Long Thoracic Nerve Palsy & Scapular Winging: Diagnosis, Anatomy & Biomechanics
Key Takeaway
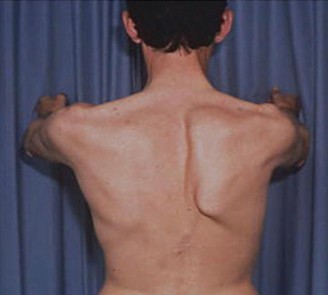
Long Thoracic Nerve (LTN) palsy results from injury to the LTN, often paralyzing the serratus anterior muscle. This crucial muscle stabilizes the scapula and enables protraction. Its dysfunction causes the scapula to protrude abnormally from the thoracic cage, a condition known as medial scapular winging, leading to significant functional impairment and pain.
Introduction & Epidemiology
Scapular winging, characterized by the abnormal protrusion of the scapula from the thoracic cage, is a debilitating condition that significantly impairs shoulder function and often causes considerable pain. While several etiologies can lead to this presentation, dysfunction of the long thoracic nerve (LTN) causing paralysis of the serratus anterior (SA) muscle is the most frequently encountered cause of isolated medial scapular winging. This condition severely compromises scapular stabilization, protraction, and upward rotation, profoundly impacting activities requiring overhead motion.
Epidemiologically, LTN palsy is relatively rare, with an estimated incidence of 1-10 cases per 100,000 population. However, its true prevalence may be underestimated due to missed diagnoses or spontaneous resolution. Common mechanisms of injury include:
*
Traction Injuries
: Most common, often seen after vigorous exercise, heavy lifting, or repetitive overhead activities (e.g., throwing sports, weightlifting).
*
Direct Trauma
: Blows to the lateral chest wall, penetrating injuries, or iatrogenic injuries during thoracic or axillary surgery (e.g., axillary lymph node dissection, mastectomy, chest tube insertion).
*
Compression
: Entrapment of the nerve, though less common, can occur from masses, accessory muscles, or fibrous bands.
*
Inflammatory/Viral
: Post-viral syndromes (e.g., Parsonage-Turner syndrome/brachial neuritis), often idiopathic in nature.
*
Idiopathic
: A significant proportion of cases lack a clear precipitating event, with spontaneous resolution noted in many.
Differential diagnoses for scapular winging extend beyond LTN palsy. Lateral scapular winging, characterized by a more prominent inferior scapular pole that shifts laterally, is typically indicative of trapezius muscle paralysis due to spinal accessory nerve (SAN) injury. Less common causes include dorsal scapular nerve palsy (rhomboid dysfunction), which presents with more subtle winging and difficulty retracting the scapula, or combined nerve injuries. Correct differentiation is paramount, as the specific nerve involved dictates the appropriate diagnostic workup and subsequent management strategy. This review will primarily focus on the diagnosis and surgical management of long thoracic nerve palsy.
Surgical Anatomy & Biomechanics
A thorough understanding of the intricate anatomy and biomechanics of the shoulder girdle is indispensable for the accurate diagnosis and effective management of scapular winging. The long thoracic nerve and its effector muscle, the serratus anterior, are central to scapular stability and kinematics.
The Long Thoracic Nerve (LTN)
- Origin : The LTN originates from the ventral rami of the C5, C6, and C7 nerve roots. Individual contributions vary, but C5 and C6 are typically dominant.
- Course : After emerging from the cervical spine, the nerve roots coalesce and descend through the posterior triangle of the neck. Critically, the LTN passes posterior to the brachial plexus and anterior to the scalenus medius muscle. This unique course renders it vulnerable to injury during neck hyperextension, lateral flexion, or direct trauma. As it enters the axilla, it travels along the lateral thoracic wall, lying on the external surface of the serratus anterior muscle, deep to the latissimus dorsi and external oblique fascia.
- Innervation : The LTN provides segmental motor innervation to each of the digitations of the serratus anterior muscle. Its lengthy, superficial course and tethering points make it susceptible to traction injuries during arm movements, particularly overhead activities, or compression along its path.
The Serratus Anterior (SA) Muscle
- Origin : The SA muscle originates from the outer surfaces of the upper eight to nine ribs. Its superior digitation arises from the first and second ribs, while its middle and inferior digitations originate from the second through ninth ribs.
- Insertion : The muscle inserts along the entire medial border of the scapula on its costal surface. The superior fibers insert onto the superior angle, the middle fibers onto the medial border, and the inferior fibers onto the inferior angle. The powerful inferior fibers are particularly crucial for upward rotation.
-
Actions
: The SA muscle is a primary mover and stabilizer of the scapula. Its key functions include:
- Protraction : Pulling the scapula anteriorly around the chest wall (e.g., during a push-up or punch).
- Upward Rotation : Working synergistically with the upper and lower trapezius during overhead elevation. This action positions the glenoid to maintain optimal alignment with the humeral head.
- Stabilization : Anchoring the scapula firmly against the posterior thoracic wall, preventing winging during arm movements.
Other Relevant Nerves and Muscles
- Spinal Accessory Nerve (SAN) : Innervates the trapezius muscle. Injury results in lateral scapular winging, shoulder droop, and weakness in shoulder elevation and retraction.
- Dorsal Scapular Nerve (DSN) : Typically arises from C5 and supplies the rhomboid major and minor, and sometimes the levator scapulae. Palsy leads to subtle medial winging, difficulty with scapular retraction, and pain.
- Pectoralis Major Muscle : While not directly involved in scapular winging as a cause, its sternal head is a common donor for muscle transfer procedures to stabilize the scapula in cases of irreparable LTN palsy. It is innervated by the medial and lateral pectoral nerves.
- Latissimus Dorsi Muscle : Innervated by the thoracodorsal nerve (TDN). The TDN is a crucial donor nerve for nerve transfer procedures to reinnervate the LTN.
Biomechanics of Scapular Winging
The scapula acts as the stable base for glenohumeral motion. Its precise kinematic control is achieved through the coordinated action of 17 muscles attaching to it.
*
LTN Palsy (Serratus Anterior Paralysis)
: Loss of SA function leads to a characteristic medial winging of the scapula. This occurs because the SA can no longer effectively hold the medial border of the scapula against the rib cage. The unopposed action of the rhomboids and levator scapulae, combined with the weight of the arm, causes the scapula to rotate internally and tilt anteriorly. This compromises the glenoid's position, leading to:
* Loss of upward rotation during arm elevation, limiting overhead reach.
* Loss of scapular protraction and stability, leading to a "pseudo-rotator cuff" weakness.
* Significant pain due to muscle fatigue (compensatory muscles), glenohumeral instability, and impingement.
*
SAN Palsy (Trapezius Paralysis)
: Presents as lateral scapular winging, with the entire scapula shifting inferiorly and laterally, and the inferior angle becoming prominent. Patients exhibit significant difficulty in elevating the arm, particularly above 90 degrees, and shoulder girdle depression.
*
DSN Palsy (Rhomboid Paralysis)
: Causes more subtle winging, as the rhomboids are primarily retractors. Patients may have difficulty actively retracting the scapula and may exhibit a slight upward rotation of the inferior pole.
Understanding these distinctions is crucial for clinical examination and electrodiagnostic evaluation to pinpoint the exact nerve and muscle involved, guiding appropriate surgical or non-surgical intervention.
Indications & Contraindications
The decision-making process for managing scapular winging due to long thoracic nerve palsy is multifaceted, balancing the potential for spontaneous recovery with the severity of symptoms and chronicity of the condition.
Non-Operative Indications
Conservative management is typically the first line of treatment, especially in acute presentations where the potential for nerve recovery is high.
- Acute Injuries (<6-12 months) : A period of observation is warranted, as many cases of neurapraxia or mild axonotmesis recover spontaneously, particularly those of idiopathic or post-viral etiology.
- Mild Symptoms/Good Functional Compensation : Patients with minimal pain, acceptable function, or who have developed effective compensatory strategies may not require surgical intervention.
- Electrodiagnostic Evidence of Reinnervation : EMG/NCS showing nascent motor unit potentials or decreasing fibrillation potentials indicates ongoing nerve recovery, supporting continued non-operative management.
- Neurapraxia or Axonotmesis : These nerve injury classifications carry a better prognosis for recovery compared to neurotmesis.
- Significant Medical Comorbidities : Patients with severe co-existing medical conditions that increase surgical risk may be managed conservatively, even if symptomatic.
- Patient Preference : Those unwilling to undergo surgery, after thorough counseling regarding prognosis and alternative options.
- Physical Therapy : A structured program focusing on scapular stabilization, core strength, postural correction, and pain management is crucial. Modalities such as TENS, heat, and massage may be used for symptom relief.
Operative Indications
Surgical intervention is considered for patients with persistent, debilitating symptoms that have failed to improve with a dedicated course of non-operative management. The optimal timing for surgery is critical and depends on the type of intervention planned.
- Failed Conservative Management (>6-12 months) : Persistent, severe pain, functional impairment, and cosmetic deformity despite dedicated physical therapy.
- Documented Denervation Without Reinnervation : EMG/NCS showing persistent denervation potentials and no signs of reinnervation after 6-12 months strongly suggests a poor prognosis for spontaneous recovery.
- Neurotmesis : Complete transection or severe crush injury of the nerve, where spontaneous recovery is unlikely.
- Identifiable Compressive Lesion : Surgical decompression is indicated if imaging (MRI, CT) reveals a tumor, cyst, or fibrous band compressing the LTN.
-
Specific Surgical Targets
:
- Nerve Transfers (e.g., Thoracodorsal to LTN) : Generally considered for chronic injuries (6-18 months) where direct repair is not feasible, but the serratus anterior muscle still shows some viability (not completely atrophied). Early intervention after failed conservative management improves outcomes.
- Muscle Transfers (e.g., Pectoralis Major to Scapula) : The preferred option for chronic, irreparable LTN palsies (>18-24 months post-injury), or when nerve transfers have failed, and the serratus anterior muscle is severely atrophied and fibrotic. This procedure provides mechanical stabilization rather than reinnervation of the original muscle.
Contraindications
- Acute Phase with Potential for Spontaneous Recovery : Premature surgical intervention can interfere with natural healing and lead to unnecessary procedures.
- Severe, Irreversible Muscle Atrophy/Fibrosis : If the serratus anterior muscle has been denervated for several years (typically >2-3 years), it may be beyond the point of effective reinnervation, even with nerve transfer. In such cases, muscle transfer is usually the only viable option for stabilization.
- Unrealistic Patient Expectations : Patients must be thoroughly counseled on the potential outcomes, recovery timeline, and possibility of residual deficits.
- Active Infection : Any active infection in the surgical field is an absolute contraindication.
- Inability to Participate in Post-Operative Rehabilitation : The success of surgical intervention heavily relies on patient adherence to a rigorous rehabilitation program.
Table: Operative vs. Non-Operative Indications
| Indication Feature | Non-Operative Management | Operative Management |
|---|---|---|
| Timing Post-Injury | Acute (0-6 months); up to 12 months for observation | Chronic (>6-12 months, depending on type) |
| Symptoms | Mild to moderate pain, manageable functional deficit | Severe, debilitating pain; significant functional loss |
| Electrodiagnostics | Neurapraxia, axonotmesis; signs of reinnervation | Neurotmesis; persistent denervation without reinnervation (>6-12 months) |
| Muscle Status | Healthy, viable SA muscle | Atrophy of SA muscle; potentially fibrotic (chronic) |
| Cause | Idiopathic, post-viral, traction (mild) | Compressive lesion, severe trauma, iatrogenic |
| Previous Treatment | Initial presentation; response to PT | Failed conservative management |
| Surgical Type | N/A | Nerve transfer (6-18 months); Muscle transfer (>18-24 months) |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is essential for optimizing surgical outcomes, minimizing complications, and ensuring patient safety.
Pre-operative Evaluation
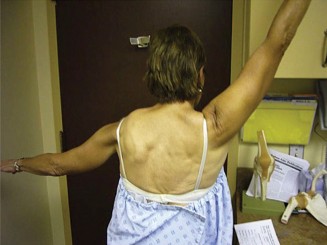
- Detailed History and Physical Examination : Crucial for identifying the extent of neurologic deficit, duration of symptoms, and impact on daily activities. Assess specific muscle weakness (SA, trapezius, rhomboids), range of motion, and pain generators. The "wall push-up test" is classic for demonstrating medial scapular winging.
-
Electrodiagnostic Studies (EMG/NCS)
: These are paramount.
- Nerve Conduction Studies (NCS) : Can identify conduction block or axonal loss in the LTN. However, the LTN is a pure motor nerve and can be challenging to test directly.
-
Electromyography (EMG)
: This is the definitive study. It assesses the serratus anterior, trapezius, rhomboids, and deltoid to differentiate LTN palsy from other conditions.
- Acute denervation : Fibrillation potentials and positive sharp waves (appear 2-4 weeks post-injury).
- Chronic denervation : Reduced recruitment of motor units, polyphasic potentials.
- Reinnervation : Nascent motor unit potentials, indicating ongoing recovery.
- EMG/NCS provides critical information on the severity of nerve injury (neurapraxia, axonotmesis, neurotmesis), the chronicity of the injury, and the potential for recovery, guiding the choice between nerve repair/transfer and muscle transfer.
-
Imaging Studies
:
- MRI of the Brachial Plexus and Shoulder : Essential for ruling out compressive lesions (tumors, cysts), assessing the integrity of the brachial plexus, and evaluating for muscle atrophy, fatty infiltration, or edema in the serratus anterior. Can sometimes visualize the LTN directly.
- Cervical Spine MRI : To exclude cervical radiculopathy or disc herniation mimicking nerve compression.
- CT Scan : May be useful for bony anomalies or to assess the rib cage if considering rib-based anchors for muscle transfers.
- Plain Radiographs : To rule out skeletal abnormalities or fractures.
Patient Education and Consent
- Thorough discussion of the proposed surgical procedure, including its rationale, potential benefits (pain relief, improved function, cosmetic improvement), and realistic outcomes.
- Detailed explanation of potential complications (infection, nerve injury, hematoma, failure of reinnervation/stabilization, donor site morbidity).
- Emphasis on the long and often arduous rehabilitation process, particularly for nerve transfers where functional return can take 12-24 months.
- Discussion of alternative treatments, including continued conservative management.
Pre-operative Medical Clearance
Standard medical clearance based on patient age, comorbidities, and the planned anesthetic technique.
Patient Positioning
Proper positioning is critical for surgical access, nerve protection, and ergonomic comfort for the surgeon.
*
Nerve Decompression/Proximal LTN Exploration (Supraclavicular)
:
*
Position
: Supine, head turned to the contralateral side. A shoulder roll may be placed under the ipsilateral shoulder to extend the neck and open the posterior triangle.
*
Arm
: Ipsilateral arm tucked or abducted slightly for comfort.
*
Distal LTN Exploration / Nerve Transfer (Thoracodorsal to LTN)
:
*
Position
: Lateral decubitus position is preferred. The patient is secured with beanbag positioning and chest/hip straps.
*
Arm
: The operative arm is prepped and draped freely across a sterile arm support or overhead suspension system (e.g., an arm holder or traction tower) to allow full range of motion intraoperatively for exposure and tension-free nerve coaptation. Adequate padding for all pressure points is crucial.
*
Pectoralis Major Transfer
:
*
Position
: Lateral decubitus or semi-Fowler position.
*
Arm
: The operative arm is prepped and draped freely, similar to nerve transfer, to allow manipulation and appropriate tensioning of the transferred muscle.
Anesthesia
General anesthesia is typically used. For nerve transfers, nerve stimulators can be used to identify specific nerves and branches. Intraoperative nerve monitoring (electromyography of target muscles) may be employed, especially for complex or revision cases, though its routine use for LTN transfer is debated.
Detailed Surgical Approach / Technique
Surgical management of long thoracic nerve palsy primarily involves either nerve reconstruction (decompression, neurolysis, or nerve transfer) or muscle transfer. The choice hinges on the chronicity of injury, the extent of nerve damage, and the viability of the serratus anterior muscle.
1. Nerve Decompression / Neurolysis
- Indication : Compressive neuropathies, often identified by MRI (e.g., fibrous bands, hypertrophied scalene muscles, mass lesions) or intraoperative findings.
-
Approach
: Varies based on the location of compression.
- Supraclavicular Approach (Proximal LTN) : An oblique incision parallel to the posterior border of the sternocleidomastoid, extending into the posterior triangle. Careful dissection through the skin, platysma, and investing fascia. The LTN is identified deep to the brachial plexus, lying on the scalenus medius. Any constricting bands or masses are meticulously released.
- Axillary/Lateral Thoracic Approach (Distal LTN) : An oblique incision along the anterior axillary line, or a vertical incision along the midaxillary line, allowing access to the nerve along the lateral chest wall.
- Technique : Once the LTN is identified, it is carefully mobilized. Adhesions, fibrous bands, or any extrinsic compressing structures are precisely released. Micro-surgical loupes or an operating microscope are often used to ensure complete neurolysis without nerve damage. Intraoperative nerve stimulation can confirm nerve function.
2. Nerve Transfer: Thoracodorsal Nerve (TDN) to Long Thoracic Nerve (LTN)
- Rationale : This is the most common and successful nerve transfer for LTN palsy. The TDN, typically a fascicle or branch to the latissimus dorsi (LD), is sacrificed to reinnervate the denervated LTN. The LD usually tolerates partial denervation without significant functional deficit due to its broad innervation and compensatory function. This procedure aims for dynamic reinnervation of the serratus anterior.
- Patient Position : Lateral decubitus with the ipsilateral arm draped free, allowing full mobility.
- Incision : An oblique incision is made along the posterior axillary line, extending inferiorly from the axilla towards the inferior angle of the scapula. This provides access to both the TDN and the LTN.
-
Dissection
:
-
Identify Latissimus Dorsi and Thoracodorsal Nerve
: The latissimus dorsi muscle is identified. The TDN and its associated thoracodorsal artery and vein enter the deep surface of the LD muscle. The nerve is typically identified near the lateral border of the muscle. Careful dissection proximally along the nerve helps to identify its individual branches to the LD. A suitable motor branch (often one of the more distal branches) is selected for harvest, ensuring minimal compromise to the remaining LD function.
- Image Context : This image could illustrate the anatomical relationship of the thoracodorsal nerve with the latissimus dorsi muscle, highlighting the region where the nerve branches are identified and harvested for transfer. The precise point of selection for a donor branch is critical.
- Identify Long Thoracic Nerve : The LTN runs superficially on the serratus anterior muscle, deep to the latissimus dorsi and external oblique fascia, along the midaxillary line. It is usually found by dissecting along the lateral thoracic wall, often tracing it proximally from its terminal branches on the SA. Careful blunt dissection with identification of the SA digitations helps locate the nerve.
- Prepare Recipient (LTN) : Once the LTN is identified, it is transected distally to the presumed point of injury or denervation. The nerve ends are trimmed back to healthy fascicles, ensuring optimal conditions for regeneration. The target site for coaptation on the LTN should be as proximal as possible to minimize regeneration distance.
- Prepare Donor (TDN) : The selected motor branch of the TDN is transected as distally as possible to maximize its length. This is crucial for achieving a tension-free repair.
-
Coaptation
: The TDN branch is meticulously coapted to the LTN under an operating microscope. A tension-free repair is paramount. This is typically an epineurial repair using fine, non-absorbable monofilament sutures (e.g., 9-0 or 10-0 nylon). If a direct, tension-free repair is not possible, a short nerve graft (e.g., sural nerve) may be considered, though direct coaptation is preferred for optimal regeneration.

- Image Context : This image could depict the micro-surgical coaptation of the thoracodorsal nerve branch to the long thoracic nerve, showing the fine sutures used for precise nerve repair under magnification.
-
Identify Latissimus Dorsi and Thoracodorsal Nerve
: The latissimus dorsi muscle is identified. The TDN and its associated thoracodorsal artery and vein enter the deep surface of the LD muscle. The nerve is typically identified near the lateral border of the muscle. Careful dissection proximally along the nerve helps to identify its individual branches to the LD. A suitable motor branch (often one of the more distal branches) is selected for harvest, ensuring minimal compromise to the remaining LD function.
- Closure : The wound is irrigated, and a drain may be placed. Layered closure of fascia, subcutaneous tissue, and skin.
3. Muscle Transfer: Pectoralis Major Transfer (Sahrmann Procedure)
- Rationale : This procedure is indicated for chronic LTN palsies (typically >18-24 months) or failed nerve reconstructions where the serratus anterior is irreversibly atrophied and fibrotic. It provides mechanical stabilization of the scapula rather than reinnervation of the original muscle.
- Patient Position : Lateral decubitus or semi-Fowler position, with the ipsilateral arm draped free.
- Incision : A curvilinear incision is made from the anterior axillary fold, extending distally towards the chest wall, and then curving posteriorly towards the inferior angle of the scapula. This can be a single incision or two separate incisions connected by a subcutaneous tunnel.
-
Dissection
:
- Harvest Pectoralis Major (PM) : The sternal head of the pectoralis major muscle is identified. It is carefully detached from its humeral insertion, ensuring its neurovascular pedicle (medial and lateral pectoral nerves, thoracoacromial vessels) remains intact. This is critical for muscle viability. The muscle is mobilized extensively from the chest wall, allowing for adequate excursion.
- Create Subcutaneous Tunnel : A subcutaneous tunnel is created using blunt dissection from the anterior chest wall inferior to the axilla, extending posteriorly towards the medial border or inferior angle of the scapula. This tunnel must be wide enough to accommodate the muscle belly without compression.
- Pass Muscle : The harvested pectoralis major muscle flap is carefully passed through the subcutaneous tunnel to the posterior aspect of the scapula.
- Re-insertion : The PM tendon is securely re-attached to the inferior angle or medial border of the scapula. This is typically achieved by drilling two or three small holes through the scapula (inferior angle or just superior to it along the medial border). Strong, non-absorbable sutures (e.g., FiberWire, Ethibond) are passed through the PM tendon and then through the drill holes in the scapula, and tied securely. The goal is to provide posterior and slightly lateral traction to counteract the medial winging. The tension of the transfer is adjusted to provide maximal scapular stability without over-tensioning.
- Closure : Layered closure with drainage.
4. Fascial Graft Sling (Less Common)
- Rationale : A salvage procedure for severe cases where nerve or muscle transfers are not feasible or have failed. It provides passive mechanical stability to the scapula.
- Technique : A strip of autogenous fascia lata (from the thigh) or allograft fascia is harvested. This strip is then used to create a sling, typically anchored from the ribs (e.g., 5th-7th ribs) to the medial border of the scapula, effectively tethering the scapula to the chest wall. While providing stability, it lacks dynamic function.
Complications & Management
Surgical interventions for scapular winging, like any complex procedure, carry inherent risks. A thorough understanding of potential complications, their incidence, and appropriate management strategies is crucial for academic orthopedic surgeons.
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (%) | Management / Salvage Strategy |
|---|---|---|
| Infection (SSI) | 1-5 | Superficial: Oral antibiotics, local wound care. Deep: IV antibiotics, surgical debridement, irrigation, wound vac. May require hardware removal if present. |
| Hematoma/Seroma | 5-10 | Small, asymptomatic: Observation, compression. Large, symptomatic: Aspiration (ultrasound-guided), surgical drainage/evacuation, re-exploration to identify and ligate bleeding vessels. |
| Nerve Graft/Repair Failure | 20-40 | Persistent denervation/no reinnervation: Re-exploration, revision nerve transfer (if feasible within timeframe), or consideration of muscle transfer (e.g., pectoralis major transfer) as a salvage procedure. |
| Donor Site Morbidity |
Thoracodorsal nerve harvest:
Mild latissimus dorsi weakness (rarely symptomatic in daily activities, <5%). Management: Physical therapy to compensate, strengthening of remaining LD.
Pectoralis major harvest: Mild chest wall asymmetry, weakness in specific adduction/internal rotation tasks (rarely significant). Management: PT, activity modification. |
|
| Persistent Winging/Pain | 20-50 | Incomplete correction: Intensive physical therapy focusing on scapular stabilization, core strength. If nerve transfer failed: Consider revision nerve transfer (if within timeframe) or conversion to muscle transfer. If muscle transfer failed: Revision muscle transfer, tension adjustment, or consider fascial sling. Pain management, including blocks or medication. |
| Adhesions/Scarring | Common | Physical therapy focusing on gentle ROM, soft tissue mobilization, scar massage. Rarely requires surgical tenolysis or adhesiolysis. |
| Scapular Dyskinesis | High | Even after successful surgery, subtle dyskinesis can persist. Management: Long-term physical therapy focusing on dynamic scapular control, proprioception, and motor re-education. |
| Neurovascular Injury | <1 | Intraoperative identification and repair of injured nerve/vessel. Requires immediate vascular surgery consult for arterial/venous injury. Careful dissection is key to prevention. |
| Hardware Complications | <1 | (Specific to muscle transfers with drill holes/suture anchors) Suture pull-out, osteolysis around anchors. Management: Revision fixation, hardware removal, or alternative fixation methods. |
| Brachial Plexus Injury | Rare | Careful anatomical dissection and identification of structures. Management: Depending on the severity, conservative management, or surgical repair/grafting if significant injury. |
| Pneumothorax | Rare | (Especially with rib-based procedures or deep dissection near chest wall) Intraoperative recognition and chest tube placement. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is as critical as the surgical procedure itself in achieving optimal functional outcomes for patients undergoing surgery for scapular winging. Protocols are tailored to the specific surgical intervention performed (nerve vs. muscle transfer) and progress through distinct phases.
General Principles
- Protection : Safeguarding the surgical repair site from undue stress or tension.
- Gradual Progression : Slowly advancing activity levels and range of motion to allow for tissue healing and adaptation.
- Pain Management : Effective control of post-operative pain to facilitate participation in therapy.
- Patient Education : Continual reinforcement of restrictions, exercises, and expected timeline.
- Focus : Restoration of glenohumeral and scapulothoracic kinematics, strengthening of the entire shoulder girdle, and functional retraining.
Phase I: Immobilization & Early Protection (0-4/6 Weeks)
- Goal : Protect the surgical repair, control pain and swelling, initiate early passive motion.
-
Immobilization
:
- Nerve Transfer : Shoulder immobilizer or sling with an abduction pillow, worn continuously (except for hygiene) for 4-6 weeks to prevent tension on the nerve coaptation.
- Muscle Transfer : Shoulder immobilizer, often for 4-6 weeks, to protect the muscle re-insertion site. For pectoralis major transfer, avoid excessive shoulder abduction and external rotation.
-
Activities
:
- Passive Range of Motion (PROM) : Gentle, controlled PROM of the glenohumeral joint within protected limits , avoiding external rotation >0-30 degrees and flexion/abduction >90 degrees. This is often performed by the therapist or with patient assistance (e.g., pulley system) but without active muscle contraction of the operated side .
- Elbow, Wrist, Hand : Active ROM exercises to prevent stiffness.
- Posture : Education on maintaining good posture and avoiding slumping.
- Pain & Edema Control : Ice, elevation, gentle massage.
- Scapular Mobilization : Gentle, passive scapular glides without stressing the repair.
Phase II: Controlled Active Motion & Early Strengthening (4/6-12 Weeks)
- Goal : Gradually restore active range of motion, begin light strengthening, and initiate neuromuscular control.
- Sling Discontinuation : Gradually wean off the sling as pain allows and muscle control improves.
-
Range of Motion
:
- Active-Assisted ROM (AAROM) : Progress from PROM to AAROM for shoulder flexion, abduction, and rotation.
- Active ROM (AROM) : Advance to pain-free AROM.
-
Strengthening
:
- Isometrics : Gentle isometric contractions of the rotator cuff (subscapularis, infraspinatus, teres minor), deltoid, and periscapular muscles excluding the directly operated or transferred muscle .
- Scapular Stabilization : Focus on exercises for the uninjured scapular muscles (e.g., lower trapezius, rhomboids) to promote scapular control.
- Nerve Glides : For nerve transfer patients, gentle upper limb nerve gliding exercises may be introduced to promote nerve mobility.
- Neuromuscular Re-education : Emphasis on controlled movements, proper movement patterns, and avoiding compensatory strategies.
- Light Resistance : Begin with light resistance bands or bodyweight exercises for non-operative muscles.
Phase III: Progressive Strengthening & Functional Integration (12 Weeks - 6 Months)
- Goal : Achieve full, pain-free ROM, progressive strengthening of all shoulder girdle muscles, and integrate functional movements.
-
Strengthening
:
- Progressive Resistance Exercises : Utilize weights, resistance bands, and machines to strengthen the entire shoulder girdle.
-
Targeted Strengthening
:
- Nerve Transfer : Specific exercises to facilitate serratus anterior reinnervation and strengthening (e.g., wall slides, push-up plus, rhythmic stabilization). Visual feedback and EMG biofeedback can be valuable. Note: Clinical reinnervation of the serratus anterior typically takes 6-12 months, sometimes longer.
- Muscle Transfer : Focus on strengthening the transferred pectoralis major in its new role as a scapular stabilizer, while also strengthening other periscapular muscles.
- Eccentric Control : Emphasize eccentric strengthening to improve dynamic stability.
-
Functional Training
:
- Incorporate activities of daily living (ADLs) and work-specific tasks.
- Begin sport-specific drills for athletes.
- Focus on overhead mechanics and proper lifting techniques.
Phase IV: Return to Activity (6 Months - 1 Year+)
- Goal : Gradual and safe return to full, unrestricted activities, including sports and heavy labor.
- Return to Activity : Based on functional assessment, strength testing (isokinetic if available), and patient-reported outcomes.
- Nerve Transfer Specific : Continued gains in strength and function can occur for up to 2-3 years post-operatively as nerve regeneration matures.
- Maintenance Program : Ongoing home exercise program to maintain strength, flexibility, and scapular control.
- Monitoring : Regular follow-up with the surgeon and therapist to monitor progress and address any residual issues.
Summary of Key Literature / Guidelines
The management of scapular winging, particularly due to long thoracic nerve palsy, has evolved significantly, with modern surgical techniques demonstrating improved outcomes over historical approaches. Current literature emphasizes a multidisciplinary approach and individualized treatment based on the etiology, chronicity, and patient's functional demands.
Diagnostic Cornerstones
- Clinical Examination : The "wall push-up test" remains the gold standard for identifying medial scapular winging. Observing the scapular kinematics during active shoulder movements is critical.
- Electrodiagnostic Studies (EMG/NCS) : Indispensable for confirming the diagnosis of LTN palsy, quantifying the severity of nerve injury (neurapraxia, axonotmesis, neurotmesis), assessing the viability of the serratus anterior muscle, and monitoring for signs of reinnervation. Serial EMGs are vital in the acute phase to guide timing of intervention.
- Imaging (MRI) : MRI of the brachial plexus and shoulder is crucial to rule out compressive lesions, evaluate nerve integrity, and assess muscle atrophy or fatty infiltration of the serratus anterior, which helps in predicting the potential for muscle reinnervation.
Conservative Management
- Initial Approach : For acute injuries (typically <6 months), and especially for idiopathic or post-viral cases, conservative management with physical therapy and observation is the recommended first-line treatment.
- Physical Therapy Focus : Scapular stabilization, core strengthening, postural re-education, and pain management. The goal is to maximize the function of compensatory muscles while awaiting potential spontaneous LTN recovery.
- Spontaneous Recovery : Reported rates vary widely (40-90%), often dependent on the etiology and severity of the initial injury. Most recovery occurs within the first 12-18 months.
Surgical Intervention
The choice of surgical procedure is highly dependent on the chronicity of the injury and the status of the serratus anterior muscle.
-
Nerve Reconstruction (Decompression or Nerve Transfer)
:
- Timing : Preferred for injuries where the serratus anterior is still viable but spontaneous recovery has failed (typically 6-18 months post-injury).
- Nerve Decompression/Neurolysis : Indicated for identified compressive neuropathies. Outcomes are generally good with resolution of compression.
- Nerve Transfer (Thoracodorsal Nerve to Long Thoracic Nerve) : Considered the procedure of choice for dynamic reinnervation of the serratus anterior when direct nerve repair is not feasible. Studies by Wood et al., Elhassan et al., and others have demonstrated high rates of improved scapular stability, pain relief, and functional gains, often with 60-80% good to excellent results. While full strength recovery is rare, significant improvement in overhead activities and reduction of winging are common. Functional recovery can be slow, spanning 12-24 months or more.
-
Muscle Transfer (Pectoralis Major Transfer - Sahrmann Procedure)
:
- Timing : The workhorse procedure for chronic, irreparable LTN palsies (>18-24 months post-injury), or in cases where nerve reconstruction has failed or is contraindicated due to severe serratus anterior atrophy and fatty infiltration.
- Rationale : Provides a static and dynamic mechanical tether to stabilize the scapula to the chest wall.
- Outcomes : Generally provides excellent scapular stability and significant pain relief. However, the transferred muscle may not fully replicate the dynamic function of the serratus anterior, and some limitations in overhead motion may persist. Overall patient satisfaction and functional scores are often very good.
-
Fascial Graft Sling
:
- Timing : Reserved for complex or salvage situations where nerve and muscle transfers are not feasible or have failed.
- Outcomes : Provides passive stability but lacks dynamic muscle function. Often associated with less robust functional improvement compared to nerve or muscle transfers.
Outcomes and Current Guidelines
- A recent systematic review suggests that both nerve transfers and pectoralis major transfers are effective treatments for LTN palsy, offering significant improvements in pain and function. Nerve transfers tend to yield better dynamic results if performed within the optimal window.
- The decision between nerve and muscle transfer should be guided by EMG findings (presence/absence of reinnervation potentials, muscle viability), chronicity of injury, patient age, and functional goals.
- A multidisciplinary team approach involving orthopedic surgeons, neurologists, and physical therapists is recommended for optimal patient care, from diagnosis through rehabilitation.
- Long-term follow-up is crucial as functional improvements, especially after nerve transfers, can continue for several years.
You Might Also Like