Humeral Shaft IMN: Antegrade Technique Masterclass & Anatomical Pearls
Key Takeaway
Welcome to the OR, fellows. Today, we're tackling intramedullary nailing for humeral shaft fractures. This masterclass will guide you through meticulous preoperative planning, precise patient positioning, and the intricate, real-time execution of the antegrade technique. We'll delve into critical surgical anatomy, neurovascular protection, and the exact steps for optimal hardware placement, ensuring you master this essential procedure for successful patient outcomes.
Welcome, colleagues, to the operating theater. Today, we are going to dissect the nuances of antegrade intramedullary nailing (IMN) for humeral shaft fractures. This procedure, while seemingly straightforward in its fundamental mechanics, demands meticulous attention to detail, a profound understanding of three-dimensional surgical anatomy, and precise execution to achieve optimal outcomes and minimize devastating complications. Let us walk through this masterclass together, step by step, as if you are scrubbed in right beside me, navigating the complexities of the humeral diaphyseal corridor.
Comprehensive Introduction and Patho-Epidemiology
Humeral shaft fractures represent a significant and challenging portion of upper extremity trauma, accounting for approximately 3% to 5% of all skeletal fractures. Epidemiologically, they exhibit a classic bimodal distribution that dictates our clinical index of suspicion and treatment algorithms. We typically encounter these injuries in young males (21 to 30 years old) secondary to high-energy trauma such as motor vehicle collisions or sports injuries, and in older females (60 to 80 years old) resulting from low-energy mechanisms like simple falls from a standing height or rotational injuries superimposed on osteoporotic bone. Roughly 5% of these presentations involve open fractures, demanding immediate surgical prioritization, while the vast majority (around 63%) fall into the AO/ASIF type A classification, indicating simple, non-comminuted fracture patterns.
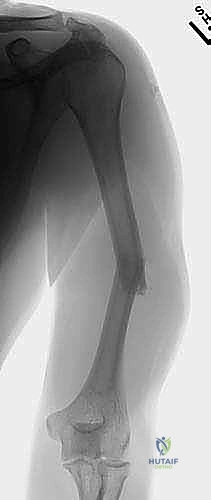
The AO/ASIF classification is not merely an academic exercise; it is crucial for understanding fracture morphology, predicting the biological and mechanical environment, and guiding our fixation strategies. Type A fractures are simple fractures (spiral, oblique, or transverse) where there remains significant cortical contact (greater than 90%) between the main proximal and distal fragments after reduction. Type B fractures involve a wedge or butterfly fragment, indicating some cortical contact between the main fragments but with a distinct, separate third fragment that compromises the load-sharing capacity of the bone. Type C fractures are complex or comminuted fractures, characterized by a complete lack of contact between the main fragments, almost universally the result of high-energy mechanisms, and present the greatest challenge for achieving stable fixation and predictable union.
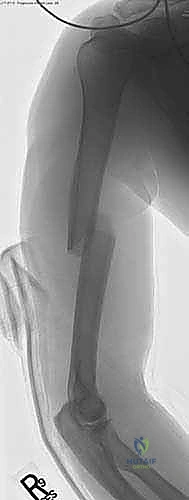
Intramedullary nailing offers a biologically compelling and biomechanically robust solution for stabilizing fractures spanning a specific anatomical safe zone: from approximately 2 cm distal to the surgical neck to about 3 cm proximal to the olecranon fossa. This specific range is absolutely critical. Fixation outside this zone risks inadequate purchase in the metaphyseal bone, hardware prominence, and catastrophic impingement on the glenohumeral or ulnohumeral joints. By respecting these boundaries, IMN leverages the mechanical advantage of a central axis implant while preserving the critical soft tissue envelope.
The evolution of humeral IMN has been driven by the desire to minimize the surgical footprint. Unlike traditional dynamic compression plating, which necessitates extensive soft tissue stripping and periosteal devascularization, IMN is a minimally invasive technique. It allows for indirect diaphyseal fracture reduction and metaphyseal fracture approximation, meticulously preserving the fracture hematoma. This hematoma is not merely a byproduct of injury; it is a biologically active milieu, rich in osteoprogenitor cells and osteoinductive cytokines, essential for the secondary bone healing cascade.
Detailed Surgical Anatomy and Biomechanics
Understanding the intricate and unforgiving anatomy surrounding the humeral shaft is paramount to preventing iatrogenic injury during IMN. The humerus is a long bone with several unique morphological characteristics that directly influence our surgical approach and implant selection. Unlike the diaphysis of the femur or tibia, which maintain a relatively consistent medullary canal before flaring widely at the metaphyses, the humeral medullary canal terminates abruptly at the metaphyseal-epiphyseal junction, not extending fully into the proximal epiphysis. The narrowest part of the canal, the isthmus, is typically located at the middle-to-distal third of the humerus, a stark contrast to the proximal-to-middle third location seen in lower extremity long bones.
The proximal two-thirds of the humeral canal is relatively cylindrical and accommodating. However, as we progress distally, the medullary canal rapidly tapers to a prismatic end within the diaphysis, transitioning sharply into dense, hard cortical bone. This contrasts sharply with the wide flare of the proximal metaphysis, which is composed of softer, yielding cancellous bone. This "trumpet shape" makes achieving a true, rotationally stable interference fit with an intramedullary nail exceptionally challenging. Consequently, proximal and distal static locking has become the absolute standard of care for humeral IMN to control both axial length and rotational alignment.
From a biomechanical standpoint, the humerus is remarkably well-enveloped in a robust muscular sleeve, which contributes to its generally excellent prognosis for healing in uncomplicated fractures. Intramedullary nails are positioned centrally, closer to the normal mechanical axis of the bone. This central placement allows them to function as highly efficient load-sharing devices when cortical contact is present. Because the bending stiffness of an implant is proportional to the fourth power of its radius, a centrally placed nail is subjected to significantly lower bending moments compared to an eccentrically placed plate. This inherent load-sharing capability significantly reduces the risk of fatigue failure of the implant and mitigates cortical osteopenia secondary to stress shielding.

The proximity of vital neurovascular structures to the humeral shaft necessitates extreme caution and precise anatomical knowledge. The axillary nerve is highly vulnerable during the proximal approach. It lies approximately 6.1 ± 0.7 cm from the proximal humerus, 1.7 ± 0.8 cm from the surgical neck, and 5 to 6 cm from the distal edge of the acromion. When performing an antegrade approach, a deltoid split should strictly not exceed 4 to 5 cm distally from the acromion to avoid denervating the anterior deltoid. Similarly, the radial nerve poses a constant threat along the shaft. It crosses the lateral intermuscular septum approximately 17.0 cm from the proximal humerus, 12.0 cm from the olecranon fossa, and 16.0 cm from the distal humerus. It spirals around the posterior humerus and pierces the lateral intermuscular septum to lie anteriorly in the distal third, making it exquisitely vulnerable during blind passage of instruments or distal locking procedures.
Exhaustive Indications and Contraindications
While we are focusing on operative fixation, a master surgeon must intimately understand when nonoperative management is the superior choice. The vast majority of non-displaced or minimally displaced humeral shaft fractures can heal successfully without surgery, with union rates routinely exceeding 90%. Common closed treatment modalities include the hanging arm cast (excellent for displaced, midshaft oblique or spiral fractures with shortening) and the functional brace (Sarmiento brace), which relies on hydrostatic compression of the soft tissue envelope to maintain alignment. Acceptable nonoperative alignment parameters are surprisingly generous: up to 3 cm of shortening, 30 degrees of varus/valgus angulation (though less tolerated in obese patients or those with pendulous breasts), and 20 degrees of anterior/posterior angulation.
However, specific clinical scenarios unequivocally demand surgical intervention. Absolute indications include open fractures requiring urgent debridement and stabilization, fractures associated with vascular injuries requiring repair, and the "floating elbow" (ipsilateral humeral and forearm fractures) which severely compromises the ability to utilize gravity-assisted closed management. Furthermore, fractures in polytraumatized patients often require IMN to facilitate early mobilization, ease of nursing care, and pulmonary toilet. Pathologic or impending pathologic fractures are perhaps the most universally agreed-upon indication for IMN, as the nail protects the entire length of the bone from subsequent failure.
The management of a humeral shaft fracture presenting with a concurrent radial nerve palsy remains one of the most debated topics in orthopedic trauma. Historically, a radial nerve palsy was considered an automatic indication for surgical exploration. Today, the paradigm has shifted. For closed fractures with a primary radial nerve palsy, observation is generally recommended, as upwards of 70-90% will spontaneously recover (neurapraxia or axonotmesis). However, if there is an obvious radial nerve palsy in an open fracture, a neurologic deficit following a penetrating stab injury, or a secondary palsy that develops after an attempted closed reduction (suggesting nerve entrapment in the fracture site, particularly in Holstein-Lewis type distal third spiral fractures), immediate surgical exploration and internal fixation are mandatory.
To synthesize our decision-making process, we must weigh the clinical variables carefully. Below is a comprehensive breakdown of indications and contraindications for Humeral IMN.
| Category | Specific Conditions | Rationale |
|---|---|---|
| Absolute Indications | Open fractures (Gustilo-Anderson I-IIIA) | Requires rigid stabilization for soft tissue management. |
| Associated vascular injury | Skeletal stability is prerequisite for vascular repair. | |
| Floating elbow | Loss of gravitational traction mechanism. | |
| Pathologic fractures | Prophylactic or therapeutic full-length bone protection. | |
| Relative Indications | Polytrauma / severe head injury | Facilitates early mobilization and nursing care. |
| Morbid obesity / pendulous breasts | High risk of varus malunion with functional bracing. | |
| Segmental fractures | High risk of nonunion with conservative care. | |
| Failure of conservative management | Progressive deformity or delayed union > 8-12 weeks. | |
| Contraindications | Active medullary infection | High risk of pan-osteomyelitis. |
| Extremely narrow medullary canal | Precludes passage of even the smallest diameter nails. | |
| Fractures < 2cm from surgical neck | Inadequate proximal fixation / risk of joint penetration. | |
| Fractures < 3cm from olecranon fossa | Inadequate distal fixation / risk of articular damage. |
Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning is the foundation of a successful intramedullary nailing procedure; battles in the operating room are frequently won or lost in the templating suite. A meticulous physical examination is non-negotiable. Documenting the exact baseline status of the radial, ulnar, and median nerves (both motor and sensory) is critical for medicolegal protection and postoperative comparison. Standard orthogonal anteroposterior (AP) and lateral radiographs of the humerus, including the shoulder and elbow joints, are mandatory. When obtaining these, move the C-arm or the patient's body rather than rotating the fractured limb, which causes excruciating pain and risks secondary neurovascular injury.
Digital templating utilizing contralateral, uninjured humerus radiographs is invaluable. This allows the surgeon to accurately estimate the required nail diameter and length. The goal is to select a nail that achieves a snug fit at the isthmus without causing iatrogenic comminution, and a length that spans the fracture adequately while remaining buried beneath the articular cartilage proximally and stopping short of the olecranon fossa distally. Traction radiographs under fluoroscopy can be utilized in the holding area for highly comminuted fractures to assess reducibility and restore length, preventing the common pitfall of over-distraction during surgery, which is a primary driver of nonunion.
Patient positioning is a critical step that dictates the ease of the entire operation. For antegrade nailing, the patient is typically positioned in a modified beach-chair position with the back elevated approximately 30 to 45 degrees. The patient must be positioned laterally enough on the table to allow the injured shoulder to clear the edge, providing unimpeded access for the C-arm and the reaming instruments. The head and neck must be securely stabilized in a neutral position. Alternatively, a supine position on a radiolucent flat table can be used, which simplifies distal locking but can make the proximal starting point more challenging to access.
Fluoroscopy setup must be confirmed prior to prepping and draping. The C-arm should be able to obtain perfect AP and lateral views of the proximal humerus, the fracture site, and the distal humerus without compromising the sterile field. Typically, the C-arm comes in from the head of the bed or from the contralateral side. The entire forequarter, from the base of the neck to the fingertips, is prepped and draped free. A sterile tourniquet is not used. The arm is draped in a manner that allows it to be freely manipulated across the chest for lateral imaging and adducted for proximal access.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach for antegrade nailing requires precision to protect the rotator cuff and the axillary nerve. A 3 to 5 cm anterolateral incision is made starting at the anterolateral tip of the acromion and extending distally. The deltoid muscle is split in line with its fibers. It is imperative not to extend this split beyond 5 cm from the acromion to avoid catastrophic denervation of the anterior deltoid via injury to the axillary nerve. The subdeltoid bursa is excised to expose the rotator cuff, specifically the supraspinatus tendon.
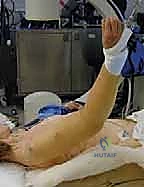
Identifying the correct starting point is arguably the most critical step of the procedure. The optimal entry portal is located at the apex of the humeral head, just medial to the greater tuberosity and posterior to the bicipital groove, directly in line with the medullary canal. A starting point that is too lateral (on the greater tuberosity) will force the nail into varus, potentially causing iatrogenic comminution of the medial cortex and a varus malunion. A longitudinal incision is made through the supraspinatus tendon (or precisely at the musculotendinous junction) to access the bone. An awl or a rigid guide pin is utilized to breach the cortex, confirming the trajectory with multi-planar fluoroscopy.
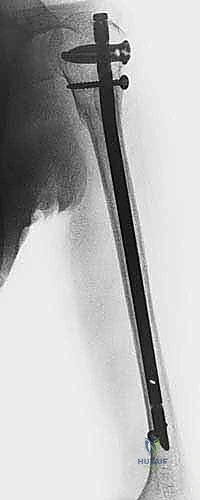
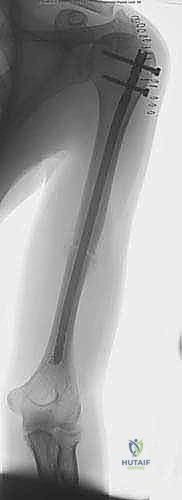
Once the medullary canal is accessed, a ball-tipped guide wire is passed down the proximal fragment. Closed reduction of the fracture is then performed. This often requires a combination of longitudinal traction, manipulation of the fragments via the soft tissue envelope, and sometimes the use of percutaneous joy-sticks (Schanz pins) or a temporary reduction tool. The guide wire is carefully advanced across the fracture site and seated centrally in the distal metaphysis, just proximal to the olecranon fossa. Reaming is then performed sequentially. While unreamed nails are an option, gentle reaming (0.5 to 1.0 mm larger than the selected nail diameter) increases the cortical contact area, provides autologous bone graft to the fracture site, and facilitates easier nail passage, reducing the risk of iatrogenic fracture.
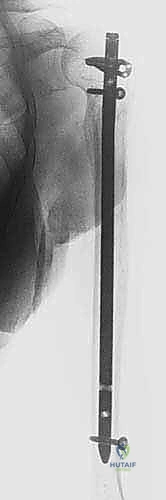
The selected intramedullary nail is assembled on the insertion jig and advanced over the guide wire. It is crucial to monitor the fracture site under fluoroscopy during insertion to ensure no distraction occurs. The proximal end of the nail must be countersunk beneath the articular cartilage of the humeral head (typically 2-4 mm) to prevent postoperative subacromial impingement, a leading cause of chronic shoulder pain. Proximal locking is performed through the targeting jig. Distal locking is performed using a freehand "perfect circle" technique under fluoroscopy. Care must be taken during anterior-to-posterior distal locking to protect the musculocutaneous nerve, and during lateral-to-medial locking to protect the radial nerve. Blunt dissection to the bone and the use of tissue protectors are mandatory during these steps.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, complications following humeral IMN can occur and require astute management. The most frequently cited complication is postoperative shoulder pain and dysfunction, reported in 10% to 30% of patients. The etiology is multifactorial, often stemming from proximal nail prominence causing subacromial impingement, iatrogenic injury to the rotator cuff during the entry portal creation, or the development of adhesive capsulitis. Prevention is paramount: strict adherence to the medial starting point, meticulous countersinking of the nail, and meticulous repair of the rotator cuff split are essential. If symptomatic hardware prominence occurs after fracture union, nail removal is indicated and typically resolves the impingement.
Delayed union and nonunion are significant concerns, occurring in approximately 5% to 10% of cases. Risk factors include severe fracture comminution, open fractures, smoking, and crucially, iatrogenic distraction of the fracture site during nail insertion. The humerus does not tolerate distraction well. If a nonunion develops (typically defined as lack of progression to healing at 6 months), salvage management usually involves exchange nailing (using a larger diameter reamed nail) or, more reliably, removal of the nail, compression plating, and application of autologous iliac crest bone graft.

Iatrogenic nerve injuries, particularly to the radial nerve, are devastating complications. The radial nerve is at highest risk during closed reduction maneuvers, passage of the reamer or nail across a distal third spiral fracture, and during lateral-to-medial distal locking. Incidence rates of iatrogenic radial nerve palsy range from 2% to 5%. If a nerve palsy is noted immediately postoperatively in a previously neurologically intact patient, the surgeon must have a low threshold for early exploration to ensure the nerve is not entrapped within the fracture site or tethered by a locking screw.
| Complication | Estimated Incidence | Primary Etiology | Salvage / Management Strategy |
|---|---|---|---|
| Shoulder Pain / Impingement | 10% - 30% | Prominent proximal nail; Rotator cuff injury. | Corticosteroid injections; Physical therapy; Hardware removal after union. |
| Nonunion / Delayed Union | 5% - 10% | Fracture distraction; Poor biology; Smoking. | Exchange nailing; Plate augmentation with autologous bone grafting. |
| Iatrogenic Radial Nerve Palsy | 2% - 5% | Entrapment during reduction; Injury during distal locking. | Observation (if confident nerve is free); Early exploration if entrapment suspected. |
| Iatrogenic Fracture Comminution | 3% - 8% | Eccentric starting point; Inadequate reaming; Forceful insertion. | Intraoperative cable/wire fixation; Conversion to long plate; Protected weight-bearing. |
| Infection | 1% - 3% | Open fractures; Poor soft tissue envelope. | Irrigation & Debridement; Suppressive antibiotics; Hardware removal if chronic. |
Phased Post-Operative Rehabilitation Protocols
The ultimate success of a humeral intramedullary nailing is heavily reliant on a structured, phased postoperative rehabilitation protocol. The primary goal is to maximize functional recovery of the shoulder and elbow while protecting the mechanical integrity of the healing fracture. The immediate post-operative phase (Weeks 0-2) focuses on pain control, edema management, and early mobilization of adjacent joints. The patient is typically placed in a simple sling for comfort. Active range of motion (ROM) of the fingers, wrist, and elbow is initiated immediately to prevent stiffness and facilitate venous return. Gentle, passive pendulum exercises for the shoulder are encouraged to prevent adhesive capsulitis, strictly avoiding any active abduction or forward elevation that would stress the rotator cuff repair.
Entering the intermediate phase (Weeks 2-6), clinical and radiographic evaluations are performed to assess early callus formation. As pain subsides, the patient is gradually weaned from the sling. Active-assisted ROM exercises for the shoulder are introduced, utilizing pulleys or a wand, focusing on restoring forward flexion and external rotation. Isometrics for the deltoid and rotator cuff can begin, provided they do not elicit pain at the fracture site. The surgeon must closely monitor for any signs of hardware failure or loss of fixation during this vulnerable period before robust bridging callus is present.
The advanced rehabilitation phase (Weeks 6-12) is contingent upon radiographic evidence of progressive union. Once clinical stability is achieved, the patient transitions to full active ROM and progressive resistance strengthening. Emphasis is placed on restoring the strength and endurance of the rotator cuff, deltoid, and periscapular stabilizers. Proprioceptive exercises and functional task simulation are integrated. Return to heavy manual labor or contact sports is generally delayed until there is unequivocal radiographic consolidation of the fracture (typically 3 to 6 months) and the patient has regained at least 80% to 90% of their contralateral limb strength and motion.
Summary of Landmark Literature and Clinical Guidelines
The contemporary management of humeral shaft fractures is heavily informed by a robust body of orthopedic literature. A landmark meta-analysis by Rommens et al. critically evaluated the outcomes of intramedullary nailing versus dynamic compression plating. While both techniques yield high union rates, the literature consistently highlights the biological advantages of IMN, specifically the preservation of the periosteal blood supply and the lower incidence of major soft tissue complications, albeit with a trade-off of higher rates of postoperative shoulder morbidity if the starting point is not perfectly executed.
The management algorithm for radial nerve palsies has been significantly shaped by the comprehensive review by Shao et al. Their work demonstrated that the vast majority of primary radial nerve palsies associated with closed humeral shaft fractures represent neurapraxia and will spontaneously resolve with expectant management over 3 to 6 months. This literature shifted the paradigm away from immediate, mandatory exploration for all palsies, reserving surgical nerve evaluation for specific indications such as open fractures, penetrating trauma, or secondary palsies developing after manipulation, where the risk of laceration or entrapment is exponentially higher.
Biomechanical studies have definitively established the critical nature of the proximal starting point. Research has shown that a starting point lateral to the greater tuberosity significantly increases the risk of iatrogenic medial cortex comminution and varus malalignment due to the mismatch between the nail's trajectory and the anatomical axis of the medullary canal. Current consensus guidelines, supported by the Orthopaedic Trauma Association (OTA), reinforce that while functional bracing remains a highly effective and acceptable standard of care for isolated, closed, low-energy fractures, antegrade intramedullary nailing is the gold standard for impending pathologic fractures, segmental patterns, and polytraumatized patients requiring immediate skeletal stability.
