Pediatric Osteomyelitis: Epidemiology, Surgical Anatomy, and Clinical Approach
Key Takeaway
Pediatric osteomyelitis is a serious bone infection in children, often affecting long bone metaphyses. *Staphylococcus aureus* is the leading pathogen. Unique pediatric skeletal anatomy, such as transphyseal vessels in infants and the thick periosteum, profoundly impacts infection spread. Prompt and precise evaluation is critical to mitigate long-term morbidity and preserve growth.
Introduction & Epidemiology
Pediatric osteomyelitis represents a complex and challenging infectious process involving bone and bone marrow, often requiring prompt and precise evaluation to mitigate long-term morbidity. While traditionally considered a rare condition, its incidence appears stable, affecting approximately 1 in 5,000 children annually, with slight variations globally. The acute hematogenous form is the most prevalent, accounting for 85-90% of cases, primarily affecting long bone metaphyses due to their rich, slow-flowing sinusoidal venous channels that predispose to bacterial seeding.
The etiology is predominantly bacterial, with
Staphylococcus aureus
remaining the most common pathogen across all age groups, including Methicillin-Resistant
S. aureus
(MRSA) which dictates specific antimicrobial strategies. Other significant pathogens vary by age:
*
Neonates (<3 months):
Group B
Streptococcus
, Gram-negative bacilli (e.g.,
E. coli
),
S. aureus
. Multi-focal involvement and joint effusion are more common due to the presence of transphyseal vessels.
*
Infants/Young Children (3 months to 5 years):
S. aureus
,
Kingella kingae
(increasingly recognized, especially in the pre-ambulatory child, often subtle presentation, difficult to culture),
Streptococcus pyogenes
(Group A Strep).
*
Older Children (>5 years):
S. aureus
,
Streptococcus pyogenes
, Coagulase-negative staphylococci (especially in implant-related infections).
*
Specific contexts:
Pseudomonas aeruginosa
in puncture wounds through athletic footwear or in immunocompromised children;
Salmonella
species in children with sickle cell disease;
Mycobacterium tuberculosis
in endemic areas or immunocompromised hosts.
Risk factors include preceding trauma, immunosuppression, indwelling catheters, recent surgery, and underlying conditions like sickle cell anemia. A history of preceding upper respiratory tract infection or transient bacteremia is common. Delayed diagnosis or inadequate treatment carries significant risks, including chronic osteomyelitis, pathological fractures, physeal damage leading to growth arrest or angular deformity, and septic arthritis. Therefore, a high index of suspicion and a structured approach to evaluation are critical for optimal outcomes.
Surgical Anatomy & Biomechanics
Understanding the unique anatomical features of the pediatric skeleton is paramount in managing osteomyelitis, as these characteristics dictate the spread of infection and guide surgical intervention.
Unique Pediatric Skeletal Anatomy:
- Metaphyseal Vascularity: The metaphysis of long bones is highly vascularized with a network of tortuous, fenestrated capillaries and venous sinusoids that create an ideal environment for bacterial deposition and microthrombi formation. These terminal capillary loops turn sharply, predisposing to reduced blood flow and vulnerability to bacterial colonization.
-
Physis (Growth Plate):
- Infants (<18 months): Transphyseal vessels are present, connecting the metaphysis to the epiphysis. This allows direct spread of infection from the metaphysis into the epiphysis and subsequently into the adjacent joint, explaining the higher incidence of septic arthritis accompanying osteomyelitis in this age group (e.g., proximal femur, humerus, ankle).
- Children (>18 months): The physis becomes a cartilaginous barrier, typically preventing direct spread of infection from the metaphysis into the epiphysis and joint. However, extensive metaphyseal infection can rupture through the cortex into the subperiosteal space, or spread along the fascial planes to involve adjacent structures.
- Epiphysis: In neonates and infants, epiphyseal ossification centers are small and mostly cartilaginous, making MRI a superior imaging modality for early detection of epiphyseal or joint involvement.
- Periosteum: The pediatric periosteum is thick, robust, and loosely attached to the cortex, particularly in the diaphysis. This loose attachment facilitates the formation of large subperiosteal abscesses, which can strip the periosteum from the bone, compromising cortical blood supply and leading to sequestrum formation or pathological fracture. Conversely, a thick periosteum can contain the infection, preventing rapid soft tissue spread.
- Haversian and Volkmann Canals: These microscopic channels within the cortical bone can serve as pathways for infection spread from the medullary cavity to the periosteum and vice versa, contributing to cortical destruction.
- Joint Capsule: For bones where the metaphysis is intracapsular (e.g., proximal femur, proximal humerus), a metaphyseal infection can easily rupture into the joint space, causing a septic arthritis, even in older children where the physis is a barrier. This is a critical consideration for sites like the hip.
Biomechanical Considerations:
Infection within bone compromises its structural integrity. Pus accumulation creates increased intraosseous pressure, which can reduce local blood flow and exacerbate bone ischemia, leading to necrosis. Bone destruction from proteolytic enzymes released by inflammatory cells and bacteria weakens the bone, increasing the risk of pathological fracture. The presence of a subperiosteal abscess can compromise periosteal blood supply, leading to cortical necrosis and sequestrum formation, particularly in acute, aggressive infections. Surgical debridement aims to remove infected and necrotic tissue while preserving vital bone and growth centers, thus maintaining biomechanical stability and facilitating healing.
Indications & Contraindications
The decision for operative versus non-operative management in pediatric osteomyelitis is multifactorial, balancing the need for source control with the desire to avoid unnecessary surgical morbidity. Early, prompt evaluation is key to guiding this decision.
Indications for Operative Management:
Surgical intervention is indicated when medical management alone is insufficient to control the infection or when specific complications arise.
*
Abscess Formation:
*
Subperiosteal Abscess:
Large, clearly demarcated abscesses that strip the periosteum from the cortex, particularly when compromising blood supply or causing significant pain.
*
Intraosseous Abscess (Brodie's Abscess):
Chronic, walled-off abscesses that do not respond to antibiotic therapy.
*
Septic Arthritis:
When osteomyelitis extends into an adjacent joint, causing a septic arthritis, surgical drainage of the joint is usually indicated to prevent cartilage destruction.
*
Failure of Medical Management:
Persistent fever, elevated inflammatory markers (ESR, CRP), worsening pain, or lack of clinical improvement after 48-72 hours of appropriate intravenous antibiotic therapy.
*
Radiographic Progression:
Worsening imaging findings (e.g., increased bone destruction, sequestrum formation, developing pathological fracture) despite antibiotics.
*
Atypical or Resistant Organisms:
Infections caused by organisms less susceptible to empiric antibiotic regimens or those forming biofilms (e.g., some Gram-negative bacteria, fungi), where source control is paramount.
*
Chronic Osteomyelitis:
Established chronic infection with sequestrum, involucrum, or sinus tract formation, requiring debridement of necrotic bone and soft tissue.
*
Pathological Fracture:
Infection-induced bone weakening leading to fracture often necessitates debridement and stabilization.
*
Implant-Related Infection:
Osteomyelitis associated with internal fixation devices often requires hardware removal and aggressive debridement.
*
Inconclusive Diagnosis:
If initial aspiration or biopsy for culture is inconclusive and empiric antibiotic therapy is failing, surgical exploration and biopsy may be required to obtain definitive cultures and guide treatment.
Contraindications for Operative Management:
Relative contraindications for immediate surgical intervention include:
*
Early Stage, Responding to Antibiotics:
Clinical improvement (afebrile, decreasing pain, improving inflammatory markers) within 24-48 hours of appropriate IV antibiotics, with no evidence of significant abscess formation or joint involvement on imaging.
*
Small, Contained Fluid Collections:
Very small subperiosteal fluid collections that are not causing significant symptoms and are expected to resolve with antibiotics.
*
Medically Unstable Patient:
Severe comorbidities or critical illness that precludes general anesthesia and surgery, in which case initial stabilization and medical optimization are prioritized. However, even in unstable patients, life- or limb-threatening infections may necessitate urgent drainage once stable enough for anesthesia.
*
Absence of Abscess/Sequestrum:
No evidence of pus collection or necrotic bone on imaging.
Summary of Operative vs. Non-Operative Indications:
| Feature/Parameter | Operative Management | Non-Operative Management |
|---|---|---|
| Clinical Response | No improvement or worsening after 48-72h IV antibiotics | Clinical improvement within 24-48h IV antibiotics |
| Imaging Findings | Subperiosteal abscess >1-2 cm, intraosseous abscess, septic arthritis, sequestrum, involucrum, impending pathological fracture, significant cortical destruction | Small subperiosteal fluid collection, no abscess, no joint effusion |
| Microbiology | Atypical organisms, fungal infections, highly resistant bacteria, implant-related infections | Typical pathogens (e.g., S. aureus ) responding to empiric antibiotics |
| Pain/Swelling | Severe, unremitting pain; rapidly worsening local signs | Decreasing pain and swelling |
| Inflammatory Markers | Persistently elevated or rising ESR/CRP | Rapidly declining ESR/CRP |
| Biopsy/Culture | Indicated for definitive diagnosis if aspirate is negative/inconclusive, or if medical treatment fails | Often initiated after positive aspiration or based on strong clinical suspicion and response to empiric treatment |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential to optimize surgical outcomes, minimize complications, and ensure efficient operating room time.
Pre-Operative Planning:
- Diagnosis Confirmation: Review clinical history, physical exam, laboratory results (CBC, ESR, CRP, blood cultures), and imaging studies (plain radiographs, MRI, ultrasound, bone scan). MRI is the gold standard for defining the extent of bone and soft tissue involvement, localizing abscesses, and assessing joint involvement. Ultrasound is useful for detecting subperiosteal fluid collections.
- Microbiology: Review any available culture results from blood or previous aspirations. If cultures are pending, ensure broad-spectrum empiric antibiotics are started, tailored to the patient's age and local epidemiology, with plans to de-escalate based on intraoperative cultures.
- Anesthesia Consultation: Assess the child's general health, airway, and any specific anesthetic risks. Discuss pain management strategies, including regional blocks if appropriate.
- Surgical Site Localization: Precisely identify the involved bone and the location of any abscesses on imaging. Mark the proposed incision site on the skin pre-operatively, referencing anatomical landmarks.
- Tourniquet Planning: For extremity surgery, determine if a tourniquet will be used. While controversial for infection due to theoretical concerns of local ischemia, a tourniquet provides a bloodless field, which can improve visualization and reduce operative time. If used, ensure proper size and pressure.
- Instrument Preparation: Ensure availability of appropriate instruments, including small osteotomes, curettes, drills, bone wax, culture swabs, and copious irrigation solutions.
- Informed Consent: Discuss the procedure, potential benefits, risks (e.g., infection, bleeding, nerve/vessel injury, recurrence, growth disturbance, pathological fracture), and alternative treatments with the parents/guardians.
Patient Positioning:
Patient positioning depends critically on the affected anatomical site to ensure adequate exposure, protect neurovascular structures, and allow for intraoperative imaging if needed.
*
General Principles:
*
Padding:
Ensure all pressure points are well-padded to prevent nerve palsies or skin breakdown.
*
Access:
Position the limb to allow full range of motion, sterile draping for the entire limb, and access for the surgical team.
*
Intraoperative Imaging:
If fluoroscopy or plain radiographs are anticipated, ensure C-arm access and clear fields for imaging.
*
Specific Positions:
*
Proximal Femur (Hip):
Supine position, with the affected hip slightly abducted and externally rotated. A bump under the ipsilateral buttock can facilitate exposure.
*
Distal Femur/Proximal Tibia (Knee):
Supine, with the knee flexed to 90 degrees and supported, or prone for posterior approaches.
*
Tibia/Fibula Shaft:
Supine with the limb freely draped or lateral decubitus, depending on the approach.
*
Proximal Humerus:
Beach chair or supine position with an arm table for support, allowing full shoulder mobility.
*
Distal Humerus/Proximal Ulna/Radius (Elbow):
Supine with an arm table, allowing for flexion and extension.
Prophylactic antibiotics, typically a first or second-generation cephalosporin, should be administered within 60 minutes prior to incision, unless the patient is already on therapeutic antibiotics for the infection.
Detailed Surgical Approach / Technique
The primary surgical goal for pediatric osteomyelitis is to debride all necrotic and infected tissue, drain abscesses, obtain cultures, and preserve healthy bone and vital structures, particularly the physis.
General Principles of Debridement:
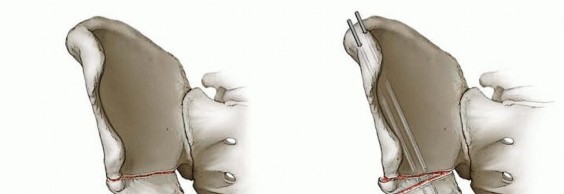
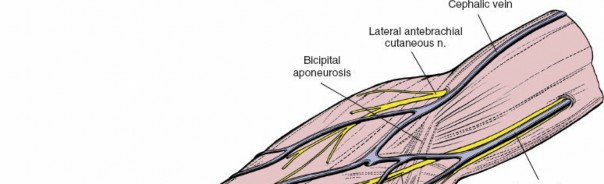
- Incision: A longitudinal incision, carefully planned to allow adequate exposure, avoid neurovascular structures, and align with skin tension lines for optimal wound healing. If an abscess is identified, the incision should be centered over it.
-
Soft Tissue Dissection:
Dissect through subcutaneous tissue and muscle layers, meticulously using sharp and blunt dissection. Identify and protect neurovascular bundles.
- Internervous Planes: Utilize internervous planes whenever possible to minimize muscle damage and functional impairment. For example, the interval between the rectus femoris and tensor fascia lata for an anterior hip approach, or between the tibialis anterior and extensor hallucis longus for an anterolateral lower leg approach.
- Periosteal Elevation: Once the bone is reached, carefully incise the periosteum longitudinally and elevate it subperiosteally to expose the affected bone. If a subperiosteal abscess is present, its drainage will be evident upon incision of the periosteum. Collect pus for aerobic, anaerobic, fungal, and mycobacterial cultures.
-
Cortical Fenestration / Window:
- Drill Holes: For metaphyseal osteomyelitis without overt cortical destruction, multiple small drill holes (e.g., 2.0-3.5 mm) can be made in the cortex overlying the area of infection. This allows for decompression of intraosseous pressure and egress of pus.
- Cortical Window: If a well-localized intraosseous abscess or significant cortical destruction is present, a cortical window may be created using osteotomes or a burr. The window should be large enough for adequate debridement but conservative to preserve bone integrity. The entry point should be carefully selected to avoid the physis.
- Debridement and Curettage: Using small curettes, meticulously debride all necrotic bone, granulation tissue, and pus from the medullary canal and abscess cavity. All devitalized tissue (sequestrum) must be removed.
- Irrigation and Lavage: Copious irrigation with sterile saline solution (pulsatile lavage can be effective) is performed to mechanically remove debris, bacteria, and inflammatory mediators. At least 3-6 liters are typically used for a major bone debridement.
- Tissue Sampling: Multiple deep tissue samples (bone, periosteum, pus) should be sent for Gram stain, aerobic and anaerobic cultures, fungal cultures (if suspected), and histopathology.
- Drainage: A suction drain (e.g., Jackson-Pratt) is often placed into the debrided cavity to prevent hematoma formation and allow continued drainage. The drain is typically removed when output is minimal (<20-30 mL/day).
- Wound Closure: The wound can be closed primarily over a drain, or in cases of extensive contamination or soft tissue swelling, a delayed primary closure or even leaving the wound open for serial debridement may be considered. Skin and subcutaneous tissues are closed in layers.

Figure 1: Intraoperative view during debridement for pediatric osteomyelitis. Note the exposed cortical bone and the presence of purulent material within the medullary canal, which is being meticulously curetted.
Specific Surgical Approaches (Examples):
-
Proximal Femur (Metaphyseal/Neck):
- Approach: Lateral approach (Fahey-Becker variant) through the interval between the gluteus medius and tensor fascia lata. Incision over the greater trochanter extending distally.
- Technique: Expose the proximal femur. If a subperiosteal abscess is present, drain it. For metaphyseal lesions, a small cortical window or multiple drill holes can be made in the lateral cortex of the femoral neck/metaphysis, distal to the physis, to decompress and debride. Careful avoidance of the lateral epiphyseal vessels and physis is crucial. If septic arthritis is present, a separate incision for hip arthrotomy (anterior or anterolateral) is required for drainage.
-
Distal Femur (Metaphyseal):
- Approach: Medial or lateral approach, depending on the location of the abscess. A longitudinal incision over the distal metaphysis.
- Technique: Dissect to the femur, carefully protecting the knee joint capsule and neurovascular structures (popliteal artery/vein posteriorly, common peroneal nerve laterally). Create drill holes or a small cortical window in the metaphysis, distal to the distal femoral physis.
-
Proximal Tibia (Metaphyseal):
- Approach: Anteromedial approach is common, longitudinal incision over the proximal tibia.
- Technique: Dissect to the tibia, avoiding the patellar tendon and anterior neurovascular bundle. Create drill holes or a window in the anteromedial cortex of the proximal metaphysis, distal to the proximal tibial physis.
-
Humerus (Proximal Metaphyseal):
- Approach: Deltopectoral approach for anterior/anteromedial lesions, or posterior approach for posterior lesions.
- Technique: Expose the proximal humerus. Be mindful of the axillary nerve posteriorly and the musculocutaneous nerve anteriorly. Create drill holes or a window in the metaphysis, carefully avoiding the physis and the circumflex humeral vessels.
Post-Debridement Management:
- Antibiotic Beads: In cases of severe chronic osteomyelitis or extensive dead space, biodegradable antibiotic beads (e.g., PMMA loaded with vancomycin or tobramycin) may be considered, but their use in pediatric acute osteomyelitis is less common.
- Wound Closure: Primary closure is preferred once adequate debridement and hemostasis are achieved. If there is significant soft tissue loss or uncertainty regarding infection control, the wound may be left open and managed with negative pressure wound therapy (NPWT) or planned for delayed primary closure.
Complications & Management
Despite meticulous surgical technique and appropriate antibiotic therapy, pediatric osteomyelitis can lead to various complications, ranging from acute surgical issues to long-term sequelae affecting growth and function. Vigilant post-operative monitoring and prompt intervention are critical for effective management.
Acute Complications:
-
Incomplete Debridement/Recurrence (Incidence: 5-10%):
- Management: Persistent fever, pain, or rising inflammatory markers post-operatively warrant re-evaluation with imaging (MRI) and potential repeat surgical debridement and culture. Adjustment of antibiotic regimen based on new cultures.
-
Bleeding/Hematoma (Incidence: <5%):
- Management: Meticulous hemostasis during surgery. Post-operatively, monitor drain output. Significant hematoma may require return to operating room for evacuation and hemostasis.
-
Neurovascular Injury (Incidence: Rare, <1%):
- Management: Prevention through meticulous dissection and knowledge of anatomy. Immediate surgical repair if recognized intraoperatively. Post-operative neurological deficits require urgent evaluation and imaging to determine etiology (compression vs. direct injury).
-
Superinfection/Secondary Infection (Incidence: <5%):
- Management: Rare in the acute phase, but can occur. New signs of infection, especially with a different presentation or different site. Requires new cultures and specific antibiotic therapy.
-
Septic Arthritis (Co-occurrence, not complication, but can be missed):
Often co-occurs, especially in infants. If not adequately drained, can lead to rapid cartilage destruction.
- Management: Prompt surgical drainage and lavage of the affected joint, often simultaneously with osteomyelitis debridement.
Chronic/Late Complications:
-
Chronic Osteomyelitis (Incidence: 5-15%):
Persistent infection despite treatment, characterized by sequestra, involucrum, sinus tracts, and pathological bone remodeling.
- Management: Aggressive and often staged surgical debridement, sequestrectomy, removal of infected involucrum, dead space management (e.g., antibiotic-loaded cement, muscle flaps), and prolonged, culture-specific antibiotic therapy (often 6 weeks IV then 3-6 months oral).
-
Growth Disturbance (Incidence: 5-20% depending on site/severity):
- Physeal Arrest/Damage: Direct damage to the physis from infection, surgical trauma, or hyperemia leading to premature physeal closure, resulting in leg length discrepancy or angular deformity. More common in younger children.
- Management: Regular follow-up with clinical examination and standing radiographs (scanograms). For significant length discrepancies, epiphysiodesis of the contralateral limb, lengthening procedures, or epiphyseodesis of the affected limb may be required. Angular deformities may require osteotomy.
-
Pathological Fracture (Incidence: 2-5%):
Weakening of bone due to infection can lead to fracture, either pre-operatively or post-operatively, especially after aggressive debridement.
- Management: Stabilization of the fracture (casting, bracing, internal fixation) alongside continued infection management (debridement, antibiotics). Healing may be delayed.
-
Soft Tissue Contracture/Scarring (Incidence: Variable):
- Management: Physical therapy, splinting, and in severe cases, surgical release or flap coverage.
-
Joint Stiffness/Degeneration (Incidence: Variable, especially if septic arthritis occurred):
- Management: Early mobilization protocols (once deemed safe), physical therapy. In severe cases of joint destruction, reconstructive surgery may be necessary later in life.
Table of Common Complications and Management:
| Complication | Incidence (%) | Salvage/Management Strategies |
|---|---|---|
| Recurrence / Incomplete Debridement | 5-10 | Repeat debridement, re-culture, MRI, antibiotic adjustment |
| Chronic Osteomyelitis | 5-15 | Staged debridement, sequestrectomy, dead space management, prolonged antibiotics |
| Growth Disturbance (Physeal Arrest/Deformity) | 5-20 (site/age dependent) | Scanograms, contralateral epiphysiodesis, lengthening, osteotomy |
| Pathological Fracture | 2-5 | Fracture stabilization (cast/ORIF), continued infection control |
| Septic Arthritis (Missed/Inadequately Treated) | Often co-occurs, higher in neonates | Urgent surgical drainage, lavage, culture-specific antibiotics |
| Neurovascular Injury | <1 | Immediate repair if recognized, observation, physical therapy |
| Deep Wound Infection / SSI | <2 | Wound opening, debridement, antibiotics, wound vac |
| Soft Tissue Contracture | Variable | Physical therapy, splinting, surgical release |
Post-Operative Rehabilitation Protocols
Post-operative care for pediatric osteomyelitis is critical for optimal recovery, successful eradication of infection, and minimization of long-term sequelae. Rehabilitation protocols are tailored to the individual child, the extent of the infection, the surgical procedure performed, and the stability of the remaining bone.
Immediate Post-Operative Period (Days 0-7):
- Pain Management: Multimodal analgesia, including opioid and non-opioid medications, nerve blocks if performed, to ensure adequate pain control and facilitate early mobilization.
- Antibiotic Therapy: Continue intravenous antibiotics, typically broad-spectrum initially, then tailored based on intraoperative cultures and sensitivities. Duration of IV therapy is usually 2-7 days, depending on clinical response, followed by oral antibiotics.
- Wound Care: Daily wound checks for signs of infection (erythema, swelling, discharge), drain output monitoring. Drains are removed when output is minimal (e.g., <20-30 mL/day). Sterile dressing changes.
- Immobilization: Depending on the extent of debridement and risk of pathological fracture, immobilization (cast, splint, brace) may be used for a short period (1-3 weeks) to protect the bone and promote healing. For simple drainage and stable bone, immobilization may not be necessary.
- Early Mobilization: As soon as pain allows and if bone stability permits, gentle range of motion exercises (active and passive) of adjacent joints are initiated to prevent stiffness and promote circulation.
Subacute Rehabilitation (Weeks 1-6):
- Antibiotic Transition: Once the child is clinically stable (afebrile, decreasing inflammatory markers) and tolerating oral intake, transition to oral antibiotics is typically made. The total duration of antibiotic therapy (IV + oral) is usually 4-6 weeks for acute osteomyelitis, and often longer (3-6 months or more) for chronic or recurrent cases, guided by infectious disease specialists and inflammatory markers.
-
Weight-Bearing / Activity:
- Non-weight-bearing (NWB) or Touch-down weight-bearing (TDWB): For significant bone debridement, pathological fracture, or involvement of major weight-bearing bones (femur, tibia), NWB or TDWB with crutches or walker may be required for 2-4 weeks, or until radiographic evidence of bone healing.
- Protected Weight-Bearing: Gradually progress to protected weight-bearing (partial weight-bearing, PWB) as tolerated, with emphasis on proper gait mechanics.
- Full Weight-Bearing (FWB): Progress to FWB when pain-free, with good bone stability clinically and radiographically.
- Physical Therapy: Focus on restoring range of motion, muscle strength, balance, and proprioception. Therapeutic exercises, functional activities, and gait training are prescribed. Scar management may be initiated.
- Monitoring: Regular clinical follow-up, monitoring of inflammatory markers (ESR, CRP) to ensure infection resolution. Repeat imaging (X-rays) may be obtained at 2-4 weeks to assess bone healing and rule out early complications like pathological fracture.
Long-Term Follow-up (Months to Years):
- Clinical Surveillance: Routine follow-up visits are essential for several years, especially for younger children, to monitor for late complications such as recurrence, growth disturbance (leg length discrepancy, angular deformity), and chronic osteomyelitis.
- Growth Assessment: For children with physeal involvement or very young children, annual clinical evaluation for leg length discrepancy and angular deformity, along with standing plain radiographs (scanograms) as needed, until skeletal maturity.
- Activity Modification: Most children return to full activity without restriction. However, in cases of extensive bone loss or severe physeal damage, certain high-impact sports might need to be modified or avoided to prevent re-injury or fracture.
- Psychosocial Support: A prolonged illness and recovery can be challenging for children and families. Providing appropriate psychosocial support is important.
The goal of rehabilitation is to return the child to their previous level of function and activity while meticulously monitoring for any signs of residual infection or developing musculoskeletal complications.
Summary of Key Literature / Guidelines
Current management of pediatric osteomyelitis is guided by a combination of clinical experience, robust research, and consensus guidelines from major orthopedic and infectious disease societies. Prompt evaluation and a multidisciplinary approach are consistently emphasized.
Diagnosis:
- Clinical Presentation: A high index of suspicion is paramount. Key indicators include localized pain, tenderness, swelling, limping or refusal to bear weight, and systemic signs like fever. However, presentations can be subtle, especially in neonates, infants, and those with Kingella kingae infections.
-
Laboratory Studies:
- White Blood Cell Count (WBC): Often elevated, but can be normal in up to 50% of cases, especially early or with Kingella kingae .
- Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP): These acute phase reactants are consistently elevated and are crucial for both diagnosis and monitoring treatment response. CRP typically rises rapidly and normalizes quickly (days to 1-2 weeks), while ESR rises slower and normalizes over weeks to months.
- Blood Cultures: Positive in 30-60% of cases. Obtain before initiating antibiotics if possible.
- Aspiration/Biopsy: Definitive diagnosis requires isolation of the pathogen from bone or pus. Image-guided aspiration (ultrasound or fluoroscopy) of subperiosteal fluid, joint effusions, or direct bone biopsy is recommended when blood cultures are negative and the diagnosis is uncertain or empiric therapy is failing.
-
Imaging:
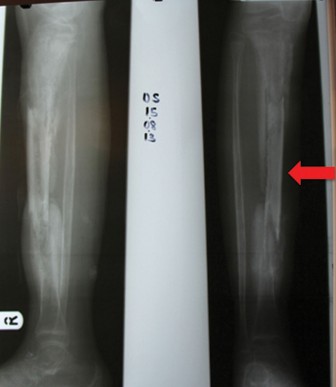
- Plain Radiographs: Often normal in the first 7-10 days. Later findings include periosteal reaction, osteopenia, and bone destruction. Useful for baseline and monitoring for pathological fracture or chronic changes.
- Ultrasound: Excellent for detecting subperiosteal fluid collections, joint effusions, and guiding aspiration, particularly in superficial sites.
- Magnetic Resonance Imaging (MRI): The gold standard for early diagnosis, defining the extent of bone and soft tissue infection, identifying abscesses (subperiosteal, intraosseous, intramuscular), and assessing joint involvement. Gadolinium enhancement further delineates areas of inflammation.
- Bone Scintigraphy (Technetium-99m): Highly sensitive (90-95%) but less specific. Useful for multifocal disease screening or when MRI is contraindicated/unavailable.
Antibiotic Therapy:
-
Empiric Coverage:
Initial empiric antibiotics are critical and should cover common pathogens based on age and local epidemiology, primarily
S. aureus
(including MRSA) and
Kingella kingae
.
- For presumed S. aureus , vancomycin or clindamycin are often used for MRSA coverage, or oxacillin/cefazolin for MSSA.
- For Kingella kingae , beta-lactams (e.g., ampicillin, cefazolin) are generally effective.
- Duration: Typically 4-6 weeks total (IV followed by oral) for acute osteomyelitis without complications. Chronic or recurrent osteomyelitis may require longer durations (months). Transition from IV to oral therapy is based on clinical improvement and normalizing inflammatory markers. Oral bioavailability of chosen antibiotics is paramount.
Surgical Intervention:
- Timing: Indicated for abscess drainage, failure of medical therapy, or when septic arthritis coexists. The decision should be made promptly if criteria are met. Delay in drainage of abscesses or septic joints can lead to significant morbidity.
- Technique: Debridement involves removal of necrotic tissue, aspiration of pus, and copious irrigation. Preservation of the physis is a primary concern in pediatric surgery.
Key Guidelines and Literature:
- Infectious Diseases Society of America (IDSA): Publishes guidelines for the diagnosis and management of bone and joint infections, which offer comprehensive recommendations for pediatric cases.
- Pediatric Orthopaedic Society of North America (POSNA) and American Academy of Orthopaedic Surgeons (AAOS): These organizations provide clinical practice guidelines and educational resources emphasizing early diagnosis, appropriate imaging, and timely surgical intervention when indicated.
- Emphasis on Kingella kingae : Recent literature highlights the increased recognition of Kingella kingae as a cause of osteomyelitis and septic arthritis in young children, often presenting subtly and requiring specific PCR-based diagnostic methods due to its fastidious nature.
In conclusion, the effective management of pediatric osteomyelitis hinges on an acute awareness of its varied presentations, prompt and accurate diagnostic workup, judicious use of imaging, targeted antimicrobial therapy, and timely surgical intervention when indicated. A multidisciplinary team approach involving pediatric orthopedic surgeons, infectious disease specialists, and radiologists is crucial for optimizing outcomes and minimizing the long-term impact on the growing child.
Clinical & Radiographic Imaging

You Might Also Like