These grafts undergo a proprietary chemical and radiation process that removes cellular material (eliminating immunogenicity and the need for systemic immunosuppression) while preserving the native three-dimensional endoneurial extracellular matrix. They are highly useful in restoring sensation for gaps up to 5 cm, bridging the gap between synthetic conduits (limited to short gaps) and autografts (which cause donor morbidity).
MICROSURGICAL TECHNIQUES IN NERVE GRAFTING
Meticulous microsurgical technique under operating microscope magnification is mandatory. The recipient nerve ends must be resected back to healthy, unscarred fascicular tissue (the "pouting" fascicle sign).
Pitfall: Failure to adequately resect the proximal neuroma or distal glioma is the most common cause of nerve graft failure. You must resect until healthy, bleeding fascicles are clearly visible under high magnification.
Perineurial (Fascicular) Neurorrhaphy
This technique is indicated when the nerve has a distinct, oligofascicular pattern (e.g., the distal median or ulnar nerves) where specific motor and sensory fascicular groups can be anatomically identified and matched.
Step A: The epineurium is carefully excised and stripped back a few millimeters to expose the underlying fascicles. This prevents epineurial tissue from invaginating into the repair site, which would block axonal growth.

Step B: A 10-0 or 11-0 nylon suture on a micro-spatula needle is passed through the perineurium of corresponding fascicles on either side of the cut surface of the nerve.

Step C: The neurorrhaphy is completed, usually requiring two 10-0 nylon sutures in each fascicle to ensure precise coaptation without buckling.

Epineurial-Perineurial Neurorrhaphy
This hybrid technique is highly effective for polyfascicular nerves. It utilizes the mechanical strength of the epineurium while ensuring precise alignment of the peripheral fascicles.
Step A: The epineurium is excised and retracted slightly. The first suture is placed through the epineurium, near a large, identifiable fascicle at the periphery of the nerve, and then passed directly through the perineurium of that specific fascicle.

Step B: The suture is then passed through the perineurium of the matching fascicle on the opposite side of the cut surface of the nerve, and finally out through the epineurium.

Step C: The repair is completed after suturing other suitably matched fascicles around the periphery. This technique provides excellent rotational alignment and mechanical stability.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a nerve graft relies heavily on the postoperative rehabilitation protocol.
- Immobilization: The extremity is immobilized in a bulky, non-compressive splint for 2 to 3 weeks. The joints are positioned to minimize any tension on the graft site, though extreme flexion is avoided.
- Progressive Mobilization: At 3 weeks, the splint is modified, and a supervised, progressive range-of-motion (ROM) program is initiated. Extension is regained gradually over the next 3 to 4 weeks to prevent sudden traction on the maturing fibrin cable and early regenerating axons.
- Sensory Re-education: Once advancing Tinel's signs indicate that axons have crossed the distal coaptation site and reached the target end-organs, a formal sensory re-education program is initiated. This involves cortical remapping exercises, starting with low-frequency vibration and progressing to texture discrimination and object identification.
- Motor Rehabilitation: For mixed or motor nerves, electrical stimulation of the denervated muscle may be considered to maintain muscle bulk while awaiting reinnervation, followed by biofeedback and strengthening once voluntary motor unit potentials return.
By adhering to strict microsurgical principles, respecting the biomechanics of tension, and selecting the appropriate autograft or synthetic conduit, the orthopaedic surgeon can optimize the environment for axonal regeneration and maximize functional recovery in severe peripheral nerve injuries.
You Might Also Like