Double Fascicular & Nerve Transfers in Brachial Plexus
Key Takeaway
The double fascicular transfer, popularized by Mackinnon and Colbert, restores elbow flexion in brachial plexus injuries by transferring redundant motor fascicles from the ulnar and median nerves to the brachialis and biceps branches of the musculocutaneous nerve. This highly reliable technique, often combined with spinal accessory to suprascapular nerve transfers for shoulder abduction, provides robust motor reinnervation while minimizing donor site morbidity through precise intraoperative electrostimulation and tension-free microneural coaptation.
INTRODUCTION TO NERVE TRANSFERS IN BRACHIAL PLEXUS RECONSTRUCTION
The surgical management of upper trunk (C5-C6) brachial plexus injuries has undergone a paradigm shift over the last two decades. Historically, reconstruction relied heavily on supraclavicular exploration and long interpositional nerve grafting. However, the advent of distal nerve transfers has revolutionized outcomes, offering faster reinnervation by bypassing the zone of injury and placing the neurorrhaphy in close proximity to the target motor endplates.
The double fascicular transfer, pioneered and popularized by Mackinnon and Colbert (building upon the foundational single ulnar-to-biceps transfer described by Oberlin), represents the gold standard for restoring elbow flexion. By transferring redundant, synergistic motor fascicles from the intact ulnar and median nerves directly to the biceps and brachialis branches of the musculocutaneous nerve, surgeons can achieve robust, reliable elbow flexion (typically Medical Research Council [MRC] grade M4 or higher) with negligible donor site morbidity.
Furthermore, comprehensive upper trunk reconstruction often necessitates the restoration of shoulder abduction and external rotation. This is frequently achieved concurrently via the spinal accessory nerve (SAN) to suprascapular nerve (SSN) transfer, providing a synergistic functional recovery of the entire shoulder-elbow axis.
INDICATIONS AND PATIENT SELECTION
Careful patient selection is the cornerstone of successful nerve transfer surgery. The double fascicular transfer is highly specific in its indications and requires a thorough preoperative clinical and electrodiagnostic evaluation.
Primary Indications
- Upper Trunk Brachial Plexus Injuries (C5-C6): Classic Erb's palsy presentations where shoulder abduction/external rotation and elbow flexion are lost, but hand and wrist function (C8-T1) remain entirely intact.
- C5-C6-C7 Injuries: Can be considered if the ulnar and median nerve contributions from C8-T1 are clinically and electrodiagnostically robust.
- Timing: Optimal surgical intervention is between 3 to 6 months post-injury. Intervening before irreversible motor endplate degradation (typically occurring at 12–18 months) is critical.
Contraindications
- Lower Trunk Involvement: Any clinical or electromyographic (EMG) evidence of C8-T1 compromise precludes the use of the ulnar or median nerves as donors.
- Late Presentation: Injuries older than 12–18 months may have irreversible muscle atrophy and fibrosis, rendering nerve transfers ineffective. In such cases, free functioning muscle transfers (e.g., gracilis) are indicated.
- Concomitant Peripheral Nerve Injury: Distal injuries to the median or ulnar nerves in the affected limb.
💡 Clinical Pearl: The "Rule of 3s" in Nerve Transfers
For optimal outcomes, nerve transfers should ideally be performed within 3 to 6 months of the injury, utilizing a donor nerve that is at least M4 in strength, and coapted within 3 inches of the target motor endplate to ensure rapid reinnervation (nerve regeneration occurs at approximately 1 mm/day).
SURGICAL ANATOMY AND NEUROPHYSIOLOGY
A profound understanding of the internal topography of the peripheral nerves in the arm is mandatory for safe fascicular dissection.
The Recipient Nerves (Musculocutaneous Branches)
The musculocutaneous nerve arises from the lateral cord (C5-C7). In the proximal arm, it pierces the coracobrachialis and descends between the biceps brachii and brachialis muscles.
* Biceps Branch: Typically branches more proximally.
* Brachialis Branch: Typically branches more distally and lies deeper.
The Donor Nerves (Ulnar and Median Fascicles)
The success of this procedure relies on the redundancy of the motor fascicles supplying the extrinsic wrist and finger flexors.
* Ulnar Nerve: The motor fascicles supplying the flexor carpi ulnaris (FCU) and the ulnar half of the flexor digitorum profundus (FDP) are typically located in the lateral or central portion of the nerve in the mid-arm.
* Median Nerve: The motor fascicles supplying the flexor carpi radialis (FCR), flexor digitorum superficialis (FDS), and palmaris longus (PL) are typically located on the medial aspect of the nerve.
SURGICAL TECHNIQUE: DOUBLE FASCICULAR TRANSFER (MACKINNON AND COLBERT)
1. Patient Positioning and Preparation
- Position: The patient is placed supine with the operative arm abducted to 90 degrees and externally rotated on a radiolucent hand table.
- Anesthesia: General anesthesia is required. Crucially, long-acting paralytic agents must be avoided to allow for intraoperative electrical nerve stimulation. A short-acting paralytic may be used for intubation but must be fully reversed prior to nerve mapping.
- Tourniquet: A sterile tourniquet is applied high on the arm but is often left uninflated unless brisk bleeding obscures the microsurgical field.
2. Surgical Approach and Exposure
- Make a longitudinal skin incision in the central portion of the medial bicipital groove, extending approximately 10 to 12 cm in the mid-arm.
- Carry the dissection down through the subcutaneous tissue to the deep fascia.
- Incise the deep fascia and open the interval between the biceps anteriorly and the triceps posteriorly.
- Identify the neurovascular bundle. The brachial artery serves as the central landmark.
- Identify the ulnar nerve lying medial to the brachial artery.
- Identify the median nerve lying on the lateral surface of the brachial artery.
- Retract the biceps muscle laterally to expose its deep medial surface. Palpate and identify the musculocutaneous nerve as it courses between the biceps and brachialis.
3. Identification and Preparation of Recipient Nerves
- Trace the musculocutaneous nerve distally to identify its terminal motor branches to the biceps and brachialis muscles.
- Electrophysiological Confirmation: Perform electrical stimulation (typically 2.0 mA) of the biceps and brachialis branches to definitively confirm the lack of motor function (verifying the C5-C6 injury).
- Dissect the biceps and brachialis branches at their points of proximal separation from the main trunk of the musculocutaneous nerve.
- Drape these recipient branches medially toward the ulnar and median nerves to assess excursion and plan the coaptation sites.
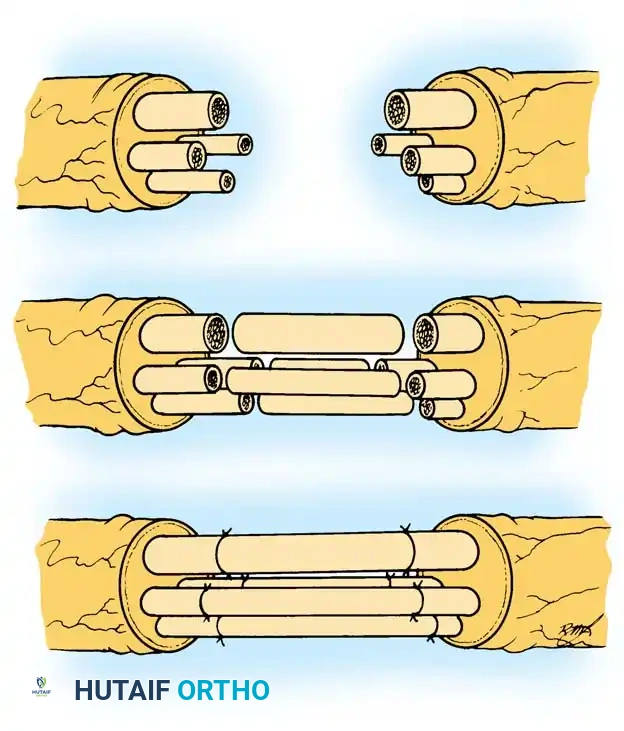
4. Donor Fascicle Selection and Internal Neurolysis
Donor fascicle selection is dictated by anatomical proximity to the recipient nerves to ensure a tension-free repair.
* Typical Configuration: The biceps branch is usually closer to the median nerve, and the brachialis branch is closer to the ulnar nerve. However, anatomical variations exist, and the configuration should be tailored to the specific patient.
* Rule-of-Thumb for Division: To maximize length and prevent tension, divide the donor nerve distally and the recipient nerve proximally.
Ulnar Nerve Dissection (Oberlin Technique Component)
- Incise the epineurium of the ulnar nerve longitudinally over a distance of 2 to 3 cm.
- Perform internal neurolysis under loupe or microscopic magnification.
- Use a handheld nerve stimulator at low intensity (0.5 to 1.0 mA) to precisely distinguish between sensory and motor fascicles.
- Identify a redundant motor fascicle that produces a strong twitch in the extrinsic flexors (FCU). As noted, this fascicle is often located anteriorly and medially (or laterally/centrally depending on the exact level) within the ulnar nerve.
- Separate the chosen fascicle from the rest of the ulnar nerve over 2 cm and divide it at its most distal point.
- Rotate the distal part of the divided fascicle medially toward the recipient brachialis (or biceps) branch.
Median Nerve Dissection
- Similarly, incise the epineurium of the median nerve.
- Identify redundant motor fascicles to the FCR, FDS, or PL (typically on the medial aspect of the nerve).
- Stimulate to confirm robust distal flexion.
- Isolate, divide distally, and transpose the fascicle toward the remaining recipient branch.
🔪 Surgical Warning: Fascicular Mapping
Never divide a donor fascicle without first confirming its motor territory via electrostimulation. Inadvertent division of sensory fascicles (e.g., the sensory component of the median nerve to the hand) will result in devastating sensory loss and potential neuroma formation.
5. Microsurgical Coaptation
- Take the elbow through a full range of motion (extension and flexion) to ensure the planned repair sites will be absolutely tension-free.
- Bring the operating microscope into the field.
- Turn the donor fascicles laterally and coapt them to their respective biceps and brachialis recipients.
- Perform an epineurial/perineurial repair using 3 to 4 interrupted 9-0 or 11-0 nylon sutures. The repair is typically performed anterior to the brachial vascular bundle.
- Augmentation: Add fibrin glue to the repair site. Fibrin glue provides immediate structural stability, seals the coaptation to prevent axonal escape, and reduces the number of sutures required, thereby minimizing foreign body reaction and scarring.
6. Closure
- Ensure meticulous hemostasis.
- Place a closed-suction drain and an indwelling pain pump catheter if dictated by institutional protocol.
- Close the subcutaneous tissues with absorbable sutures and the skin with a subcuticular closure.
CONCURRENT PROCEDURE: SPINAL ACCESSORY TO SUPRASCAPULAR NERVE TRANSFER
In upper trunk injuries, restoring elbow flexion is only half the battle; restoring shoulder abduction and external rotation is equally critical. The transfer of the distal spinal accessory nerve (SAN) to the suprascapular nerve (SSN) is the workhorse procedure for this purpose.
Rationale and Biomechanics
The SAN (cranial nerve XI) innervates the trapezius. By transferring its distal portion (preserving the proximal branches to the upper trapezius to maintain shoulder elevation) to the SSN, surgeons can reinnervate the supraspinatus and infraspinatus muscles. This restores initiation of shoulder abduction and vital external rotation.
Surgical Technique (Posterior Approach)

FIGURE 62-24A: Planned incision for the posterior approach to the suprascapular nerve.
- Positioning: The patient can be in the lateral decubitus or prone position (if performed isolated), though a supraclavicular approach is often used if the patient is supine for the double fascicular transfer. The posterior approach described here offers excellent visualization of the suprascapular notch.
- Incision: A transverse incision is made parallel and just superior to the spine of the scapula.
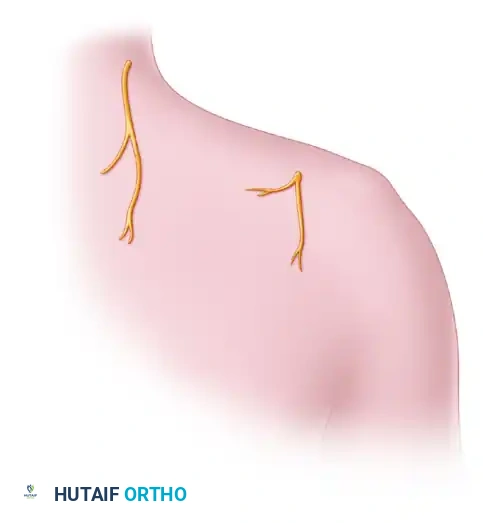
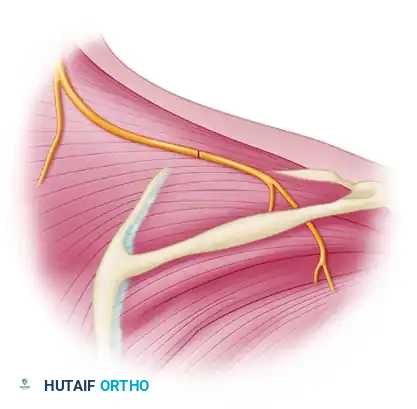
FIGURE 62-24B: Anatomy before nerve transfer. The trapezius is retracted to expose the supraspinatus and the underlying suprascapular nerve.
- Dissection: The trapezius muscle is identified and split or retracted. The distal branches of the spinal accessory nerve are identified on the deep surface of the trapezius.
- The supraspinatus muscle is mobilized to expose the suprascapular notch. The transverse scapular ligament is identified and released to decompress the suprascapular nerve.
- The suprascapular nerve is divided as proximally as possible (often requiring dissection into the supraclavicular fossa) to provide adequate length.
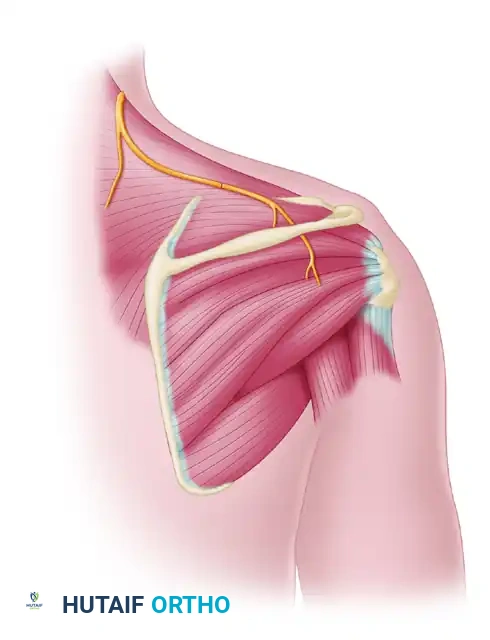
FIGURE 62-24C: Nerve transfer for suprascapular nerve function. The distal spinal accessory nerve is coapted to the distal suprascapular nerve.
- Coaptation: The distal SAN is routed toward the SSN. A tension-free, end-to-end microsurgical coaptation is performed using 9-0 nylon sutures and augmented with fibrin glue.
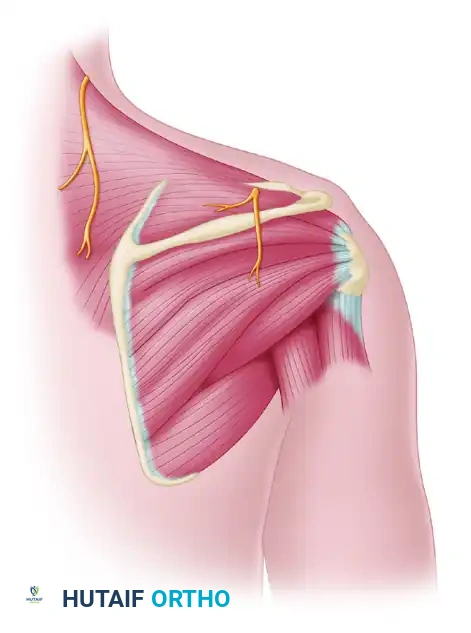
FIGURE 62-24D: Final view of the completed spinal accessory to suprascapular nerve transfer, demonstrating the anatomical relationship between the trapezius, supraspinatus, and infraspinatus muscles.
💡 Clinical Pearl: Suprascapular Nerve Release
Always release the transverse scapular ligament during this transfer. The denervated suprascapular nerve is often tethered at the notch; releasing the ligament prevents postoperative compression of the regenerating axons and provides valuable millimeters of extra nerve length for a tension-free repair.
POSTOPERATIVE CARE AND REHABILITATION PROTOCOL
The success of nerve transfers relies as much on postoperative cortical re-education as it does on microsurgical precision. The brain must learn to fire wrist flexors to achieve elbow flexion.
Phase 1: Protection (Weeks 0–2)
- Immobilization: Immediately postoperatively, the patient’s arm is placed in a shoulder immobilizer or a broad arm sling. The elbow is maintained in approximately 90 degrees of flexion to remove all tension from the nerve repair sites.
- The shoulder immobilizer is strictly maintained for the first 7 to 10 days.
- Wound care and strict elevation are observed to minimize edema.
Phase 2: Gentle Mobilization (Weeks 2–6)
- At 2 weeks, the immobilizer is removed, and gentle, intermittent passive range of motion (ROM) of the elbow and shoulder is initiated.
- Extension of the elbow is achieved gradually to prevent traction injuries to the maturing microneural coaptations.
- Active ROM of the hand and wrist is highly encouraged to maintain the gliding planes of the donor nerves and prevent joint stiffness.
Phase 3: Motor Re-education and Cortical Plasticity (Months 3–12)
- Clinical signs of reinnervation (Tinel's sign advancing along the musculocutaneous nerve, palpable flicker in the brachialis/biceps) typically appear between 3 to 6 months.
- Biofeedback Training: Once a flicker of movement is detected, aggressive motor re-education begins. Patients are instructed to actively flex their wrist and fingers (firing the donor median/ulnar fascicles) while simultaneously attempting to flex their elbow.
- Over time, through neuroplasticity, the brain dissociates the wrist flexion from the elbow flexion, allowing the patient to achieve spontaneous, independent elbow flexion without needing to consciously fire the hand.
Phase 4: Strengthening (Months 6+)
- Once the patient achieves antigravity elbow flexion (MRC Grade M3), progressive resistance training is initiated.
- Strengthening continues for up to 24 months, as muscle hypertrophy and axonal maturation continue to improve functional power.
COMPLICATIONS AND PITFALLS
While highly successful, surgeons must be vigilant to avoid specific technical errors:
- Tension at the Coaptation Site: The most common cause of failure. If tension exists, the limb must be immobilized in greater flexion, or a short interpositional nerve graft must be used (though this significantly downgrades the outcome).
- Incorrect Fascicle Selection: Failing to use intraoperative stimulation can lead to the transfer of sensory fascicles, resulting in no motor recovery and painful neuromas.
- Donor Site Morbidity: While rare, excessive dissection or harvesting too many fascicles can lead to transient or permanent weakness in grip strength or wrist flexion. Adhering to the principle of taking only redundant fascicles is paramount.
- Failure of Cortical Integration: A small subset of patients may struggle with the cognitive remapping required to fire the transferred nerves. Early and persistent involvement of specialized occupational hand therapists is critical to mitigate this risk.
📚 Medical References
- double fascicular transfer for elbow fl exion, J Hand Surg 30A:978, 2005.
- Matsuzaki H, Yoshizu T, Maki Y, et al: Long-term clinical and neurologic recovery in the hand after surgery for severe cubital tunnel syndrome, J Hand Surg 29A:373, 2004.
- Muermans S, De Smet L: Partial medial epicondylectomy for cubital tunnel syndrome: outcome and complications, J Shoulder Elbow Surg 11:248, 2002.
- Ogino T, Minami A, Fukuda K: Tardy ulnar nerve palsy caused by cubitus varus deformity, J Hand Surg 11B:352, 1986.
- Parisien S, Kaplan J: A case of recurrent symptomatic dislocation of the ulnar nerve at the elbow, Orthopedics 5:1323, 1982.
- Richmond JC, Southmayd WW: Superfi cial anterior transposition of the ulnar nerve at the elbow for ulnar neuritis, Clin Orthop Relat Res 164:42, 1982.
- Takebe K, Kanbara Y, Mizuno K, et al: Tardy ulnar nerve palsy associated with the isolated dislocation of the head of the radius, Clin Orthop Relat Res 167:260, 1982.
- Tanabu S, Yamauchi Y, Fukushima M: Hypoplasia of the trochlea of the humerus as a cause of ulnar nerve palsy: report of two cases, J Bone Joint Surg 67A:151, 1985.
- Taniguchi Y, Takami M, Takami T, et al: Simple decompression with small skin incision for cubital tunnel syndrome, J Hand Surg 27B:559, 2002.
- Tomaino MM, Brach PJ, Vansickle DP: The rationale for the effi cacy of surgical intervention for electrodiagnostic-negative cubital tunnel syndrome, J Hand Surg 26A:1077, 2001.
- Trail AI: Delayed repair of the ulnar nerve, J Hand Surg 10B:345, 1985.
- Vastamäki PK, Kallio PK, Solonen KA: The results of secondary microsurgical repair of ulnar nerve injury, J Hand Surg 18B:323, 1993.
- Watchmaker GP, Lee G, Mackinnon SE: Intraneural topography of the ulnar nerve in the cubitus tunnel facilitates anterior transposition, J Hand Surg 19A:915, 1994.
- Median Nerve Browett JP, Fiddian NJ: Delayed median nerve injury due to retained glass fragments: a report of two cases, J Bone Joint Surg 67B:382, 1985.
- Dellon AL: Musculotendinous variations about the medial humeral epicondyle, J Hand Surg 11B:175, 1986.
- Dellon AL, Mackinnon SE: Musculoaponeurotic variations along the course of the median nerve in the proximal forearm, J Hand Surg 12B:359, 1987.
- Gessini L, Jandolo B, Pietrangeli A: Entrapment neuropathies of the median nerve at and above the elbow, Surg Neurol 19:112, 1983.
- Hartz CR, Linscheid RL, Gramse RR, et al: The pronator teres syndrome: compressive neuropathy of the median nerve, J Bone Joint Surg 63A:885, 1981.
- Johnson RK, Spinner M, Shrewsbury MM: Median nerve entrapment syndrome in the proximal forearm, J Hand Surg 4:48, 1979.
- Kallio PK, Vastamäki M: An analysis of the results of late reconstruction of 132 median nerves, J Hand Surg 18B:97, 1993.
- Mackinnon SE, Dellon AL: Surgery of the peripheral nerve, New York, 1988, Thieme. Nicholson OR, Seddon HJ: Nerve repair in civil practice: results of treatment of median and ulnar nerve lesions, BMJ 2:1065, 1957.
- Olehnik WK, Manske PR, Szerzinski J: Median nerve compression in the proximal forearm, J Hand Surg 19A:121, 1994.
- Pritchard DJ, Linscheid RL, Svien HJ: Intraarticular median nerve entrapment with dislocation of the elbow, Clin Orthop Relat Res 90:100, 1973.
- Rana NA, Kenwright J, Taylor RG, et al: Complete lesion of the median nerve associated with dislocation of the elbow joint, Acta Orthop Scand 45:365, 1974.
- Rappaport NH, Clark GL, Bora WF Jr: Median nerve entrapment about the elbow, Adv Orthop 8:270, 1985.
- Simesen K, Haase J, Bjerre P: Interfascicular transplantation in medial nerve injuries, Acta Orthop Scand 51:243, 1980.
- Spinner M: The anterior interosseous-nerve syndrome: with special attention to its variations, J Bone Joint Surg 52A:84, 1970.
- Spinner M, Schreiber SN: Anterior interosseous-nerve paralysis as a complication of supracondylar fractures of the humerus in children, J Bone Joint Surg 51A:1584, 1969.
- Swiggert R, Ruby LK: Median nerve compression neuropathy by the lacertus fi brosus: report of three cases, J Hand Surg 11A:700, 1986.
- Wiggins CE: Pronator syndrome, South Med J 75:240, 1982.
- Wolfe JS, Eyring EJ: Median-nerve entrapment within a greenstick fracture: a case report, J Bone Joint Surg 56A:1270, 1974.
- Femoral Nerve Barrington RL: Haemorrhagic femoral neuropathy, Injury 14:170, 1982.
- Worth RM, Kettelkamp DB, Defalque RJ, et al: Saphenous nerve entrapment: a cause of medial knee pain, Am J Sports Med 12:80, 1984.
You Might Also Like