Etiology, Clinical Evaluation, and Management of Peripheral Nerve Injuries
Key Takeaway
Peripheral nerve injuries in orthopedic trauma demand meticulous clinical and electrodiagnostic evaluation. Mechanical trauma, including gunshot wounds and long bone fractures, frequently causes neurapraxia, axonotmesis, or neurotmesis. Accurate assessment requires deep anatomical knowledge to avoid diagnostic pitfalls like trick movements. Electrodiagnostic studies, particularly nerve conduction velocity and electromyography, provide objective evidence of Wallerian degeneration and nerve continuity, guiding surgical decision-making and prognostic expectations in complex musculoskeletal trauma.
INTRODUCTION TO PERIPHERAL NERVE INJURIES
Peripheral nerve injuries (PNIs) represent a formidable challenge in orthopedic trauma and reconstructive surgery. The functional outcome of a salvaged limb is often dictated not by the osseous union, but by the recovery of the peripheral nervous system. While peripheral nerves can be compromised by a myriad of systemic conditions—including metabolic disorders, collagen vascular diseases, malignancies, and endogenous or exogenous toxins—the orthopedic surgeon is primarily concerned with injuries resulting from thermal, chemical, or mechanical trauma.
Every patient presenting with trauma to an extremity or limb girdle must undergo a rigorous, systematic evaluation for concomitant musculoskeletal, vascular, and peripheral nerve damage. Failure to identify a peripheral nerve injury at the time of initial presentation can lead to devastating functional deficits, severe medicolegal ramifications, and missed windows for optimal surgical intervention.
Clinical Pearl: The initial trauma assessment must always include a documented neurovascular examination before and after any manipulation, reduction, or splinting of a fracture or dislocation. An undocumented nerve is presumed to be an unexamined nerve.
ETIOLOGY AND PATHOMECHANICS OF MECHANICAL TRAUMA
Mechanical trauma to peripheral nerves can be broadly categorized into lacerations (sharp or blunt), traction/stretch injuries, compression, and high-energy concussive injuries. The biomechanical properties of peripheral nerves allow them to withstand a certain degree of stretch (up to 8-10% of their resting length) before structural failure of the endoneurial tubes and axonal disruption occurs. Beyond this physiological limit, the intraneural microcirculation is compromised, leading to ischemia, edema, and subsequent fibrosis.
Gunshot Wounds and Ballistic Trauma
Gunshot wounds (GSWs) are frequently complicated by peripheral nerve injuries. The pathophysiology of nerve injury in ballistic trauma is highly dependent on the velocity of the projectile.
- Low-Velocity Injuries (e.g., civilian handguns): These typically cause nerve damage through direct laceration or localized contusion.
- High-Velocity Injuries (e.g., military rifles): These projectiles create a massive temporary cavitational effect. The kinetic energy transfer produces a concussive shockwave that can severely stretch and contuse nerves located centimeters away from the actual bullet tract.
Spontaneous recovery is expected in over 50% of peripheral nerve injuries associated with gunshot wounds. The expected time to recovery ranges from 3 to 9 months. High-velocity injuries inherently take longer to heal due to the extensive zone of injury and severe intraneural fibrosis. In ballistic trauma, neurapraxia (temporary conduction block without axonal disruption) and axonotmesis (axonal disruption with intact supporting stroma) occur with roughly equal frequency. Neurotmesis (complete nerve transection) is relatively rare unless there is a direct hit by the projectile.
Surgical Warning: Immediate exploration of a nerve injury associated with a high-velocity gunshot wound is generally contraindicated unless there is an expanding hematoma or concomitant vascular injury requiring repair. The extensive zone of injury makes it impossible to accurately determine the viable margins of the nerve for early grafting.
Fracture and Joint Dislocation Associations
Bone or joint injuries are intimately associated with peripheral nerve lesions. Primary injury to a peripheral nerve may result directly from the initial traumatic force. However, secondary neural injuries are equally critical and can be caused by:
1. Displaced osseous fragments impaling or lacerating the nerve.
2. Severe stretching over a dislocated joint (e.g., axillary nerve in anterior shoulder dislocations).
3. Iatrogenic injury during closed manipulation, reduction, or surgical fixation.
Based on the classic epidemiological data reported by Spurling (analyzing 300 cases of long bone fractures with nerve involvement), the frequency of specific nerve injuries follows distinct anatomical patterns:
Upper Extremity (74% of total cases):
* Humerus Fractures: Radial nerve (70%), Ulnar nerve (22%), Median nerve (8%). The radial nerve is exceptionally vulnerable as it traverses the spiral groove and pierces the lateral intermuscular septum.
* Radius and/or Ulna Fractures: Ulnar nerve (41%), Radial nerve (35%), Median nerve (24%).
Lower Extremity (20% of total cases):
* Femur Fractures: Complete sciatic nerve (60%), Tibial component only (20%), Peroneal component only (20%).
* Tibia and/or Fibula Fractures: Peroneal nerve (70%), Both nerves (23%), Tibial nerve (7%). The common peroneal nerve is highly susceptible to stretch and compression as it winds around the fibular neck.
ELECTRODIAGNOSTIC EVALUATION
Electrodiagnostic studies—comprising Nerve Conduction Studies (NCS) and Electromyography (EMG)—are indispensable extensions of the clinical examination. They provide objective, quantifiable evidence of nerve conductivity, the severity of the insult, and the progression of recovery.
Timing and Pathophysiology of Nerve Degeneration
The timing of electrodiagnostic testing is critical. Immediately following an injury, it is physiologically impossible to determine the ultimate severity of the axonal insult.
When a nerve is stimulated proximal to, distal to, and across the level of injury, the responses vary based on the time elapsed since the trauma:
* Immediate Phase (0-5 days): Stimulation proximal and distal to the insult will elicit a normal Compound Muscle Action Potential (CMAP) and Sensory Nerve Action Potential (SNAP) in the distal segment, even if the nerve is completely transected. This occurs because the distal axonal segment has not yet undergone structural degradation. Stimulation across the injured segment, however, will show a conduction block.
* Wallerian Degeneration Phase (5-10 days): As Wallerian degeneration ensues, the axoplasm and myelin sheath distal to the injury degrade. There is a progressive reduction in the amplitude and an alteration in the configuration of the evoked potentials. By day 10, if axonal continuity is lost (axonotmesis or neurotmesis), the distal nerve segment becomes completely inexcitable.
Differentiating Neurapraxia from Axonal Loss
If the traumatic insult produced only a temporary physiological block (neurapraxia), the structural integrity of the axon remains intact. In this scenario, conductivity distal to the lesion will remain completely normal even after 10 to 14 days. This finding carries a highly favorable prognosis, indicating that spontaneous recovery will occur once the local myelin is repaired (typically within weeks to a few months).
Diagnostic Pitfall: Ordering an EMG/NCS within the first 72 hours of a closed nerve injury provides little prognostic value regarding axonal loss. Baseline studies are generally deferred until 3 to 4 weeks post-injury, at which point fibrillation potentials and positive sharp waves (indicating muscle denervation) will be clearly visible on needle EMG.
CLINICAL EVALUATION OF PERIPHERAL NERVES
A profound understanding of peripheral neuroanatomy, including common anatomical variations (e.g., Martin-Gruber anastomoses), is non-negotiable for the orthopedic surgeon. The clinical evaluation must be meticulous, isolating specific sensory zones and individual muscle bellies.
Sensory Examination and Autonomic Function
Sensory evaluation should map the autonomous zones of the peripheral nerves—areas where there is no overlapping innervation from adjacent nerves.
* Median Nerve: Volar tip of the index finger.
* Ulnar Nerve: Volar tip of the little finger.
* Radial Nerve: Dorsal first web space.
* Axillary Nerve: Lateral aspect of the proximal arm (regimental badge area).
* Common Peroneal Nerve: Dorsum of the first web space (deep peroneal) and lateral aspect of the leg/dorsum of the foot (superficial peroneal).
Furthermore, autonomic (sympathetic) function must be assessed. Denervated skin loses its sudomotor function, resulting in an absence of sweating (anhidrosis). This can be clinically observed, palpated (the skin feels unusually smooth and dry), or measured using the Ninhydrin test or by assessing skin resistance (which increases in denervated areas).
Motor Evaluation and the Deception of Trick Movements
Evaluation of motor loss is the cornerstone of PNI assessment. However, this evaluation is only accurate if the examiner physically palpates or directly visualizes the specific tendon or muscle belly under consideration. Relying solely on the gross analysis of joint movement is a dangerous trap due to "trick movements" and muscle substitution.
Common Trick Movements to Avoid:
1. Thumb Opposition: Many patients can accomplish apparent opposition of the thumb to the little finger even when the median nerve is completely severed and the opponens pollicis is paralyzed. This is achieved by the ulnar-innervated deep head of the flexor pollicis brevis and the adductor pollicis. Solution: Palpate the opponens pollicis muscle belly over the thenar eminence during the maneuver.
2. Wrist Extension: The wrist can be partially extended even when the radial nerve is completely paralyzed. This is accomplished by forceful, simple flexion of the fingers, which utilizes the tenodesis effect to pull the wrist into extension. Solution: Test wrist extension with the fingers completely relaxed or passively extended, and palpate the extensor carpi radialis longus/brevis.
3. Elbow Flexion: The elbow can be forcefully flexed even when the musculocutaneous nerve is completely severed and the biceps/brachialis are paralyzed. This is achieved by substitution of the radial-innervated brachioradialis. Solution: Palpate the biceps tendon and muscle belly during resisted supination and flexion.
While some muscles cannot be easily tested by palpation (e.g., lumbricals, short adductor of the thumb, interossei except the first dorsal), there are sufficient superficial muscles supplied by each nerve to allow for an accurate diagnosis.
Grading Muscle Strength
A clinical assessment of muscle strength must be standardized. The scale recommended by Highet (and adopted by the Medical Research Council - MRC) is universally accepted in orthopedic and neurosurgical literature:
- Grade 0: Total paralysis; no evidence of contractility.
- Grade 1: Muscle flicker or trace of contraction; no joint movement.
- Grade 2: Muscle contraction resulting in joint movement with gravity eliminated.
- Grade 3: Muscle contraction resulting in joint movement against gravity.
- Grade 4: Muscle contraction against gravity and against some external resistance.
- Grade 5: Normal muscle contraction against full resistance, compared with the contralateral side.
SURGICAL MANAGEMENT AND DECISION MAKING
The management of peripheral nerve injuries requires a delicate balance between allowing time for spontaneous recovery and intervening before irreversible motor endplate degeneration occurs (typically 12 to 18 months post-injury).
Indications for Surgical Exploration
- Immediate Exploration: Indicated for open injuries with sharp lacerations (e.g., knife wounds, glass cuts) where nerve transection is highly probable. Immediate primary repair yields the best functional outcomes. It is also indicated when a nerve injury is associated with a vascular injury requiring repair, or in the presence of an expanding hematoma or compartment syndrome.
- Early Exploration (within weeks): Indicated for iatrogenic injuries where the nerve was known to be intact preoperatively but is completely non-functional postoperatively, particularly if the surgical approach placed the nerve at high risk of entrapment or transection.
- Delayed Exploration (3 to 6 months): The standard of care for closed traction injuries, crush injuries, and gunshot wounds. If clinical and electrodiagnostic evaluations show no signs of reinnervation by 3 to 4 months, surgical exploration is warranted.
Surgical Approaches and Biomechanics of Repair
When exploring a peripheral nerve, the surgical approach must utilize extensile incisions that allow for identification of the nerve in pristine, uninjured tissue both proximal and distal to the zone of injury.
- Positioning and Tourniquet: The patient must be positioned to allow access to the entire limb. A pneumatic tourniquet is utilized to maintain a bloodless field, but it should be deflated prior to final hemostasis and nerve repair to ensure the intraneural microvasculature is intact and to prevent ischemic compounding of the nerve injury.
- Neurolysis: If the nerve is found in continuity but encased in scar tissue, an external neurolysis (freeing the nerve from surrounding scar) is performed. Internal neurolysis is rarely indicated as it risks damaging the delicate interfascicular plexus.
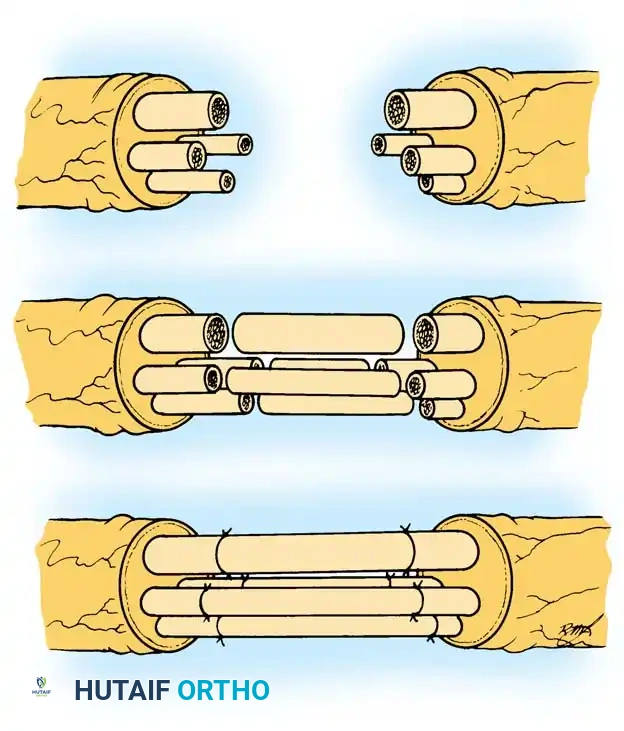
- Nerve Repair: If the nerve is transected, the neuromas must be resected back to healthy fascicles (the "bread loafing" technique). The repair must be absolutely tension-free. Tension across a nerve repair causes ischemia and catastrophic failure of axonal regeneration.
- Nerve Grafting: If a tension-free primary repair is impossible, autologous nerve grafting is the gold standard. The sural nerve is the most common donor. The grafts are reversed to prevent axonal escape through branching points.
Postoperative Protocols
Postoperative management is critical to the success of nerve reconstruction.
1. Immobilization: The limb is typically immobilized in a well-padded splint for 2 to 3 weeks to protect the microscopic repair from tension.
2. Mobilization: Following the initial immobilization phase, a carefully supervised, progressive range-of-motion protocol is initiated.
3. Rehabilitation: Physical therapy focuses on maintaining joint suppleness and preventing contractures of paralyzed muscles. As reinnervation occurs, sensory re-education and motor strengthening protocols are instituted. The patient must be counseled that nerve regeneration occurs at a rate of approximately 1 mm per day (or 1 inch per month), requiring immense patience and compliance with long-term rehabilitation.
===
You Might Also Like