Capitellar OCD: Etiology, Diagnosis, & Optimized Treatment Strategies for Athletes
Key Takeaway
Osteochondritis dissecans (OCD) of the capitellum is a subchondral bone necrosis primarily affecting adolescent athletes due to repetitive microtrauma. It causes elbow pain and stiffness. Treatment involves a comprehensive assessment, with stable lesions often managed conservatively. Unstable or persistent lesions typically require optimized surgical intervention to restore articular congruity and prevent long-term damage.
Optimized Treatment of OCD of the Capitellum: Arthroscopic Insights
Introduction & Epidemiology
Osteochondritis dissecans (OCD) of the capitellum is a significant pathology predominantly affecting the adolescent elbow, particularly in overhead and weight-bearing athletes such as baseball pitchers, gymnasts, and tennis players. This condition represents a subchondral bone necrosis with subsequent cartilaginous involvement, potentially leading to delamination, fragmentation, and free body formation within the joint. The etiology is widely considered multifactorial, involving repetitive microtrauma, often exacerbated by valgus stress and axial compression across the radiocapitellar joint during active elbow extension and pronation, leading to impaired blood supply to the vulnerable anterolateral capitellum. Genetic predispositions and endocrine factors may also play a role, though the primary driver remains biomechanical stress in a skeletally immature elbow.
The incidence of capitellar OCD is increasing, paralleling the rise in organized youth sports specialization and intensity. While exact prevalence figures are difficult to ascertain due to varying diagnostic criteria and populations studied, it is a leading cause of elbow pain, stiffness, and functional limitation in this demographic. Untreated or poorly managed lesions can progress to irreversible articular damage, premature osteoarthritis, and chronic instability, significantly impacting an athlete's career and long-term joint health. Therefore, early diagnosis and a tailored, optimized treatment strategy are paramount to preserving joint congruity and function.
Surgical Anatomy & Biomechanics
A thorough understanding of the elbow's intricate anatomy and biomechanics is fundamental to the successful treatment of capitellar OCD. The capitellum, a spheroidal eminence on the distal humerus, articulates with the concave fovea of the radial head. This articulation, alongside the ulnohumeral joint, forms the primary components of elbow flexion and extension, while the radiocapitellar and proximal radioulnar joints facilitate forearm rotation.
The blood supply to the capitellum is derived primarily from the radial recurrent artery, a branch of the radial artery, and the interosseous recurrent artery, a branch of the posterior interosseous artery. These vessels form an anastomotic network around the elbow, with terminal branches penetrating the subchondral bone. The anterolateral aspect of the capitellum, a common site for OCD lesions, is thought to have a more tenuous blood supply and is subjected to higher compressive forces, predisposing it to ischemic events and subsequent osteonecrosis.
Biomechanically, the radiocapitellar joint experiences significant compressive loads, particularly during activities involving valgus stress, such as throwing. As the elbow extends rapidly, the radial head impacts the capitellum. In throwers, the cumulative effect of these forces, often exceeding physiological limits, can lead to repetitive microtrauma, stress shielding, and compromise of the subchondral vascularity. Concomitant laxity or injury to the ulnar collateral ligament (UCL) can exacerbate valgus instability, further increasing stress on the radiocapitellar articulation. Understanding these forces helps in identifying patients at risk and guiding post-operative rehabilitation. Neurovascular structures, including the ulnar nerve medially, the radial nerve and its posterior interosseous branch laterally, and the median nerve anteriorly, must be meticulously protected during any surgical intervention.
Indications & Contraindications
The decision-making process for managing capitellar OCD is complex and hinges on a comprehensive assessment of the patient's symptoms, activity level, lesion stability, and skeletal maturity. Conservative management is typically the first line of treatment for stable, non-displaced lesions in skeletally immature patients without loose bodies or significant mechanical symptoms. This involves activity modification, rest, bracing, and physical therapy aimed at improving range of motion and strength while protecting the elbow from further stress.
Surgical intervention is indicated when conservative measures fail or when the lesion exhibits characteristics associated with a poor prognosis without intervention. The primary goal of surgery is to restore articular congruity, promote healing of the subchondral bone, and alleviate symptoms, thereby preventing progression to degenerative arthritis.
Operative vs. Non-Operative Indications for Capitellar OCD
| Feature/Indication | Non-Operative Treatment | Operative Treatment |
|---|---|---|
| Symptoms | Mild, intermittent pain, no mechanical symptoms. | Persistent pain despite conservative management, mechanical symptoms (catching, locking). |
| Lesion Stability (MRI) | Stable lesion (Type I/II), intact articular cartilage, no displacement, minimal subchondral edema. | Unstable lesion (Type III/IV), articular cartilage fissuring/fragmentation, displaced fragment, loose bodies, significant subchondral bone changes. |
| Skeletal Maturity | Skeletally immature patients, open physes. | Skeletally mature or nearly mature patients, closed/closing physes. |
| Lesion Size/Depth | Small lesions (<10 mm), superficial. | Larger lesions (>10 mm), deep, involving significant subchondral bone. |
| Duration of Symptoms | Acute presentation, short duration. | Chronic symptoms (>3-6 months), progressive worsening. |
| Functional Limitation | Minimal or no impact on daily activities or sports. | Significant limitation in range of motion, strength, or ability to participate in sports. |
| Imaging Findings | Intact cartilage, no loose bodies, minimal subchondral edema. | Fragmented cartilage, loose bodies present, collapse of subchondral bone, cyst formation. |
Contraindications for Surgery:
Absolute contraindications for arthroscopic intervention are rare but include active local or systemic infection. Relative contraindications encompass poor overall health status precluding anesthesia, severe concomitant elbow arthritis where joint preservation is no longer feasible, or unrealistic patient expectations regarding post-operative recovery and return to sport. In situations of a completely resolved lesion without residual symptoms or imaging findings, operative intervention is naturally unwarranted.
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is essential for optimizing surgical outcomes in capitellar OCD. This includes a comprehensive review of all imaging studies and selecting appropriate arthroscopic equipment and patient positioning to facilitate exposure and maneuverability.
Pre-Operative Imaging:
Standard radiographs (anteroposterior, lateral, and oblique views) are crucial for an initial assessment of lesion size, location, and the presence of any gross displacement or loose bodies.
Magnetic Resonance Imaging (MRI) is the gold standard for evaluating capitellar OCD, providing detailed information on articular cartilage integrity, subchondral bone edema, and lesion stability. MRI allows for accurate staging of the lesion, which guides treatment selection.
Computed Tomography (CT) scans with multiplanar reconstructions can be invaluable for precise assessment of bony morphology, fragment size, degree of displacement, and identification of osseous defects, particularly when fixation is contemplated.
Arthroscopic Equipment:
A specialized set of instruments is required for elbow arthroscopy, given the relatively small joint space and critical neurovascular structures surrounding it. The following instruments are essential:
*
30-degree arthroscope:
Standard for general visualization, though a 70-degree scope can be useful for specific areas.
*
2.7-mm arthroscope:
Crucial for working in the tight confines of the direct lateral compartment and accessing challenging posterior lesions.
*
4.0-mm arthroscope:
Routinely used for anterior and posterior compartments. Its larger cannula facilitates superior inflow, joint distension, and subsequent hemostasis, which is critical for clear visualization. Inflow can be managed via the arthroscope's cannula or through a dedicated inflow portal.
*
Self-locking limb positioner:
Devices such as the Arthrex Trimano or Smith & Nephew SPIDER are indispensable. They provide stable elbow positioning, allowing for dynamic adjustments of flexion, extension, and forearm rotation, which significantly improves working space and portal safety.
*
2.9-mm full-radius shaver blade without teeth:
Essential for meticulous debridement of synovium, fibrillated cartilage, and unstable lesion margins without causing excessive tissue damage.
*
Microcurettes:
A variety of sizes and angles allows for precise debridement of the subchondral bed, removal of necrotic bone, and manipulation of cartilage fragments, particularly important for achieving better movement in tight lateral compartment.
*
Small joint obturator and cannulas:
Necessary for safe portal establishment and instrument passage into the confined joint space.
*
Small slotted cannula:
Useful for precise instrument introduction and fragment retrieval.
*
5.5-mm disposable cannula with stop cock:
Provides a reliable conduit for instrument insertion and allows for controlled irrigation.
*
Microfracture awls:
Used to create controlled perforations in the subchondral bone plate, stimulating fibrocartilage formation in situations where debridement and marrow stimulation are indicated.
*
Smooth .062-in Kirschner wire:
Versatile for temporary fragment fixation, marking drill trajectories, and creating pilot holes.
*
Sterile tourniquet:
Applied to the proximal arm to minimize bleeding and optimize visualization, crucial for complex cases.
*
1.6- and 2.0-mm smooth drills:
For creating pilot holes for fixation devices or for microfracture.
*
1.6- or 2.0-mm absorbable nails for osteochondritis dissecans (OCD) fixation:
Absorbable implants (e.g., PLLA, PGA) are commonly used for fragment fixation, with the 1.6 mm size most frequently employed for capitellar lesions due to its smaller profile and adequate strength.
Positioning:
Patient positioning is critical for optimal visualization and access to all compartments of the elbow while ensuring patient and staff safety. Two primary positions are utilized:
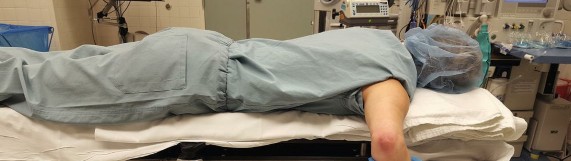
1. Prone Positioning:
This position is generally preferred for patients under 150 lbs, especially those with smaller arms, as it offers several advantages:
* Keeps the patient's body and the operating table away from the smaller arms, creating more working space around the elbow.
* Allows for easy access to both anterior and posterior compartments without repositioning.
* Facilitates the use of a self-locking limb positioner to control the elbow's flexion-extension and pronation-supination.
*

*

*
Figure 21-1 Prone positioning with self-locking limb positioner improves working space in smaller patients.
The elbow is typically flexed to 90 degrees and suspended from an overhead gantry or supported by a limb positioner, allowing for unrestricted movement. The tourniquet is applied to the upper arm.
2. Lateral Decubitus Positioning:
This position is typically preferred for patients over 150 lbs (Fig. 21-2) or when a specific preference exists.
* The patient is positioned on their side with the affected arm draped free and supported by a padded arm holder or limb positioner.
*

*
Figure 21-2 Lateral decubitus positioning for larger patients.
This allows the arm to hang freely, facilitating gravity-assisted joint distension and providing ample space for instrument manipulation. Similar to prone, careful padding is essential to prevent pressure neuropathies. The elbow is typically maintained at 90 degrees of flexion with the forearm pronated or supinated as needed.
Regardless of the chosen position, thorough padding of all bony prominences, especially the ulnar nerve at the cubital tunnel and the radial nerve in the spiral groove, is critical. A sterile tourniquet is applied to the proximal arm and inflated after exsanguination.
Detailed Surgical Approach / Technique
Arthroscopic management of capitellar OCD is a precise procedure requiring a systematic approach to ensure thorough assessment and appropriate intervention.
Portal Placement and Diagnostic Arthroscopy
The surgical procedure begins with standard anterior and posterior portal placement under direct visualization or fluoroscopic guidance to avoid neurovascular structures. The elbow joint is distended with saline solution.
- Anteromedial Portal: Typically established first, approximately 1 cm anterior and 1 cm proximal to the medial epicondyle. This portal provides access to the anterior compartment, radiocapitellar joint, and allows for visualization of the capitellar lesion.
- Anterolateral Portal: Located approximately 1 cm anterior and 1 cm proximal to the lateral epicondyle. This portal is the primary working portal for instruments, allowing for debridement, microfracture, and fragment manipulation.
- Direct Lateral Portal: Located just anterior to the lateral epicondyle, it is crucial for optimal visualization and instrumentation of the capitellum, particularly the central and anterolateral aspects where most OCD lesions occur. A smaller 2.7-mm arthroscope is often advantageous here to navigate the tight space.
- Posterolateral Portal: Approximately 2 cm proximal to the olecranon, along the lateral supracondylar ridge. Primarily used for viewing the posterior compartment and removal of posterior loose bodies.
- Posteromedial Portal: Located just medial to the triceps tendon, proximal to the olecranon. Used for viewing posterior structures and accessing the ulnar nerve.
- Trans-triceps Portal: For specific posterocentral pathology, established directly through the triceps aponeurosis.
Diagnostic arthroscopy is performed systematically to evaluate the entire elbow joint. The radiocapitellar articulation is thoroughly inspected for the OCD lesion, assessing its size, location, stability (using a probe), cartilage integrity, and the presence of associated loose bodies. The ulnohumeral joint and proximal radioulnar joint are also evaluated for any concomitant pathology.



Lesion Management Strategies
The choice of surgical technique depends on the classification of the lesion (e.g., Iwase, Baumgarten, Gudas classifications are common) and intraoperative findings regarding fragment stability and cartilage quality.
1. Debridement and Marrow Stimulation (Microfracture/Abrasion Arthroplasty):
This approach is indicated for stable lesions, particularly those with softened or fibrillated cartilage without gross displacement, or after removal of an unsalvageable fragmented lesion where a defect remains.
* Using a 2.9-mm full-radius shaver blade, unstable articular cartilage is debrided to a stable rim.
* The underlying subchondral bone is then addressed. Necrotic or sclerotic bone within the crater is meticulously removed with microcurettes to expose healthy, bleeding bone.
*

*

* Microfracture is then performed using an awl or 1.6-mm smooth drills, creating small perforations in the subchondral bone plate, typically 3-4 mm apart and 2-4 mm deep. This stimulates the release of mesenchymal stem cells from the marrow, promoting the formation of fibrocartilaginous repair tissue.
*

*

*

2. Fragment Fixation:
This is the preferred approach for unstable but salvageable fragments, especially those with intact or largely intact articular cartilage, aiming to preserve the native hyaline cartilage.
* The fragment is carefully mobilized and elevated. The base of the lesion and the undersurface of the fragment are debrided of any necrotic tissue using microcurettes, ensuring a healthy, bleeding bed.
*

*

* The fragment is then reduced precisely into its anatomical position. Temporary fixation with a smooth .062-in Kirschner wire can be helpful to maintain reduction.
*

* Permanent fixation is achieved with absorbable nails (e.g., PLLA, PGA). The number and trajectory of nails depend on fragment size and configuration. Typically, 1.6-mm nails are used, drilled perpendicular to the articular surface into the subchondral bone. For larger fragments, 2.0-mm nails or even headless compression screws can be considered, ensuring countersinking below the articular surface.
*

*

*

*

*

*

*

*

*

*

*

*

*

*

3. Osteochondral Autograft Transfer System (OATS) / Allograft:
For large, unfixable fragments or significant chondral defects, particularly in older adolescents or skeletally mature patients, restoration with an osteochondral graft may be considered.
* This involves harvesting osteochondral plugs from a less weight-bearing area (e.g., ipsilateral or contralateral knee trochlea, non-articular portion of the elbow).
* The defect on the capitellum is prepared, and the plugs are then press-fit into the defect, aiming for precise cartilage-to-cartilage match.
* Allograft options are available when autograft harvest is limited or unsuitable.
4. Particulated Juvenile Articular Cartilage (DeNovo NT) or Autologous Chondrocyte Implantation (ACI):
These advanced biological resurfacing techniques are typically reserved for larger, focal chondral defects without significant subchondral bone loss, and often in older adolescents or young adults. ACI is a two-stage procedure involving chondrocyte harvest, expansion, and subsequent implantation. DeNovo NT involves direct implantation of particulated juvenile cartilage. These techniques aim for true hyaline cartilage regeneration.
5. Loose Body Removal:
Any identifiable loose bodies, whether cartilaginous or osteochondral, must be meticulously identified and extracted from all compartments of the elbow. These can cause mechanical symptoms, further cartilage damage, and impede joint function.
Upon completion of the chosen procedure, the joint is thoroughly irrigated to remove any debris. The tourniquet is deflated, and hemostasis is ensured. Portals are closed with sterile dressings.
Complications & Management
While arthroscopic treatment of capitellar OCD is generally safe, several potential complications can arise. Understanding these risks, implementing preventive measures, and having strategies for management are crucial.
| Complication | Incidence | Prevention Strategies | Salvage Strategies / Management |
|---|---|---|---|
| Nerve Injury | 0.5-5% | Meticulous anatomical knowledge; precise portal placement using known safe zones; blunt dissection to joint capsule; careful use of electrocautery; maintaining joint distension to push neurovascular structures away. | Observation for neurapraxia; neurolysis for persistent symptoms; nerve repair/grafting for transection (rare); early referral to hand/peripheral nerve surgeon. |
| Elbow Stiffness/Loss of ROM | 5-20% | Early initiation of passive and active range of motion exercises post-operatively; appropriate pain management; avoiding prolonged immobilization; addressing pre-existing stiffness. | Aggressive physical therapy; static progressive or dynamic splinting; manipulation under anesthesia (MUA); arthroscopic or open arthrolysis for severe, recalcitrant cases. |
| Infection | <1% | Strict aseptic technique; prophylactic antibiotics; thorough joint irrigation; meticulous wound care. | Joint aspiration and culture; empiric broad-spectrum antibiotics; arthroscopic lavage and debridement; open debridement for resistant cases. |
| Failure of Fixation/Non-union | 5-15% | Appropriate patient selection for fixation; meticulous fragment preparation and reduction; stable fixation construct; adequate post-operative protection; addressing mechanical factors contributing to non-union (e.g., persistent valgus stress). | Revision fixation (stronger hardware, bone grafting); fragment removal and debridement/microfracture; osteochondral autograft/allograft; biological resurfacing. |
| Hardware-Related Issues | Variable (pins) | Ensure absorbable pins are fully countersunk below articular surface; use appropriate size and material; regular post-operative imaging to monitor absorption and integrity. | Symptomatic hardware removal (if accessible and causing irritation/synovitis); addressing associated pain or mechanical symptoms. |
| Recurrence of Symptoms/Lesion Progression | 5-10% | Optimized surgical technique; strict adherence to rehabilitation protocol; activity modification; addressing underlying biomechanical deficiencies; appropriate patient selection for lesion type. | Re-evaluation with advanced imaging; revision surgery (e.g., conversion from debridement to OATS/biological resurfacing, revision fixation, fragment removal). |
| Heterotopic Ossification | Rare (elbow specific) | Judicious use of electrocautery; careful soft tissue handling; early range of motion; prophylactic NSAIDs or radiation in high-risk patients (though rarely needed for routine OCD). | Excision of mature heterotopic bone after 6-12 months of maturation, coupled with a robust rehabilitation program and prophylaxis. |
| Post-Operative Hemarthrosis/Effusion | Uncommon | Thorough hemostasis at end of case; careful management of anticoagulation if applicable. | Aspiration for diagnostic and therapeutic purposes; compression dressing; rest; activity modification. |
Post-Operative Rehabilitation Protocols
A structured and progressive rehabilitation protocol is paramount for achieving optimal outcomes following arthroscopic treatment of capitellar OCD. The protocol must be individualized based on the surgical procedure performed (e.g., debridement, microfracture, fixation, OATS), lesion size, patient age, and skeletal maturity. The general principles aim to protect the healing tissue, restore range of motion, improve strength, and facilitate a safe return to activity.
Phase I: Protection and Early Motion (0-4 weeks)
- Goal: Protect surgical site, control pain and swelling, initiate gentle range of motion.
-
Immobilization:
- For fragment fixation or OATS, a hinged elbow brace is typically used, locked at 30-90 degrees flexion for the first 2-4 weeks to protect the repair.
- For debridement/microfracture, immediate gentle motion is encouraged, sometimes with a brief sling for comfort initially.
- Pain & Edema Control: Ice, elevation, analgesics.
-
Range of Motion (ROM):
- Passive ROM (PROM): Gentle, pain-free PROM within the protected range. For fixation, typically 30-90 degrees flexion. For debridement, a wider arc may be permitted.
- Active-Assistive ROM (AAROM): Initiated as pain allows.
- Forearm Pronation/Supination: Gentle passive and active motion, avoiding excessive stress on the radiocapitellar joint.
- Strengthening: No active strengthening of the elbow. Gentle isometric gripping exercises.
- Weight Bearing: No weight bearing through the affected arm.
Phase II: Gradual Restoration of Motion & Strength (4-8 weeks)
- Goal: Achieve full, pain-free ROM, begin gentle strengthening.
- Brace: Gradually unlock the brace (if used) to allow for increasing arcs of motion. Discontinue brace once sufficient stability and confidence are achieved, typically around 6 weeks.
-
ROM:
- Progress to full, pain-free PROM and AROM.
- Emphasize terminal extension and flexion, but avoid aggressive stretching.
-
Strengthening:
- Initiate gentle isometric exercises for elbow flexors, extensors, pronators, and supinators.
- Progress to light isotonic exercises with low resistance (e.g., 1-2 lb weights) for biceps, triceps, and forearm musculature.
- Scapular stabilization exercises.
- Functional Activities: Begin light activities of daily living.
Phase III: Progressive Strengthening & Return to Activity (8-16 weeks)
- Goal: Restore full strength, power, and endurance; introduce sport-specific movements.
- ROM: Maintain full, pain-free ROM.
-
Strengthening:
- Advance isotonic and eccentric strengthening exercises with increasing resistance.
- Incorporate plyometric exercises for upper extremity.
- Advanced core and scapular stabilization.
- Initiate rotator cuff strengthening.
- Proprioception & Neuromuscular Control: Begin sport-specific drills, initially without resistance or impact. Gradual return to activities like light throwing (long toss) or gentle racquet swings for non-overhead athletes.
- Avoid: High impact, heavy lifting, or activities that produce significant valgus stress.
Phase IV: Return to Sport (4-6 months, or longer for fixation/OATS)
- Goal: Safely return to competitive sport.
-
Criteria for Return to Sport:
- Full, pain-free ROM.
- No tenderness at the surgical site.
- Sufficient strength (typically >90% of contralateral limb).
- Completion of a progressive sport-specific throwing/activity program without symptoms.
- Physician and therapist approval.
- For overhead athletes: Interval throwing programs (ITP) are crucial, starting with light throws and gradually increasing distance, velocity, and number of throws. This phase often takes 2-4 months itself.
- Imaging: While not mandatory, follow-up MRI may be considered at 6-12 months to assess lesion healing and articular surface integrity, especially after fixation or resurfacing procedures.
Important Considerations:
*
Skeletal Maturity:
Skeletally immature athletes may require a more conservative progression, particularly regarding return to high-impact or overhead activities.
*
Lesion Type:
Fragment fixation or OATS procedures generally necessitate a longer and more cautious rehabilitation due to the need for bone healing and graft integration.
*
Individual Variability:
Each patient progresses at their own pace; the protocol serves as a guideline, not a rigid timeline. Consistent communication between the surgeon, physical therapist, and patient is essential.
Summary of Key Literature / Guidelines
The understanding and management of capitellar OCD have evolved significantly, driven by advancements in arthroscopic techniques and a growing body of literature. Current guidelines emphasize an individualized approach based on lesion stability, size, patient age, and activity level.
Classification Systems: Several classification systems guide treatment, with common ones including the Iwase classification (based on fragment stability and displacement), the Baumgarten classification (based on MRI findings of cartilage and subchondral bone integrity), and the Gudas classification. These systems aid in prognosticating outcomes and standardizing treatment approaches, though a universal consensus remains elusive.
Outcomes of Debridement and Microfracture: For stable or early unstable lesions, arthroscopic debridement of unstable cartilage and microfracture of the subchondral bed have demonstrated good to excellent results in a significant proportion of patients, particularly in skeletally immature individuals. Studies report pain relief and return to sport rates ranging from 70% to 90%. However, the long-term durability of fibrocartilage repair tissue compared to native hyaline cartilage remains a concern, with some studies indicating potential for progressive degenerative changes over time. The "microfracture first" approach is often favored for smaller, contained lesions or as a salvage procedure after fragment removal.
Outcomes of Fragment Fixation: Arthroscopic fixation of unstable but salvageable osteochondral fragments aims to preserve the native articular cartilage. Meta-analyses and systematic reviews generally report favorable outcomes with high rates of fragment union, pain relief, and return to sport. Absorbable implants, such as PLLA or PGA nails, are commonly used, minimizing the need for secondary hardware removal. Success rates typically exceed 80%, with better outcomes often seen in younger patients with smaller, less displaced fragments and shorter symptom duration. Early diagnosis and intervention are critical for maximizing the potential for successful fixation.
Outcomes of Osteochondral Autograft Transfer System (OATS) and Biological Resurfacing: For larger defects or failed primary fixation, osteochondral autograft (OATS) or allograft transplantation, as well as biological resurfacing techniques (e.g., particulated juvenile articular cartilage, ACI), offer reconstructive options. OATS has shown promising results in filling large defects with hyaline cartilage-like tissue, though potential donor site morbidity must be considered. While these techniques are more technically demanding and have longer rehabilitation protocols, they represent viable options for restoring articular surfaces and preventing degenerative changes in carefully selected patients. Long-term follow-up studies are still emerging for these advanced techniques in the capitellum.
Return to Sport Considerations: A critical aspect of capitellar OCD management is the safe return to sport. Most literature suggests a cautious approach, with return to sport typically occurring between 6 to 12 months post-surgery, highly dependent on the type of procedure and patient's functional recovery. Adherence to progressive, sport-specific rehabilitation protocols, objective strength and functional testing, and psychological readiness are essential components of return-to-play decisions. Premature return to activity can increase the risk of re-injury or progression of the lesion.
In conclusion, optimized treatment of OCD of the capitellum requires a nuanced approach, integrating comprehensive pre-operative assessment, meticulous arthroscopic technique tailored to the specific lesion characteristics, and a disciplined post-operative rehabilitation program. While arthroscopy offers excellent visualization and minimal invasiveness, long-term success is contingent upon appropriate patient selection, thorough understanding of elbow biomechanics, and vigilant post-operative care. Continued research, particularly on biological augmentation and long-term outcomes of newer techniques, will further refine our management strategies for this challenging condition.
Clinical & Radiographic Imaging







You Might Also Like