Mastering the Anterior Approach to the Cubital Fossa: Anatomy, Indications & Technique
Key Takeaway
The anterior approach to the cubital fossa provides direct surgical access to vital neurovascular structures like the median nerve, brachial artery, and proximal radial nerve, plus the distal biceps tendon. It's crucial for repairing traumatic lacerations, addressing compressive neuropathies, managing acute limb ischemia, and reinserting ruptured biceps tendons, minimizing iatrogenic injury.
Introduction & Epidemiology
The anterior approach to the cubital fossa, while perhaps less frequently utilized than some posterior or medial elbow exposures in routine orthopedic practice, remains an indispensable pathway for addressing critical pathologies involving the neurovascular structures of the elbow. This approach offers direct and efficient access to the median nerve, brachial artery, and the proximal aspect of the radial nerve and its terminal branches, as well as the distal biceps tendon and its radial insertion. Mastery of this exposure is fundamental for any orthopedic surgeon, particularly those specializing in trauma, hand, and upper extremity surgery, given the potential for devastating functional deficits if these vital structures are compromised.
Historically, surgical access to the cubital fossa has evolved from rudimentary dissections to highly refined techniques guided by a profound understanding of three-dimensional anatomy. Despite advancements in imaging, direct visualization often remains paramount for definitive diagnosis and treatment of complex injuries or chronic conditions. The epidemiology of conditions requiring this approach is diverse, ranging from acute traumatic lacerations (e.g., penetrating injuries) to chronic compressive neuropathies, biceps tendon avulsions, and tumor excisions. While not as common for routine fracture fixation as other approaches, its role in managing soft tissue compromise and nerve/vascular repair cannot be overstated. The surgeon must possess a meticulous understanding of the anatomical relationships within this confined space to minimize iatrogenic injury and optimize outcomes.
Surgical Anatomy & Biomechanics
The cubital fossa is a triangular depression located on the anterior aspect of the elbow, representing a critical anatomical crossroads. Its boundaries are defined by:
*
Superiorly:
An imaginary line connecting the medial and lateral epicondyles.
*
Medially:
The lateral border of the pronator teres muscle.
*
Laterally:
The medial border of the brachioradialis muscle.
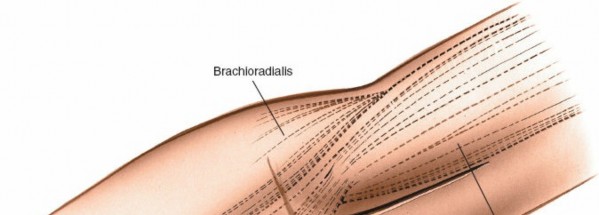
The floor of the fossa is formed by the brachialis (proximally) and supinator (distally) muscles, while the roof is constituted by skin, superficial fascia, and the bicipital aponeurosis (lacertus fibrosus).
Key structures within the cubital fossa, from medial to lateral (deep to the bicipital aponeurosis), are:
*
Median Nerve (MN):
The most medial major neurovascular structure. It lies between the brachial artery and the brachialis muscle, passing obliquely superficial to the brachialis and deep to the bicipital aponeurosis. It then enters the forearm between the two heads of the pronator teres.
*
Brachial Artery (BA):
Lies lateral to the median nerve and medial to the biceps tendon. It bifurcates at the level of the radial neck into the radial and ulnar arteries, although this bifurcation can occur higher or lower. The accompanying brachial veins (venae comitantes) are typically present, as is the basilic vein (superficial to the aponeurosis).
*
Biceps Tendon (BT):
Located lateral to the brachial artery, it inserts onto the radial tuberosity. The bicipital aponeurosis (lacertus fibrosus) extends from the medial aspect of the biceps tendon, superficially crossing the median nerve and brachial artery, to merge with the deep fascia of the forearm, providing protection to underlying structures and an additional origin for forearm flexors.
*
Radial Nerve (RN):
The most lateral of the primary neurovascular structures, it lies deep to the brachioradialis and passes anterior to the lateral epicondyle. Within the cubital fossa, it bifurcates into its two terminal branches:
*
Deep Branch of the Radial Nerve (Posterior Interosseous Nerve, PIN):
Courses posteriorly, passing between the two heads of the supinator muscle (arcade of Frohse) to supply the extensor compartment.
*
Superficial Branch of the Radial Nerve (SRN):
Remains superficial to the supinator, running distally with the radial artery, providing sensation to the dorsum of the hand.
Superficial neurovascular structures requiring careful attention during the approach include:
*
Medial Antebrachial Cutaneous Nerve (MACN):
Supplies sensation to the medial forearm.
*
Lateral Antebrachial Cutaneous Nerve (LACN):
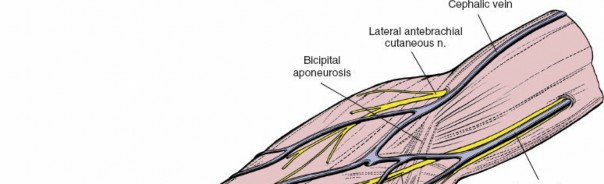
A terminal branch of the musculocutaneous nerve, it supplies sensation to the lateral forearm. It lies close to the cephalic vein.
*
Cephalic and Basilic Veins:
Major superficial veins, often used for venipuncture, which can be encountered during skin incision and superficial dissection.
Biomechanical considerations for the cubital fossa relate primarily to the function of the biceps brachii, forearm pronation/supination, and the integrity of the neurovascular supply to the forearm and hand. Injury to the biceps tendon results in significant loss of supination strength (up to 40%) and elbow flexion strength (20-30%). Disruption of the brachial artery can lead to acute limb ischemia, compartment syndrome, and potential limb loss if not promptly addressed. Nerve injuries (median, radial) can result in profound motor and sensory deficits, severely impacting hand function.
Indications & Contraindications
The anterior approach to the cubital fossa is chosen when direct visualization and access to the neurovascular bundle or distal biceps tendon are paramount.
Indications
-
Acute Trauma:
- Repair of lacerations or complete transections of the median nerve .
- Repair of lacerations or complete transections of the brachial artery , including management of acute thrombosis or pseudoaneurysm formation.
- Repair of lacerations or complete transections of the radial nerve or its superficial/deep branches (PIN, SRN) in the proximal forearm/cubital fossa region.
- Distal biceps tendon rupture: Reinsertion of the biceps tendon into the bicipital tuberosity of the radius. This is a primary indication, particularly for acute ruptures in active individuals.
- Repair of partial lacerations or contusions to the biceps tendon.
- Management of acute compartment syndrome in the forearm originating from cubital fossa pathology requiring neurovascular exploration.
-
Chronic Conditions:
- Release of post-traumatic anterior capsular contractions causing elbow stiffness. This is often performed in conjunction with a medial or lateral approach for complete arthrolysis.
- Excision of benign or malignant tumors originating from or impinging upon structures within the cubital fossa (e.g., lipomas, neurofibromas, sarcomas).
- Decompression of the median nerve (e.g., pronator syndrome, especially when pathology is proximal or recalcitrant to conservative measures) or radial nerve (e.g., PIN compression).
- Vascular procedures such as embolectomy, thrombectomy, or bypass grafting for chronic occlusions affecting the brachial artery.
Contraindications
Absolute contraindications are few, primarily related to the overall patient condition and local soft tissue viability.
*
Severe Local Infection:
Active cellulitis or abscess within the planned surgical field.
*
Severe Soft Tissue Compromise:
Extensive scarring, burns, or poor skin quality that would preclude safe incision and closure.
*
Prior Surgery with Extensive Scarring:
While not an absolute contraindication, significant scarring from previous anterior approaches can increase the difficulty and risk of iatrogenic injury.
*
Unstable Medical Comorbidities:
Patients who cannot tolerate general or regional anesthesia.
*
Alternative, Safer Approach:
In cases where another surgical approach provides superior access with less risk to critical structures (e.g., posterior approach for olecranon fractures, medial approach for ulnar nerve transposition).
Operative vs. Non-Operative Indications
| Condition | Operative Indication | Non-Operative Indication |
|---|---|---|
| Median Nerve Laceration | Complete or partial laceration with functional deficit | Minor contusion without significant deficit, mild paresthesias resolving spontaneously |
| Brachial Artery Laceration/Occlusion | Acute limb ischemia, active hemorrhage, pseudoaneurysm, thrombosis leading to critical hypoperfusion | Minor contusion without flow compromise, stable hematoma (monitor closely) |
| Radial Nerve Laceration | Complete or partial laceration with functional deficit (motor/sensory) | Minor contusion without significant deficit, mild paresthesias resolving spontaneously |
| Distal Biceps Tendon Rupture | Complete rupture in active individuals seeking full strength restoration, acute or chronic (within repair window) | Elderly, sedentary individuals with low functional demands, partial ruptures (selective) |
| Anterior Capsular Contraction | Symptomatic elbow stiffness unresponsive to extensive physical therapy | Mild stiffness responding to physical therapy, absence of functional limitation |
| Cubital Fossa Tumor | Symptomatic tumor (pain, mass effect on nerves/vessels), suspicion of malignancy | Asymptomatic benign lesions, lesions identifiable as non-concerning on imaging (monitor) |
| Pronator Syndrome | Persistent symptoms despite conservative treatment, clear anatomical compression | Mild symptoms, early stages, amenable to rest, NSAIDs, therapy |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential to minimize complications and ensure optimal outcomes.
Pre-Operative Assessment
- Clinical History and Physical Examination: Detailed neurovascular assessment (sensory/motor deficits, pulse examination, capillary refill, specific nerve testing). Document pre-operative deficits accurately. Assess range of motion and pain levels.
-
Imaging Studies:
- Plain Radiographs: Anteroposterior and lateral views of the elbow to rule out associated bony injuries or heterotopic ossification.
- Magnetic Resonance Imaging (MRI): Gold standard for soft tissue evaluation, especially for biceps tendon ruptures, nerve pathology (tumors, compression), and capsular integrity.
- Computed Tomography Angiography (CTA) or Conventional Angiography: Crucial for assessing vascular injuries (lacerations, pseudoaneurysms, thrombosis, pre-operative mapping for chronic occlusions).
- Nerve Conduction Studies (NCS) and Electromyography (EMG): Useful for quantifying nerve injury severity, localizing compression sites, and assessing prognosis in chronic nerve pathologies.
- Patient Education: While strictly doctor-to-doctor, it is imperative that the surgical team understands the potential risks and anticipated outcomes to appropriately discuss with the patient.
- Equipment: Ensure specialized instruments for microvascular repair (if applicable), nerve repair (micro-suture), and biceps tendon reinsertion (suture anchors, drill guides) are available.
Patient Positioning
The standard position for the anterior approach is
supine
on the operating table.
*
Arm Positioning:
The involved arm is placed on a padded arm board, abducted approximately 90 degrees from the torso. The elbow is typically extended to allow for maximal exposure of the cubital fossa, with the forearm supinated to expose the anterior aspect.
*
Head Positioning:
The head can be turned away from the operative field to facilitate surgeon access.
*
Tourniquet Application:
A pneumatic tourniquet is applied high on the upper arm. The limb is exsanguinated either by elevation for 3 to 5 minutes or by applying a soft rubber bandage (Esmarch). Inflation of the tourniquet (typically 250-300 mmHg, or 100 mmHg above systolic blood pressure) provides a bloodless field, crucial for precise neurovascular dissection.
*
Draping:
Standard sterile draping is performed, typically isolating the arm up to the shoulder, allowing full manipulation of the elbow and wrist if needed during the procedure (e.g., for range of motion assessment or tensioning of repairs).
Detailed Surgical Approach / Technique
The anterior approach requires meticulous dissection to protect critical neurovascular structures while achieving the desired exposure.
1. Incision
The initial incision is paramount for providing adequate exposure while minimizing damage to superficial nerves and vasculature.
* Make a curved, "boat-race" (S-shaped or lazy-S) incision over the anterior aspect of the elbow.
* Begin approximately 5 cm proximal to the elbow crease, just medial to the biceps tendon.
* Curve laterally over the elbow crease, extending distally 5 to 7 cm into the proximal forearm, just medial to the brachioradialis.
* This S-shape allows for extensile exposure both proximally and distally, accommodates elbow flexion without excessive skin tension, and places the scar in a less conspicuous location than a straight longitudinal incision.
(Illustration of the "boat-race" incision over the anterior aspect of the elbow, demonstrating its curvilinear path.)
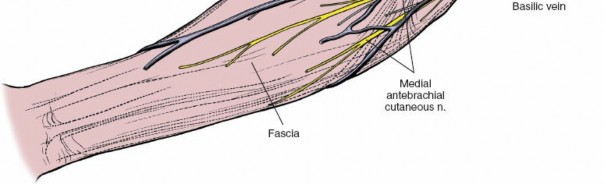
2. Superficial Dissection and Identification of Cutaneous Nerves
- Incise the skin and subcutaneous tissues. Identify and protect the superficial veins (cephalic and basilic veins) and cutaneous nerves.
- The medial antebrachial cutaneous nerve (MACN) typically lies along the medial border of the incision.
- The lateral antebrachial cutaneous nerve (LACN) , a terminal branch of the musculocutaneous nerve, is found laterally, often near the cephalic vein.
- These nerves are highly variable in their course and location; careful blunt dissection and retraction are necessary. If transected, a painful neuroma can develop, thus, protection is paramount.
- Elevate skin flaps carefully, avoiding excessive traction that could damage these delicate structures.

(Visual depiction of the initial skin incision and careful subcutaneous dissection, highlighting the identification and protection of superficial neurovascular structures.)
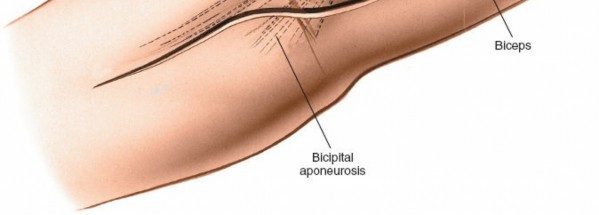
3. Exposure of the Biceps Tendon and Bicipital Aponeurosis (Lacertus Fibrosus)
- The deep fascia is incised longitudinally in line with the skin incision.
- Identify the biceps brachii muscle belly proximally and its distinct tendon distally, running obliquely across the cubital fossa.
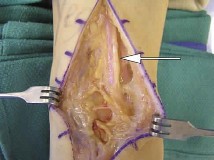
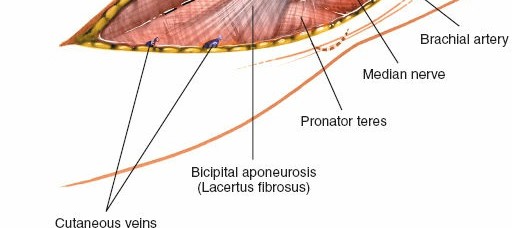
- The bicipital aponeurosis (lacertus fibrosus) originates from the medial aspect of the biceps tendon and extends obliquely over the median nerve and brachial artery, blending with the deep fascia of the forearm. This aponeurosis forms part of the roof of the cubital fossa and must be carefully addressed.

(Anatomical view demonstrating the bicipital aponeurosis (lacertus fibrosus) superficial to the median nerve and brachial artery.)
4. Incision of the Lacertus Fibrosus and Exposure of Deep Structures
- The lacertus fibrosus is incised longitudinally along its medial border, parallel to the underlying median nerve and brachial artery. Extreme caution is warranted to avoid injury to these structures, especially in cases of edema or scar tissue. A small curved hemostat can be carefully passed underneath to elevate it before incision.
- This incision reveals the median nerve and brachial artery, which typically lie directly deep to the aponeurosis.
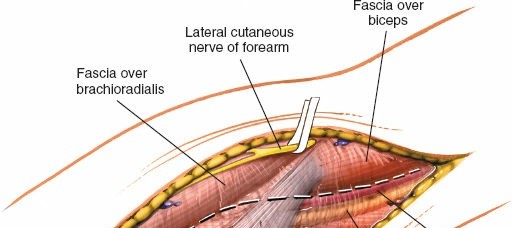
(Surgical view illustrating the careful incision of the bicipital aponeurosis to expose the underlying neurovascular bundle.)
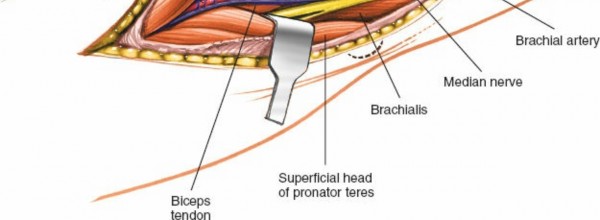
5. Identification and Protection of the Median Nerve and Brachial Artery
- Once the lacertus fibrosus is divided, the median nerve (MN) will be the most medial of the primary structures, often appearing white and cord-like.
- Immediately lateral to the median nerve lies the pulsatile brachial artery (BA) . The venae comitantes often surround the artery.
- These structures are carefully isolated with vessel loops or rubber drains for gentle retraction, allowing access to deeper planes or for repair if injured.
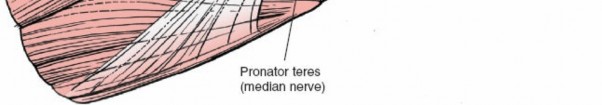
- Proximally, the median nerve and brachial artery are positioned medial to the biceps tendon. Distally, the median nerve passes deep to the pronator teres.

(Close-up image showing the identification and isolation of the median nerve and brachial artery after lacertus fibrosus release.)
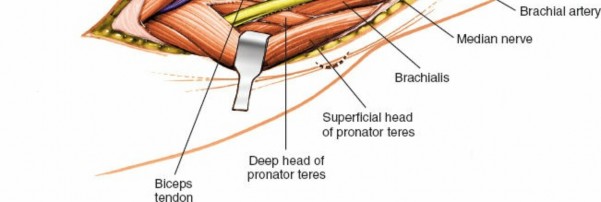
(Another view emphasizing the anatomical relationship between the median nerve and brachial artery within the cubital fossa.)
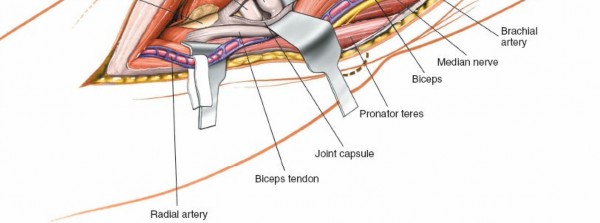
6. Managing the Biceps Tendon
- For access to structures lateral to the median nerve/brachial artery, or for biceps tendon repair, the biceps tendon is retracted.
- In cases of distal biceps tendon rupture, the ruptured tendon end will be identified. The goal is to reinsert it into the bicipital tuberosity of the radius.
- The tendon is typically located by extending the elbow and pronating the forearm.
- If the biceps tendon is intact but obscuring view, it can be gently retracted laterally.
(Image displaying the biceps tendon, possibly retracted, providing a deeper view into the fossa.)
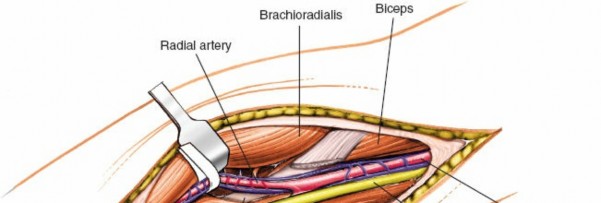
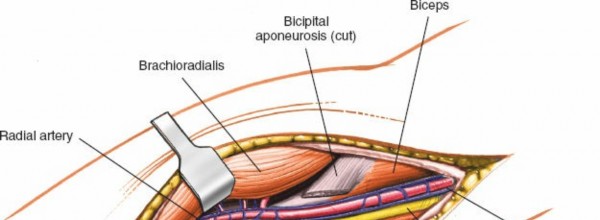
7. Exposure of the Radial Nerve and its Branches
- To expose the radial nerve, the interval between the brachialis (medial) and brachioradialis (lateral) is developed. The biceps tendon is retracted medially.
- The radial nerve (RN) emerges from under the brachialis muscle, deep to the brachioradialis. It bifurcates into the deep branch (PIN) and superficial branch (SRN) typically at the level of the radial head.
- The PIN passes between the two heads of the supinator muscle (arcade of Frohse), which often requires partial release of the supinator origin to decompress or access the nerve.
- The SRN continues distally alongside the radial artery.
- Careful blunt dissection along the lateral border of the cubital fossa is required.
(View demonstrating the exposure of the radial nerve, potentially showing its bifurcation.)
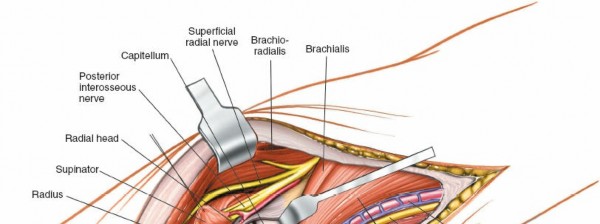
(Deeper dissection revealing the posterior interosseous nerve (PIN) as it enters the supinator muscle.)
8. Specific Surgical Maneuvers (Examples)
a. Distal Biceps Tendon Reinsertion
- Once the ruptured tendon end is identified, it is debrided and prepared.
- The radial tuberosity is identified. This may require pronation of the forearm and retraction of the surrounding muscles (brachioradialis laterally, pronator teres medially).
- A drill hole or tunnel (e.g., using a bicortical drilling technique or a single cortical hole with suture anchors) is created in the radial tuberosity.
-
Various techniques can be employed:
- Single-incision technique: Often uses a cortical button (e.g., Endobutton) or suture anchors, passed through a single anterior incision.
- Two-incision technique (Boyd-Anderson or modified): Utilizes an anterior incision for tendon retrieval and a posterior-lateral incision for radial tuberosity preparation and reattachment, particularly for chronic repairs or when addressing concomitant radial head pathology. The anterior approach described here forms the first part of this.
- Sutures are passed through the tendon and then through the bone tunnel or anchors, securing the tendon firmly to the tuberosity. The elbow is typically flexed to 60-90 degrees during reinsertion to prevent over-tensioning.
(Depiction of the biceps tendon prepared for reinsertion, possibly showing sutures passed through it.)
(Surgical image illustrating the preparation of the radial tuberosity for anchor placement or tunnel drilling.)
(Visual of suture anchors or a bone tunnel being utilized for biceps tendon reattachment.)
b. Nerve Repair
- For lacerated nerves, the epineurium is identified. Debridement of necrotic or severely damaged nerve ends is performed.
- Micro-sutures (e.g., 8-0 to 10-0 non-absorbable) are used to perform epineurial repair or fascicular repair under a microscope, aligning the fascicular bundles.
- Nerve grafts (e.g., sural nerve) may be necessary for large gaps.

(Image demonstrating a nerve being repaired, likely under magnification, with delicate sutures.)
c. Vascular Repair
- For lacerated arteries, control of hemorrhage is achieved proximally and distally.
- Debridement of damaged vessel ends and primary end-to-end anastomosis are performed using microvascular techniques (e.g., 7-0 or 8-0 non-absorbable sutures).
- Vein grafts (e.g., saphenous vein) may be required for significant arterial defects.
- Arterial repair often takes precedence to restore perfusion.
- Consider adjunctive measures such as heparinized saline irrigation and anti-coagulation based on institutional protocols.

(Surgical image depicting vascular repair, showing a vessel anastomosis with fine sutures.)
d. Anterior Capsular Release
- Once the neurovascular structures are identified and protected, the brachialis muscle is carefully elevated from the anterior capsule of the elbow.
- The anterior capsule can then be incised and released, either transversely or in a Z-plasty fashion, to address contracture. Care must be taken to avoid violating the joint capsule excessively to prevent joint instability.
- This is often combined with posteromedial or posterolateral releases for comprehensive arthrolysis.

(Image illustrating a deeper dissection, potentially showing the joint capsule or structures adjacent to it, which might be involved in capsular release.)
9. Closure
- Before closure, ensure meticulous hemostasis. The tourniquet can be deflated, and any bleeding points coagulated.
- Irrigate the wound thoroughly.
- Reapproximate the deep fascia and lacertus fibrosus if possible, ensuring no tension on nerve or vascular repairs.
- Close the subcutaneous layer, then the skin with sutures or staples.
- A sterile dressing is applied, and an appropriate splint or brace is typically used to protect the repair (e.g., 90-degree flexion for biceps repair, or position dictated by nerve/vascular repair tension).
(Final image showing wound closure, highlighting the "boat-race" incision post-suturing.)
Complications & Management
Complications following the anterior approach to the cubital fossa, though often rare with meticulous technique, can be significant due to the proximity of vital structures. Vigilant pre-operative planning and intra-operative care are essential for prevention.
| Complication | Incidence (Approx.) | Salvage / Management Strategy |
|---|---|---|
| Nerve Injury (Median, Radial, Cutaneous) | 1-5% |
Prevention:
Meticulous dissection, precise anatomical knowledge, use of nerve stimulator.
Diagnosis: Post-operative neurological examination, NCS/EMG. Treatment: - Neuropraxia/Axonotmesis: Observation, physiotherapy, pain management. Most resolve spontaneously. - Neurotmesis (Transection): Early surgical exploration and direct repair (epineurial, fascicular) or nerve grafting if a gap exists. - Neuroma: Excision and nerve transposition or grafting, targeted muscle reinnervation. |
| Vascular Injury (Brachial Artery/Veins) | <1-2% |
Prevention:
Careful blunt dissection, vessel loops for retraction, immediate control of hemorrhage.
Diagnosis: Post-operative pulse checks, Doppler, angiography, signs of ischemia. Treatment: - Acute Occlusion/Thrombosis: Immediate re-exploration, thrombectomy, arterial repair (anastomosis or graft), systemic anticoagulation. - Bleeding/Hematoma: Evacuation, ligation of small vessels, re-exploration for major bleeds. |
| Infection | <1% |
Prevention:
Strict aseptic technique, prophylactic antibiotics.
Diagnosis: Clinical signs (erythema, warmth, pain, fever, purulent discharge), elevated inflammatory markers. Treatment: - Superficial: Oral antibiotics, local wound care. - Deep: Surgical debridement, intravenous antibiotics (culture-guided), hardware removal if present and involved. |
| Heterotopic Ossification (HO) | 5-15% (after trauma) |
Prevention:
Prophylactic NSAIDs (e.g., Indomethacin) or low-dose radiation post-operatively, especially in high-risk patients (burns, head injury, extensive trauma).
Diagnosis: Progressive loss of motion, radiographic evidence. Treatment: - Early: Aggressive physical therapy. - Mature HO: Surgical excision (osteotomy) after skeletal maturity, combined with post-operative prophylaxis. |
| Elbow Stiffness/Contracture | 10-20% |
Prevention:
Early, controlled range of motion (CPM), appropriate splinting/bracing, diligent physical therapy.
Diagnosis: Limited range of motion on examination. Treatment: - Early: Intensive physical therapy, dynamic splinting. - Persistent/Severe: Surgical release (capsulotomy, arthrolysis). |
| Re-rupture (Biceps Tendon Repair) | 2-5% |
Prevention:
Strong repair technique, appropriate post-operative immobilization and progressive rehabilitation, patient compliance.
Diagnosis: Sudden pain, palpable defect, weakness. Treatment: - Early: Surgical revision, often with augmentation (e.g., allograft). - Late/Chronic: Reconstruction with graft material. |
| Painful Scar/Neuroma | Variable |
Prevention:
Careful incision planning, gentle tissue handling, precise skin closure.
Diagnosis: Localized pain, tenderness, altered sensation at scar site. Treatment: - Scar: Scar massage, silicone sheeting, corticosteroid injections. - Neuroma: Local anesthetic injections, surgical excision and burying the nerve ending into muscle or bone (target muscle reinnervation). |
| Compartment Syndrome (Forearm) | Rare |
Prevention:
Careful hemostasis, judicious fluid management, awareness in trauma.
Diagnosis: Clinical signs (pain out of proportion, pallor, paresthesias, pulselessness, paralysis), elevated compartment pressures. Treatment: Emergent fasciotomy. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is crucial for achieving optimal functional recovery and varies significantly based on the specific pathology addressed. General principles include protection of the repair, early controlled motion, progressive strengthening, and eventual return to activity.
General Principles
- Protection: The primary goal in the early phase is to protect the surgical repair from excessive stress. This typically involves immobilization in a brace or splint.
- Pain Management: Adequate pain control is essential to facilitate patient participation in therapy.
- Edema Control: Elevation and gentle compression can help minimize swelling.
- Early Motion (Controlled): Once deemed safe, initiation of controlled passive and active-assisted range of motion (PROM/AAROM) prevents stiffness and promotes healing.
- Progressive Strengthening: Gradually introduce isometric, then isotonic exercises as healing progresses.
- Functional Return: Tailor exercises to mimic occupational or recreational activities.
Specific Rehabilitation Examples
1. Distal Biceps Tendon Repair
-
Phase I (Weeks 0-4: Protection and Controlled Motion)
- Immobilization: Long arm posterior splint or hinged elbow brace locked at 90 degrees flexion and neutral rotation for 1-2 weeks. Progress to hinged brace allowing 30-90 degrees flexion, neutral pronation/supination.
- Motion: Gentle passive elbow flexion/extension (within limits), passive forearm supination/pronation (avoid resisted supination).
- Activity: No lifting, pushing, pulling. Avoid active elbow flexion/supination.
-
Phase II (Weeks 4-8: Progressive Motion and Early Strengthening)
- Motion: Gradually increase elbow ROM (full extension by 6-8 weeks). Begin active-assisted and active ROM for elbow and forearm.
- Strengthening: Initiate gentle isometric elbow flexion and supination. Introduce light resistance exercises for wrist and hand.
-
Phase III (Weeks 8-16: Advanced Strengthening and Return to Activity)
- Strengthening: Progress to progressive resistance exercises for elbow flexion, extension, pronation, and supination. Introduce functional grip and pinch strengthening.
- Activity: Gradual return to light activities. Avoid heavy lifting or sport-specific activities.
-
Phase IV (Weeks 16+ / Month 4-6: Full Return to Activity)
- Progress to sport-specific or work-specific training. Full return to activities typically at 4-6 months, once strength is symmetrical and pain-free.
2. Nerve Repair (Median or Radial Nerve)
-
Phase I (Weeks 0-3/4: Immobilization and Protection)
- Immobilization: Splinting in a position that reduces tension on the nerve repair (e.g., elbow flexed, wrist neutral for median nerve; wrist extended for radial nerve).
- Motion: Gentle passive range of motion of adjacent joints (shoulder, hand) to prevent stiffness, carefully avoiding tension on the repaired nerve.
- Sensory Re-education: Begin desensitization techniques and protective sensation education.
-
Phase II (Weeks 4-12: Gentle Motion and Sensory Re-education)
- Motion: Gradually increase passive and active range of motion, ensuring no pain or nerve tension.
- Motor Re-education: Initiate gentle active muscle contraction for reinnervated muscles, often guided by EMG biofeedback.
- Sensory Re-education: Progress with texture discrimination, localization, and stereognosis.
-
Phase III (Weeks 12+: Strengthening and Functional Integration)
- Strengthening: Progressive resistance exercises as motor return is evident.
- Functional Training: Integrate hand and arm into daily activities, focusing on coordination and dexterity.
- Splinting: Dynamic splinting may be used to assist weak muscles or prevent contractures.
3. Vascular Repair
-
Phase I (Weeks 0-2: Strict Immobilization and Monitoring)
- Immobilization: Splinting to prevent elbow motion and protect the vascular anastomosis.
- Monitoring: Frequent neurovascular checks (pulse, Doppler, capillary refill, sensation, motor function).
- Medications: Anticoagulation or antiplatelet agents as per surgeon's protocol.
-
Phase II (Weeks 2-6: Gradual, Protected Motion)
- Motion: Begin gentle passive and active-assisted ROM, avoiding extreme positions that could stretch the repair.
-
Phase III (Weeks 6+: Progressive Activity)
- Gradual increase in activity and resistance, as determined by vascular surgeon and therapist.
Summary of Key Literature / Guidelines
The anterior approach to the cubital fossa is a well-established surgical pathway, with its principles deeply rooted in classical anatomical texts and modern surgical literature.
- Anatomical Foundation: Standard anatomical texts such as Gray's Anatomy and Netter's Atlas of Human Anatomy provide the foundational understanding of the cubital fossa. Surgical anatomy texts, such as those by Hoppenfeld and deBoer's Surgical Exposures in Orthopaedics , offer detailed, step-by-step guidance on this specific approach, emphasizing internervous planes and critical landmarks.
-
Distal Biceps Tendon Repair:
The literature consistently supports surgical repair of complete distal biceps tendon ruptures in active individuals to restore strength and endurance. Both single-incision (e.g., suture anchor or cortical button techniques) and two-incision techniques have demonstrated good clinical outcomes.
- Studies by Mazzocca et al. (2007, 2010) have explored the biomechanical advantages of various fixation methods, often favoring cortical button techniques for robust fixation.
- Meta-analyses often show comparable functional outcomes between single and two-incision techniques, with potential differences in complication profiles (e.g., higher rates of sensory nerve issues with two-incision, HO with single-incision, though often technique-dependent). Ramsey et al. (1999) is a seminal paper detailing the anatomy and repair.
-
Nerve Repair:
The principles of microsurgical nerve repair are paramount.
- Sunderland (1978) and Seddon (1943) classifications remain the cornerstone for grading nerve injuries and guiding prognosis.
- Birch et al. (1998) and other micro-neurosurgeons have extensively documented outcomes of direct nerve repair and grafting, emphasizing the importance of tension-free repair and early intervention.
- The literature consistently demonstrates better outcomes for primary repair of sharp lacerations compared to blunt avulsion injuries or delayed repairs.
-
Vascular Repair:
Immediate revascularization is the guiding principle for acute arterial injuries.
- Rich and Spencer (1978) and later publications in vascular surgery provide comprehensive guidelines on the diagnosis and management of peripheral vascular trauma, emphasizing the "golden hour" for revascularization to prevent limb loss.
- Outcomes are highly dependent on the mechanism of injury, associated soft tissue damage, and time to repair.
-
Elbow Contracture Release:
The role of the anterior approach for anterior capsular release is well-documented, often as part of a comprehensive arthrolysis.
- Morrey (2000) and other elbow surgeons have published extensively on the management of elbow stiffness, highlighting the importance of thorough capsular release, prevention of heterotopic ossification, and aggressive post-operative rehabilitation.
-
Guidelines:
While specific universally accepted guidelines for the anterior cubital fossa approach are not uniformly codified due to its diverse indications, general principles from national orthopedic and hand surgery societies advocate for:
- Thorough pre-operative assessment and imaging.
- Meticulous surgical technique with emphasis on neurovascular protection.
- Appropriate post-operative immobilization and rehabilitation tailored to the specific repair.
- Prophylaxis against common complications (e.g., HO, infection).
In conclusion, the anterior approach to the cubital fossa, though requiring detailed anatomical precision, is an invaluable tool for the orthopedic surgeon. Its successful application hinges upon rigorous planning, exacting technique, and a comprehensive understanding of potential complications and structured rehabilitation.
Clinical & Radiographic Imaging

You Might Also Like