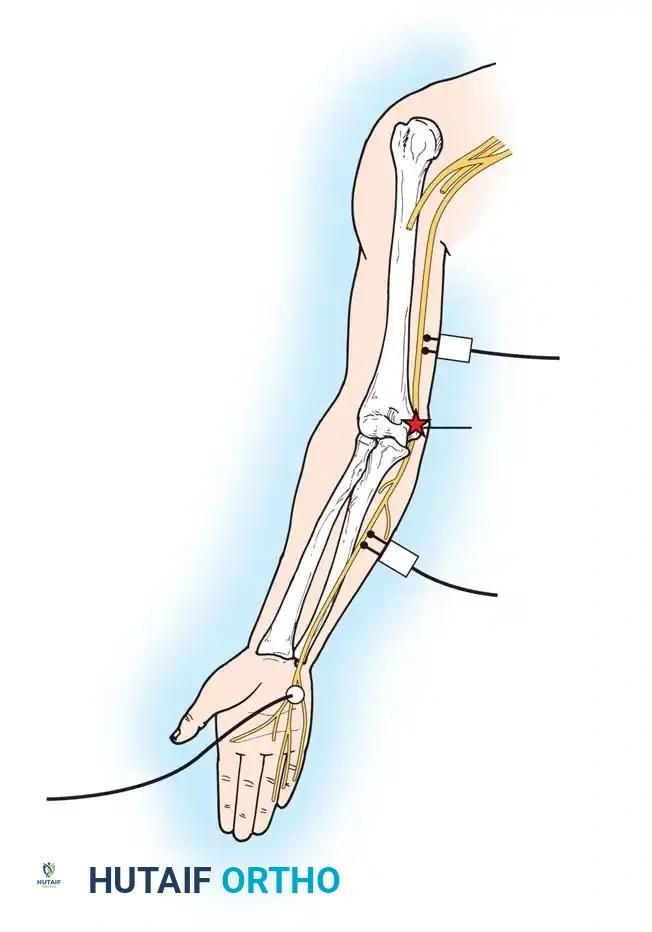
Mastering Endoneurolysis and Peripheral Nerve Repair Techniques
Key Takeaway
Endoneurolysis, or internal neurolysis, is a microsurgical technique utilized to decompress and evaluate intact nerve fascicles encased in intraneural fibrosis. This comprehensive guide details the indications, biomechanics, and step-by-step surgical approaches for endoneurolysis, partial neurorrhaphy, and gap management. Emphasizing meticulous intraneural dissection and tension-free repair, it provides orthopedic surgeons with evidence-based protocols to optimize axonal regeneration and functional recovery in complex peripheral nerve injuries.
INTRODUCTION TO PERIPHERAL NERVE MICROSURGERY
Peripheral nerve surgery demands a profound understanding of microanatomy, neurophysiology, and the biomechanical limits of neural tissue. The management of neuromas-in-continuity, partial nerve lacerations, and critical nerve gaps requires a highly nuanced approach. The overarching goal is to maximize axonal regeneration while minimizing iatrogenic trauma to the delicate vasa nervorum and intact fascicular networks. This comprehensive academic treatise explores the indications, surgical techniques, and postoperative protocols for endoneurolysis, partial neurorrhaphy, and the management of nerve gaps through mobilization, transposition, and advanced grafting techniques.
ENDONEUROLYSIS (INTERNAL NEUROLYSIS)
Endoneurolysis, frequently referred to as internal neurolysis, is the meticulous microsurgical separation of nerve fascicles (funiculi) from surrounding intraneural fibrosis. When an endoneurial exploration is undertaken, the surgeon must operate under the assumption that neurolysis, or partial or complete neurorrhaphy, may ultimately be required. Consequently, preserving as much of the intact epineurium and normal neural architecture as possible is paramount.
Indications and Preoperative Evaluation
Endoneurolysis is indicated when a nerve lesion in continuity demonstrates clinical or electrodiagnostic evidence of incomplete recovery, and intraoperative evaluation reveals dense intraneural scarring compressing otherwise viable fascicles.
Surgical Warning: Distinguishing between pathological intraneural fibrosis and normal physiological plexus formation between fascicles is exceptionally difficult. Overzealous dissection can easily devascularize the nerve or transect vital crossing axons, leading to irreversible iatrogenic deficits.
Step-by-Step Surgical Approach
- Exposure and Epineurotomy: The epineurium is incised longitudinally proximal to the lesion. This incision must begin no more than 0.5 cm from the level of gross pathological changes in the nerve, as determined by careful micro-palpation.
- Limiting the Incision: The incision is strictly limited proximally and distally. Extending the epineurotomy unnecessarily causes the epineurium to become frayed. If a neurorrhaphy subsequently becomes necessary, this frayed tissue must be sacrificed, thereby increasing the nerve gap.
- Flap Retraction: The flaps of the epineurium on each side are undermined widely and retracted laterally using fine 8-0 or 9-0 nylon stay sutures.
- Intraneural Dissection: The funiculi are separated using a pointed or diamond-bladed micro-knife. Sharp or blunt dissection is employed as dictated by the density of the fibrosis. Spring-loaded microscissors are highly advantageous during this phase. The use of high-powered magnifying loupes (minimum 4.5x) or an operating microscope is absolute mandatory to avoid injury to intact nerve tissue.
- Intraoperative Stimulation: Direct nerve stimulation is critical. If most of the fasciculi are intact, can be separated, and trace continuously through the neuroma, nothing further should be done.
- Decision to Resect: If intraoperative stimulation fails to elicit a distal motor or sensory action potential response, and few (if any) intact fasciculi can be identified, the lesion is deemed a non-functional neuroma. Resection of the neuroma followed by complete neurorrhaphy or nerve grafting is indicated.
PARTIAL NEURORRHAPHY
Partial severance of larger nerves—most notably the sciatic nerve and the cords or trunks of the brachial plexus—is a common clinical scenario in high-energy trauma. In such injuries, a partial neurorrhaphy is the gold standard. While occasionally justifiable in smaller nerves, it is technically more demanding and less predictably satisfactory than complete neurorrhaphy.
Decision-Making Matrix
The decision to perform a partial neurorrhaphy is complex and must be made only after exhaustive intraoperative investigation of the lesion.
- Large Nerves: If approximately one-half of a large nerve is disrupted, partial neurorrhaphy is highly advisable.
- Motor vs. Sensory Trade-offs: If the motor response to intraoperative stimulation of the intact portion is robust, it is unwise to risk injury to these functioning motor funiculi merely to restore sensation to a non-critical area (e.g., risking peroneal motor function for dorsal foot sensation, or ulnar motor function for little finger sensation).
- Small Nerves: If the majority of fascicles in a smaller nerve are severed, and stimulation demonstrates no critical function in the remaining few, complete resection and complete neurorrhaphy is generally superior. Suturing isolated, microscopic fascicles in small nerves is often clinically impractical and yields poor results.
Surgical Technique for Partial Neurorrhaphy
When the decision is made to proceed with partial neurorrhaphy, the surgical field must be meticulously prepared to manage the discrepancy in length between the intact and severed fascicles.

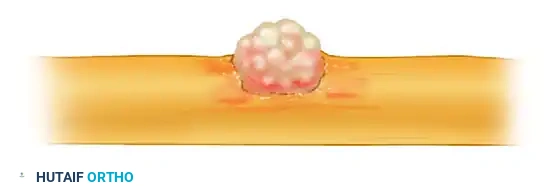
Clinical presentation of a neuroma-in-continuity with partial fascicular disruption. The intact fascicles must be carefully isolated from the fibrotic mass.
- Epineurial Extension: The longitudinal incision in the epineurium is extended proximally and distally for several centimeters to allow adequate mobilization of the individual fascicular groups.
- Fascicular Isolation: The intact funiculi are carefully dissected away from the damaged segment. This requires precise micro-dissection to preserve the vasa nervorum supplying the intact segment.

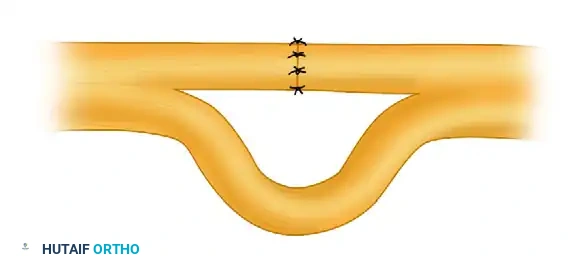
Following excision of the damaged fascicles (neuroma resection), a gap is created in the severed portion, while the intact fascicles are preserved and looped to accommodate the repair.
- Neuroma Excision: The fibrotic neuroma on the severed fascicles is resected back to healthy, pouting axonal tissue (the "mushrooming" effect).
- Tension-Free Repair: The severed ends are brought together. Because the intact fascicles will bow or loop out due to the shortening of the repaired segment, careful spatial management within the nerve bed is required to prevent kinking.
Completion of the partial neurorrhaphy. The severed fascicles are repaired with fine microsutures, while the intact fascicular loop is carefully accommodated within the soft tissue bed to prevent compression.
MANAGEMENT OF THE NERVE GAP
Closing a nerve gap without inducing excessive tension is the most critical biomechanical challenge in peripheral nerve surgery. Tension across a neurorrhaphy site causes intraneural ischemia, promotes aggressive intraneural fibrosis, and ultimately compromises axonal regeneration.
Nerve Stretching and Bulb Suture
Clinical Pearl: Stretching of a nerve at operation by anything more than gentle traction is universally condemned. While gentle traction sufficient to bring the ends together under slight tension does not hinder regeneration, forceful stretching causes immediate ischemic necrosis of the neural tissue.
Mobilization Techniques
Most small gaps can be closed by mobilizing the nerve ends for a few centimeters proximal and distal to the zone of injury. Some degree of mobilization is required in all peripheral neurorrhaphies to achieve healthy tissue margins.
However, the exact amount of mobilization a peripheral nerve can tolerate is finite. Extensive dissection strips the nerve of its segmental mesoneurial blood supply, leading to ischemia.
* Distal Vulnerability: Mobilization has been shown to be significantly more detrimental to the distal nerve segment due to the retrograde nature of its vascular arborization.
* The 2.6 cm Rule: Nicholson and Seddon demonstrated that extensive mobilization adversely affects recovery. In median nerve repairs in the forearm, only 50% of patients achieved M3 motor recovery or better if the gap exceeded 2.6 cm and required extensive mobilization.
When mobilizing, motor and essential sensory branches must be protected. Gaps distal to motor branches are easier to close. For example, the radial nerve branch to the brachioradialis commonly tethers the nerve, preventing gap closure. If the biceps brachii is functioning well, this brachioradialis branch can occasionally be sacrificed to gain length without devastating functional loss.
Critical Nerve Gap Distances
The maximum nerve gap over which mobilization remains a viable option varies by anatomical location. These distances are measured with the extremity in the anatomical position after neuroma excision.
Upper Extremity (Grantham's Values):
* Radial (Midarm): 8.0 cm
* Median (Midforearm, not transposed): 4.5 - 6.5 cm
* Ulnar (Midforearm, not transposed): 3.2 - 5.0 cm
* Posterior Interosseous (Forearm): 1.0 cm
Lower Extremity (Grantham's Values):
* Sciatic (Midthigh): 6.0 - 9.0 cm
* Tibial (Knee): 4.5 - 9.0 cm
* Peroneal (Knee): 6.4 - 8.1 cm
Positioning of the Extremity
Relaxing nerves by flexing joints, abducting, adducting, or rotating the extremity is a powerful adjunct to mobilization.
- Joint Flexion Limits: A joint should never be flexed forcibly to obtain an end-to-end suture. Extreme flexion leads to catastrophic tension when the joint is later mobilized. The strict policy is to flex the knee and elbow no more than 90 degrees, and the wrist no more than 40 degrees.
- Specific Maneuvers: External rotation and abduction are highly effective for repairing radial and axillary nerves (e.g., elevating the shoulder girdle in brachial plexus injuries). Extension of the hip is useful in proximal sciatic injuries.
- Postoperative Protocol: After sufficient wound healing (typically 3-4 weeks), the joint is extended gradually at a rate of approximately 10 degrees per week until full motion is regained.
Surgical Warning: Strong consideration must always be given to nerve grafting in preference to drastic, extreme positioning of an extremity just to force a tension-free primary neurorrhaphy.
Nerve Transposition
Altering the anatomical course of a nerve can significantly shorten the distance between severed ends.
* Ulnar Nerve: Anterior transposition at the elbow is standard for gaining length.
* Median Nerve: Can be transposed anterior to the pronator teres if the lesion is distal to its branches supplying the long flexors.
* Tibial Nerve: Can be routed superficial to the soleus or gastrocnemius if the lesion is distal to the calf muscle branches.
* Radial Nerve: Transposition of the proximal end anterior to the humerus and deep to the biceps yields substantial length. Additionally, simple external rotation of the arm gains length, provided mobilization extends into the axilla and triceps branches are dissected proximally.
Bone Resection
In civilian trauma, bone resection is almost never indicated solely to accomplish a neurorrhaphy. Intact long bones, especially in children, must never be shortened to aid nerve repair.
Bone resection is strictly reserved for cases where the bone (e.g., humerus or femur) is already fractured. If an early delayed nerve suture is performed before the fracture has united, shortening the bone is technically straightforward and highly beneficial for closing large gaps in the ulnar, radial, median, or sciatic nerves. Once the fracture has healed, corrective osteotomy for nerve repair is rarely justified. Both bones of the forearm or leg should never be shortened in the absence of a pre-existing fracture.
BRIDGING THE GAP: GRAFTS AND CONDUITS
When primary repair cannot be achieved without excessive tension, bridging the gap is mandatory. While autografting (e.g., sural nerve, medial antebrachial cutaneous nerve) remains the gold standard, alternative modalities are increasingly utilized.
Nerve Allografts
Fresh nerve allografts offer the potential for functional recovery equivalent to autografts by providing a structurally intact scaffold; however, they require rigorous systemic immunosuppression.
* Immunosuppression Protocol: Tacrolimus (Prograf) is utilized to inhibit T-cell proliferation. It is initiated 3 days preoperatively and maintained for 18 months postoperatively.
* Graft Preparation: Grafts are harvested from ABO-compatible donors and stored at 4°C in University of Wisconsin solution for up to 7 days prior to implantation. Risks include host rejection and opportunistic infections.
Acellularized Nerve Allografts:
Acellularized allografts (e.g., Avance) eliminate the need for immunosuppression by stripping the cellular components while preserving the extracellular matrix scaffold. They are available in diameters of 1 to 5 mm and lengths up to 5 cm.
* Clinical Efficacy: A multicenter retrospective study (ASSH 2010) evaluating 61 nerve repairs demonstrated functional recovery in 88% of patients. Sensory recovery (S3 to S4) was achieved in 91% of cases, while motor recovery (M3 to M5) was achieved in 67%. While autograft remains superior for critical motor nerves, acellularized allografts are an excellent alternative for sensory nerves (digital, common digital) when autograft donor sites are limited or contraindicated.
Synthetic Nerve Conduits
Synthetic conduits provide a protected microenvironment for axonal sprouting across short gaps. Materials include silicone, type I collagen, polyglactin, poly-l-lactic acid (PLLA), polyglycolic acid (PGA), and polyvinyl alcohol (PVA) hydrogel. While highly effective for small, non-critical sensory gaps (typically <3 cm), their use in large mixed-motor nerve defects remains limited compared to the robust scaffold provided by autografts or processed allografts.
CONCLUSION
The surgical management of peripheral nerve injuries via endoneurolysis and partial neurorrhaphy requires exacting microsurgical technique and profound anatomical knowledge. By adhering to strict biomechanical principles—avoiding excessive tension, respecting the vasa nervorum during mobilization, and judiciously utilizing transpositions or grafts—the orthopedic surgeon can optimize the environment for axonal regeneration, thereby maximizing functional recovery for the patient.
You Might Also Like